- First Lady Melania Trump discusses opioid abuse in Las Vegas (lasvegasnow.com)BE BEST (whitehouse.gov)
First lady Melania Trump is asking the public to look beyond the raw numbers and to see the opioid crisis as a "human story."...Mrs. Trump says that by thinking of the faces behind the statistics "we have the potential to not just reduce, but eliminate" the tens of thousands of deaths that are attributed to opioids annually. Federal statistics show that nearly 48,000 Americans died in 2017 after overdosing on the powerful painkillers...The first lady spoke Tuesday during a town hall-style conversation in Las Vegas on the opioid epidemic. She is using the event to close a two-day, three-state tour to promote her "Be Best" initiative, which includes a focus on babies born dependent on opioids...READ MORE
- FDA Commissioner Gottlieb, who raised alarms about teen vaping, resigns (washingtonpost.com)
Food and Drug Commissioner Scott Gottlieb...resigned...effective in about a month...The 46-year-old physician, millionaire and cancer survivor known for a self-assured, sometimes brash, manner lives in Westport, with his wife and three daughters – 9-year-old twins and a 5-year-old...“It was a very hard decision,” Gottlieb said in an interview. “This is the best job I will ever have. I’m leaving because I need to spend time with my family. I get home late Friday, work on weekends and come back to Washington on Sunday. I did the job 100 percent."...The resignation took some senior FDA officials by surprise, and came as Gottlieb’s signature issue – youth vaping – is being reviewed by the White House Office of Management and Budget. The plan, detailed by Gottlieb last fall, would sharply restrict the sale of flavored e-cigarettes to curb a surge in underage vaping, which he argues could lead to a whole new generation addicted to nicotine...READ MORE
- Industry Responds to Government Strategy to Improve Services for Rare Diseases (pharmtech.com)
The Association of the British Pharmaceutical Industry has responded to the United Kingdom government’s recently published plans on improvements to services for people with rare diseases...the National Genomic Healthcare Strategy...action points aimed at improving services...
• Every person with a rare disease will have a dedicated person responsible for co-ordinating their care.
• Every patient with a rare disease will be given an ‘alert card’, including information about their condition, treatment regime, and contact details for the individual expert involved in their care.
• Every child with a rare condition will be transferred to appropriate adult services when they reach the age of 18, even if that adult service is not the commissioning responsibility of NHS England.
Additionally, the strategy has set out how the genomics community can work together to make the UK a global leader in genomic healthcare...
- Novartis faces shareholder criticism over drug prices at AGM (reuters.com)
Novartis’s shift into high-tech drugs won praise for providing patients with new options but criticism over prices that may run into the millions of dollars at the Swiss drugmaker’s annual general meeting...Swiss shareholder group Actares said insurance systems are being “taken hostage” by high prices for life-saving drugs. It called out Novartis’s $475,000 cancer cell therapy Kymriah and its still-unapproved gene therapy for spinal muscular atrophy that Novartis contends is cost-effective at $4-$5 million per patient, while independent groups have concluded its value is less than that.
- Lawmakers hear bill to ban rules barring pharmacists from telling patients about less expensive, generic drugs (thenevadaindependent.com)
The legislation, which GOP Assembly members Melissa Hardy and Glen Leavitt presented to the Assembly Commerce and Labor Committee on Monday, would bar pharmacy benefit managers, or PBMs, from stopping a pharmacist from telling patients about the availability of a less expensive, generic or more effective drug, or a less expensive manner of purchasing a drug. The proposal builds upon a pharmacy gag ban passed by the Legislature in 2017 that prevented PBMs from blocking pharmacists from sharing information about the copay or coinsurance for a prescription drug or the clinical efficacy of a less expensive alternative drug...READ MORE
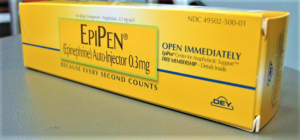
- Pfizer unit Meridian under civil investigation by U.S. Attorney (reuters.com)
Pfizer Inc said...it received a request for documents as part of a U.S. investigation related to quality issues involving the manufacture of auto-injectors at its Meridian Medical Technologies site...Meridian, a unit of Pfizer that manufactures EpiPen injectors...has been hit by a series of manufacturing problems in recent years...In 2017, Meridian had received a warning letter from the Food and Drug Administration. The FDA said Meridian had failed to thoroughly investigate product failures, including EpiPen products that were associated with patient deaths and severe illnesses. It said the company failed to take corrective actions until FDA’s inspection...
- NACDS, NCPA brief Congressional Doctors Caucus on DIR fee reform (chaindrugreview.com)
NACDS and the National Community Pharmacists Association held a briefing this week for the Congressional Doctors Caucus...chaired by Rep. Phil Roe...comprised of 16 medical providers in Congress who use their medical expertise to develop patient-centered healthcare policy...NACDS and NCPA discussed the Centers for Medicare & Medicaid Services’ recently released proposed rule—Modernizing Part D and Medicare Advantage to Lower Drug Prices and Reduce Out-of-Pocket Expenses—and explained the serious repercussions of direct and indirect remuneration (DIR) fees and the impact they can have on patient access to care and on pharmacies, who are struggling to maintain their businesses amid the uncertainty caused by the retroactive fees. The groups noted that they have advocated strongly for DIR reform in the CMS proposal because it will benefit pharmacies and the patients they serve.
- Purdue eyes bankruptcy filing to cope with mounting opioid accusations: Reuters (fiercepharma.com)Exclusive: OxyContin maker Purdue Pharma exploring bankruptcy -sources (reuters.com)
Purdue Pharma may file for bankruptcy to get out from under thousands of lawsuits blaming the opioid maker and its aggressive marketing for the addiction crisis...The drugmaker is battling a weight of litigation, including state claims that target its executives and founding family, claiming it misled doctors and patients and marketed its painkillers too aggressively, helping to create a nationwide opioid crisis. Now, the company is exploring bankruptcy as a way to cope with the amassing litigation...A bankruptcy filing would halt proceedings in the lawsuits and allow Purdue to negotiate with plaintiffs under the watch of a bankruptcy judge...READ MORE
- China slashes tax to stimulate domestic orphan drug market (pharmaceutical-technology.com)
China has announced a plan to slash the Value Added Tax on 21 rare disease therapies and four active pharmaceutical ingredients, dropping it by 80% in a bid to stimulate the domestic rare disease therapy market and drop the prices of the therapies...China is currently suffering from a shortage of rare disease therapies; of the 44 globally available rare disease therapies, only 19 are currently marketed within China, and of these 19, all are priced out of reach for the average Chinese patient. And with an estimated population of nearly 20 million people suffering from a rare disease within China, this is a huge area of unmet need...
- This Week in Managed Care: March 1, 2019 (ajmc.com)
, welcome to This Week in Managed Care from the Managed Markets News Network
Jaime Rosenberg