- Cannabis Testing Lab In Nevada Gains Accreditation For Quality Standards (kunr.org)
As the marijuana industry is expanding in Nevada, cannabis testing labs are playing an important role in the quality of the end-product...a local facility is upping the standard of its operations...Jason Strull is a toxicologist at 374 Labs located in Northern Nevada. The facility recently received additional accreditation—ISO 17025—from an independent, non-governmental auditor for its stringent testing methods, yielding consistent results regarding toxicology and potency...“And as far as cannabis testing goes, it’s sort of the wild, wild West,” Strull explains. “So what this shows to potential customers, and just the general public, is that we’re going the extra mile.”
- Five Industry Trends for U.S. Drug Wholesalers in 2017 (drugchannels.net)
Here are five significant industry trends that will substantially impact the drug wholesaling industry in the coming years.
- U.S. pharmaceutical spending – and drug prices – are still growing. - We project that in 2016, for the first time, the Big Three wholesalers’ combined drug distribution revenues will exceed $400 billion.
- The consolidation of pharmacy and provider markets shows no sign of slowing - Mergers and acquisitions among pharmacies and payers are pressuring wholesaler margins, especially as the acquiring companies consolidate buying power.
- Payers and manufacturers are narrowing drug channels, pressuring wholesalers’ profitability - Narrow pharmacy networks – either preferred or limited models – are now a widely accepted element of pharmacy benefit design.
- Large pharmacies have entered into multifaceted partnerships with wholesalers – This...consolidation is creating a channel supply alignment among the largest wholesalers, largest PBMs and largest retail chains.
- Biosimilars are unlikely to provide superior profits to wholesalers. - Competition between a biologic drug and biosimilar is therefore likely to resemble brand-to-brand competition rather than brand-to-generic competition.
- Merck wins $2.54 billion in hepatitis C drug trial against Gilead (reuters.com)
Merck & Co...was awarded $2.54 billion in royalties by a federal jury in a patent lawsuit against Gilead Sciences Inc over Gilead's blockbuster hepatitis C drugs Sovaldi and Harvoni...The jury in Delaware reached the verdict following a nearly two-week trial, finding that a patent acquired by Merck in 2014 on hepatitis C treatments was valid...Harvoni and Sovaldi have drawn attention for their breakthrough success in curing hepatitis C in more than 90 percent of patients, and for their high cost...Harvoni's list price is $1,125 per pill and $94,500 for a 12-week regimen. Foster City, California-based Gilead, one of the world's largest biotechnology companies, made nearly $20 billion on the two drugs in 2015.
- Pharmacy Week in Review: December 16, 2016 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharmacies miss half of dangerous drug combinations (chicagotribune.com)
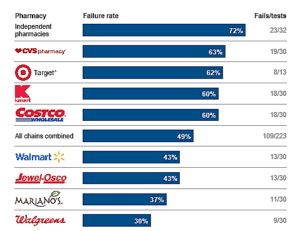
In the largest and most comprehensive study of its kind, the Tribune tested 255 pharmacies to see how often stores would dispense dangerous drug pairs without warning patients. Fifty-two percent of the pharmacies sold the medications without mentioning the potential interaction, striking evidence of an industry-wide failure that places millions of consumers at risk...CVS...had the highest failure rate of any chain in the Tribune tests, dispensing the medications with no warning 63 percent of the time. Walgreens...had the lowest failure rate at 30 percent — but that's still missing nearly 1 in 3 interactions... There is a very high sense of urgency to pursue this issue and get to the root cause...test after test, other pharmacists dispensed dangerous drug pairs at a fast-food pace, with little attention paid to customers. They failed to catch combinations that could trigger a stroke, result in kidney failure, deprive the body of oxygen or lead to unexpected pregnancy with a risk of birth defects...The Tribune study...exposes fundamental flaws in the pharmacy industry. Safety laws are not being followed, computer alert systems designed to flag drug interactions either don't work or are ignored, and some pharmacies emphasize fast service over patient safety. Several chain pharmacists, in interviews, described assembly-line conditions in which staff hurried to fill hundreds of prescriptions a day.
