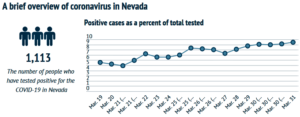
- Coronavirus contextualized: Exploring, through data, COVID-19 in Nevada and beyond (thenevadaindependent.com)
There are lots of numbers swirling around in the time of coronavirus: Confirmed cases of COVID-19, people tested, number of hospitalizations and, increasingly, new deaths...There are also other data points revealing the finer points of how the virus is affecting people, such as the age and gender of those who have tested positive and what pre-existing conditions people hospitalized after contracting the novel coronavirus have...But those numbers can be difficult to parse without context. Below, The Nevada Independent explores that data and puts it into context, walking through what we do and don’t know about coronavirus in Nevada, how Nevada stacks up against other states and projections for the future...READ MORE
- U.S. FDA says malaria drugs in shortage as coronavirus drives up demand (reuters.com)Current and Resolved Drug Shortages and Discontinuations Reported to FDA (accessdata.fda.gov)
The U.S. Food and Drug Administration said malaria drugs hydroxychloroquine and chloroquine are in shortage due to a surge in demand because of the coronavirus pandemic...The drugs, which have been tried with some success to treat the illness caused by the virus, were added to the agency's website that lists drug shortages on Tuesday...Studies are underway in a number of countries to see whether hydroxychloroquine and the related malaria drug chloroquine may be effective in controlling the spread of coronavirus, which has led to a surge in demand for the treatments...READ MORE
- Coronavirus (COVID-19) Update: FDA takes further steps to help mitigate supply interruptions of food and medical products (fda.gov)
During this COVID-19 pandemic, the FDA is working around the clock to make sure that Americans have access to safe food and medical products. The agency is continuously examining the global supply chain to identify any concerns and assess the availability of the products Americans need most. We are also partnering with the Federal Emergency Management Agency (FEMA) on supply chain issues, including importation of needed medical products to support the U.S. response. Here is a status update and details on some of the latest actions we have taken:...READ MORE
Medical Devices
Human Drugs & Biologics and Animal Drugs
Blood Supply
Human and Animal Food
Veterinary Medicine
- 7 healthcare-related items you may have missed in the $2T coronavirus stimulus package (fiercehealthcare.com)Hospitals get $100B in massive stimulus deal as facilities face COVID-19 (fiercehealthcare.com)
Here are seven things you may have missed in the enormous $2 trillion economic stimulus package...includes major requirements for insurers to cover diagnostics and services associated with COVID-19 and gives some flexibility to hospitals...But the bill, which includes massive unemployment assistance and help to businesses, includes several other healthcare provisions...READ MORE
- Requiring Medicare Part D and Medicare Advantage plans to allow for refills and fills for up to three months.
- Extending healthcare programs through November.
- Waiving site-neutral payment cuts, 50% rule for long-term care hospitals.
- Eliminating Medicare sequester and boosting Medicare payments for COVID-19 payments.
- Requiring payers to cover hospital-made tests.
- Publishing the cash price for diagnostic testing.
Requiring group and individual health plans to cover preventive services.
- Gilead fails to overturn $752M CAR-T patent verdict. Will BMS win case for larger penalties? (fiercepharma.com)
Bristol Myers Squibb and Gilead Sciences have been locked in a CAR-T patent fight, with BMS scoring a lucrative victory following a trial late last year. Now, Gilead has failed to persuade a judge to overturn the $752 million verdict—and the company could face bigger damages down the line... Judge James Otero rejected numerous arguments from Gilead’s Kite Pharma unit that patents held by BMS’ Juno Therapeutics are invalid, plus that procedural flaws in the legal process warrant a new trial...READ MORE
- Coronavirus (COVID-19) Update: FDA Continues to Accelerate Development of Novel Therapies for COVID-19 (fda.gov)
As part of the Trump Administration’s all-hands-on-deck approach across public, academic and private sectors to combat the COVID-19 pandemic, the U.S. Food and Drug Administration stood up a new program to expedite the development of potentially safe and effective life-saving treatments. The program, known as the Coronavirus Treatment Acceleration Program (CTAP), is using every tool at the agency’s disposal to bring new therapies to sick patients as quickly as possible, while at the same time supporting research to further evaluate whether these medical countermeasures are safe and effective for treating patients infected with this novel virus.,,READ MORE
- BREAKING, FDA Gives Emergency Authorization Of Trump Touted Drugs To Fight Coronavirus (citizentruth.org)
The Food & Drug Administration has authorized the use of two drugs championed by President Donald Trump as a means to fight coronavirus...On Sunday night the FDA issued an emergency authorization for the use of two anti-malaria drugs, hydroxychloroquine and chloroquine. Researchers in the United States have begun testing the drugs in some states, like New York, but the drug will now be more widely available...The FDA has allowed for the drugs to be “donated to the Strategic National Stockpile to be distributed and prescribed by doctors to hospitalized teen and adult patients with COVID-19, as appropriate, when a clinical trial is not available or feasible,” the department of Health and Human Services said in a statement...READ MORE
- FDA wants heartburn meds off the market due to contamination (yahoo.com)
U.S. health regulators are telling drugmakers to immediately pull their popular heartburn drugs off the market after determining that a contamination issue with the medications poses a greater risk than previously thought...The move from the Food and Drug Administration Wednesday applies to all prescription and over-the-counter versions of ranitidine, best known by the brand name Zantac. The drugs are widely used to treat stomach acid and ulcers...Dozens of recalls have been linked to the same probable carcinogen since last year. The FDA is still investigating the issue and has sanctioned at least one manufacturing plant in India that makes ingredients used in the medications...READ MORE
- In pharma, cancer is king Inside the industry’s quest for its next crowning achievement in oncology (pharmamanufacturing.com)
Pharma’s pursuit of innovative oncology drugs has hit a fever pitch like the industry has never felt. On the heels of a treatment revolution that has fundamentally changed how we understand, fight and, in some cases, can cure cancer, pharma is mobilizing at a dazzling speed to develop the next holy grail in oncology...The current generation of cancer treatments is also still stymied by a number of challenges including manufacturing issues, limited therapeutic benefits for many patients and sky-high prices that have drawn public ire...All told, oncology has become a segment of the industry that is both bursting at the seams and begging for further innovations. Recognizing the potential to help both patients and business, pharma has become obsessed with oncology...READ MORE
- Drug supplies, costs hurt by unintended consequences of COVID-19 policies, suppliers tell White House (fiercepharma.com)
Associations representing generic drug makers, health insurers, pharmacy benefit managers and pharmacies have sent a letter to top administration and congressional leaders laying out how some policies and proposals to fight COVID-19 are making the situation worse...In an unusual display of coordinated frankness for the industry, a coalition representing generic drug makers, insurers, pharmacies and benefit managers told Vice President Mike Pence and congressional leaders that some policies in place or under consideration to fight COVID-19 are making it difficult and more expensive for patients to get some drugs...Signers of the letter are the Academy of Managed Care Pharmacy, America’s Health Insurance Plans, the Association for Accessible Medicines, the Blue Cross Blue Shield Association, the National Association of Chain Drug Stores, the National Association of Specialty Pharmacy, the Pharmaceutical Care Management Association and Pharmaceutical Research and Manufacturers of America...READ MORE