- OTC drugmakers face an uphill battle on Amazon. But they should still be there, expert says (fiercepharma.com)
Amazon is now the third-largest digital ad platform, but pharma marketers are mostly shut out. That’s because most of Amazon’s ads are for products sold by retailers, and of course, prescription drugs can't be purchased that way...as Amazon looks for an Rx entry, it's already launched its own Basic Care OTC medicine lineup. Pharma marketers with OTC brands should be figuring out their own strategies for Amazon...Over-the-counter remedies, which are part of many of Big Pharma's consumer divisions, are burgeoning on Amazon, so much so that Amazon quietly launched its own Basic Care brand line of OTC products last year…There are two typical Amazon advertising choices: sponsored brand ads or sponsored product ads...It's not really an advertising game OTC makers can sit out. Amazon is approaching a 50% market share in U.S. e-commerce sales...
- California, Massachusetts go toe-to-toe for title of best biotech hub (biopharmadive.com)
...the California Life Sciences Association released its industry report...pitting itself again against Massachusetts for the title of being the best state for the pharmaceutical industry...the report found California-based organizations received $3.9 billion in National Institutes of Health grants this year, ahead of Massachusetts, which was second in the nation at $2.7 billion. Additionally, CLSA found California companies had more than 1,300 medicines in the pipeline and roughly 130,000 employees working for 1,500 biopharmaceuticals and medical device companies in 2018..the MassBio report found Massachusetts-based biotechs dominated the initial public offering market last year, making up nearly half of all biotech IPO money and more than a third of biopharma venture capital funding...
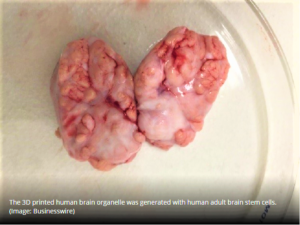
- 3D printed brain helps characterize 11 lead compounds, neuropathological research (outsourcing-pharma.com)
The stem cell research and therapeutics company Celprogen has identified and characterized 11 lead compounds for potential drug candidates for the treatment of Alzheimer’s, Parkinson’s, and Glioblastoma using its 3D model system...The 3D model system is unique since it represents the data generated closer to in-vivo data, eliminates animal studies, and represents patient’s brain physiological format...poor animal study design and reporting have raised concerns about whether current processes are the best way to conduct effective and efficient drug development...3D model systems aim to better inform risk-based decisions in drug development...
- More Nevada hospitals earn top marks on patient safety (reviewjournal.com)
The number of Nevada hospitals adhering to best practices on patient safety jumped in a new report…The state’s overall ranking rose to 16th, up from 40th in the spring...The rankings are based on the percentage of hospitals in the state earning A grades...which more than doubled in Nevada from just under 17 percent to 35 percent...Seven Nevada hospitals received top marks, including four in Southern Nevada: Henderson Hospital, Mountain View Hospital, North Vista Hospital and St. Rose Dominican Hospital, Rose de Lima campus. Northern Nevada Medical Center, Renown South Meadows Medical Center and St. Mary’s Regional Medical Center in Reno also received As.
- This Week in Managed Care: November 16, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network..
- Tens of thousands die in Africa each year due to fake drugs (reuters.com)
Tens of thousands of people in Africa die each year because of fake and counterfeit medication...The drugs are mainly made in China but also in India, Paraguay, Pakistan and the United Kingdom...Almost half the fake and low-quality medicines reported to the World Health Organization between 2013 and 2017 were found to be in sub-Saharan Africa...“Counterfeiters prey on poorer countries more than their richer counterparts, with up to 30 times greater penetration of fakes in the supply chain...The counterfeit drug market is worth around $200 billion worldwide annually...making it the most lucrative trade of illegally copied goods...
- US court says Vegas hospital must pay $820K in wages case (apnews.com)
Nevada’s public hospital in Las Vegas has been ordered to pay nearly $820,000 in sanctions and attorney fees for failing to produce required emails, text messages and computer records in an ongoing federal employee wages lawsuit...University Medical Center has until Dec. 5 to pay the penalty in the civil case alleging that thousands of nurses and other employees routinely worked through 30-minute meal periods for no pay...The court found that UMC had repeatedly violated its discovery obligations and its duty to preserve...noting that some electronically stored information that was turned over included “indecipherable codes complete with Japanese and Korean characters.”
- Draft Brexit withdrawal agreement: what does it mean for the pharma industry? (pharmaceutical-technology.com)Patients and industry fret over drug supplies, if no Brexit deal (reuters.com)
...UK Government and the European Commission published a draft Brexit withdrawal agreement. Like with every industry, the document has implications for the pharmaceutical industry, including clarification about the lack of disruption to supplies during the transition period and sharing of marketing authorisation information between the UK and EU member states...A key area of debate during the negotiation process was the impact of Brexit on the pharmaceutical industry, particularly the disruption in supply of medicines...The draft agreement stipulates that the transition period will last from 29 March 2019 until 31 December 2020. During this time the UK will continue to abide by all EU rules and remain under the jurisdiction of the European Court of Justice...The aim of the transition is to provide time for the UK to negotiate its relationship with the EU, particularly in terms of trade deals...the UK will share the “marketing authorisation dossier of a medicinal product” authorised by its Medicines and Healthcare products Regulatory Agency with EU member states or the European Medicines Authority...
- November 16 Pharmacy Week in Review: Emergency Ebola Fingerstick Test with Portable Reader Receives Authorization (pharmacytimes.com)
Laura Joszt, welcome to the Pharmacy Times News Network, Pharmacy Week in Review, , this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- HHS recommended that the DEA make kratom a Schedule I drug, like LSD or heroin (statnews.com)
The Department of Health and Human Services has recommended a ban on the chemicals in kratom that would make the popular herbal supplement as illegal as heroin or LSD…HHS asserted in a letter to the Drug Enforcement Administration that two chemicals in kratom should be classified as Schedule I substances... FDA...has said that kratom is “an opioid” and has been “associated” with dozens of deaths...Kratom should not be used to treat medical conditions, nor should it be used as an alternative to prescription opioids...Some states have already banned kratom, but it’s currently legal at the federal level. It’s sold in different forms, including dry powder and capsules. According to the American Kratom Association, millions of Americans use the substance.