- NACDS hails rule relaxing access to pharmacy rewards (chaindrugreview.com)
The Department of Health and Human Services is giving pharmacies more flexibility to extend their reward and loyalty programs to beneficiaries in government programs such as Medicare and Medicaid…it will allow government program beneficiaries to enjoy access to programs voluntarily implemented by pharmacies, which reduce health care costs, improve quality and promote patient health...This is a very positive step forward for pharmacy patient care...and of the expanded innovative programs that pharmacies provide...Programs vary in their designs but often include cost savings or other rewards for filling prescriptions or engaging in other health-related activities, such as health screenings, medication adherence programs or healthy lifestyle behaviors…
- New Data Show Prescription Profits Under Pressure at Independent Pharmacies (drugchannels.net)
I review our latest estimates on pharmacy economics and margins. The data reveal that independent pharmacy owners are starting to perform poorly. In 2015, the average pharmacist owning a single pharmacy earned about $200,000. It’s the second year in which the average owner’s compensation declined. The number of independent pharmacies and gross margins also are trending downwards...independent pharmacies are under-performing with generic substitution…
SIX OBSERVATIONS ON THE 2015 DATA
Overall independent pharmacy profit margins have remained stable.
Independent pharmacies’ prescription profit margins are trending downward.
Independent pharmacies’ gross profit per prescription was stable.
Independent pharmacies have lower generic dispensing rates.
The average pharmacist owning a single pharmacy earned about $200,000 in 2016—down for the second year.
- China’s OxyContin Boom Is a Gold Mine for This Drugmaker (bloomberg.com)
With its harsh anti-narcotics laws and painful history with debilitating opium epidemics in the 19th-century, China wouldn’t spring to mind as a promising market for OxyContin, a painkiller that has been at the center of an opioid addiction outbreak in the U.S…Yet in China, powered by soaring cancer rates and an aging population, OxyContin is turning into a hit. And the drug company behind the brand is giving sales an added boost through an outreach push to physicians and by working with the most powerful of allies—the Chinese government...OxyContin is sold...by Mundipharma (China) Pharmaceutical Co., a company associated with...Purdue Pharma LP, the seller of the long-acting opioid in the U.S...China faces ever greater challenges in managing the use of inherently risky opioids within its sprawling, state-run health-care system.
- Hospital Impact: Drug companies win, patient safety loses with 21st Century Cures Act (fiercehealthcare.com)
The 21st Century Cures Act passed the House and Senate with overwhelming bipartisan support and has now been signed into law...On its surface, there seems to be little controversy in supporting more funding to the National Institutes of Health for innovative research, more expeditious approval of promising drugs by the Federal Drug Administration, and better approaches to combat the opioid abuse epidemic...However, there are a number of troubling provisions in this potential law of which the public should be aware...To outline the advantages and concerns, I created a series of winners and losers from which readers can draw their own conclusions:
- Winners
Drug companies: It shouldn’t surprise anyone that the pharmaceutical industry is ecstatic over the law, as the bar for approval of drugs under development is made significantly lower by reducing the rigor required for approval of new drugs by the FDA. Traditionally, rigorous double-blinded peer review studies are required prior to consideration and approval. - Losers
The FDA: Already stymied with deteriorating labs, under staffing and somewhat compromised approval processes for new drugs and medical devices, the FDA is not expected to fare better under the new law...Lowering the bar for the rapid and expeditious approval of pharmaceutical and medical device products with expansion of off label uses will only further undermine the FDA’s ability to protect the public from potential harm...Evidence-based medicine: The law allows for the use of “real-world evidence”—which may include anecdotal studies or observations, patient or professional opinion, as well as non-validated or scientifically measured outcomes—to justify the approval of a new treatment product...pharmaceutical companies will only be required to submit a “data summary” for new pharmacological agents or new indications to existing products, which will make the introduction of new products far easier.
- Winners
- This Week in Managed Care: December 16, 2016 (ajmc.com)
Laura Joszt with The American Journal of Managed Care. Welcome to This Week in Managed Care, from the Managed Markets News Network.