- Pfizer to nearly halve COVID-19 vaccine production timeline, sterile injectables VP says (fiercepharma.com)
DNA production—the first step in Pfizer's vaccine manufacturing process—could soon take just nine to 10 days, rather than 16...With an upsized production goal of 2 billion COVID-19 vaccine doses this year, Pfizer and its German partner BioNTech aren’t resting on their laurels now that their shot, Comirnaty, has emergency nods in the U.S., Europe and beyond. As the companies continue to build out capacity, manufacturing efficiency is getting its own boost...The time it takes the company to produce a COVID-19 vaccine batch could soon be cut from 110 days to an average of just 60...READ MORE
- Nevada climbs out of bottom in administering vaccine, CDC says (reviewjournal.com)
Nevada no longer has one of the worst COVID-19 vaccination rates per capita in the U.S., according to federal data...The Silver State had consistently ranked among the bottom five states at administering vaccine for weeks. It now ranks 12th worst, the Centers for Disease Control and Prevention reported...READ MORE
- New Massachusetts law recognizes pharmacist as providers (pharmacist.com)
...the governor of Massachusetts signed legislation that includes language recognizing pharmacists as health care providers in the state...The new law, S.2984: An Act Promoting a Resilient Health Care System That Puts Patients First, not only expands scope of practice for certain health care providers, but also increases insurance coverage for telehealth services...“This is a culmination of nearly a decade of advocating for pharmacists and their profession in our state,” said Massachusetts Pharmacists Association President Maureen Judkins, PharmD...READ MORE
- Vizient Report Highlights Drug Pricing Trends (drugtopics.com)
A new Vizient report highlights trends in drug pricing for pharmaceutical products used across multiple health system settings...Drug price increases remain prevalent in the oncology and rheumatology therapeutic areas, while use of other high-cost drugs appears to have surged in light of the coronavirus disease 2019 (COVID-19) pandemic. However, the report also points to an overall trend toward moderation of price increases and the projected effects of biosimilar and generic competition in the marketplace...According to the report, Vizient projects a 2.67% increase in the price of pharmaceuticals purchased by health systems, academic medical centers, pediatric hospitals, and non-acute practices during this time frame...READ MORE
- AbbVie, Novo Nordisk lead pharma TV advertisers into big-spending January (fiercepharma.com)
Pharma marketers are continuing their TV ad push into 2021. January pharma TV spending picked up where December ended—matching branded ad spending among the top 10 almost dollar for dollar...The highest spenders racked up $216 million for the month after a robust $217 million December, according to data from real-time TV ad tracker iSpot.tv...Now, with the third month in a row of the top 10 clearing $200 million, are pharma companies setting a new standard for TV spending?...Maybe. Five autoimmune disease treatments and three diabetes meds at the top of the list may point to increases in categories' competition. That could, in turn, foster hand-in-hand media buying increases to get TV mindshare...READ MORE
1. Humira
2. Rybelsus
3. Dupixent
4. Skyrizi
5. Ozempic
6. Trulicity
7. Eliquis
8. Xeljanz
9. Enbrel
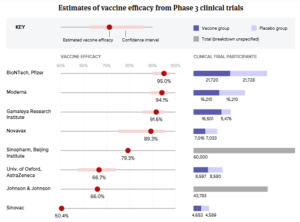
10. Opdivo and Yervoy - The first coronavirus vaccines have arrived. Here’s where the rest stand. (biopharmadive.com)
Study results showed vaccines from J&J; and Novavax to be effective against COVID-19. But seemingly weaker protection versus new virus variants have raised concerns...Scientists, drugmakers and governments have moved with unprecedented haste to develop a vaccine against the new coronavirus...The fastest of them have completed studies proving their vaccines can protect against COVID-19. A half dozen shots from developers in the U.S., U.K., Germany, China and Russia have now been cleared by regulators for emergency use...READ MORE
- AstraZeneca, Oxford race to update COVID-19 vaccine as study flags weak action against variant (fiercepharma.com)
It didn’t take long before a morale boost for AstraZeneca’s COVID-19 vaccine was overshadowed by disappointment over its waned protection against a newly emerged coronavirus variant...A new study has found AZ’s COVID-19 shot offered “minimal protection” against mild to moderate disease caused by the B.1.351 variant, which was first identified in South Africa, the University of Oxford, the original developer of the vaccine, said...READ MORE
- Nearly 60% of Critical Care Pharmacists Report Burnout (pharmacypracticenews.com)
With many ICUs reaching or exceeding maximum capacity in spring 2020 due to the COVID-19 pandemic, it may not be a surprise that about 60% of critical care pharmacists reported feeling burned out in a recent national survey...In an electronic survey that queried critical care pharmacists about their institutions and number of activities performed in May and June 2020, 128 of 221 respondents (58%) reported feelings of burnout such as emotional exhaustion and depersonalization...READ MORE
- FDA clears Lilly’s COVID-19 antibody cocktail for emergency use (biopharmadive.com)
The Food and Drug Administration...cleared an antibody drug cocktail from Eli Lilly for emergency use for treating people recently diagnosed with COVID-19...The cocktail, a combination of two coronavirus-targeting antibodies, is authorized only for people with mild or moderate symptoms of COVID-19, but who are at high risk of the disease's worst effects due to age, underlying medical conditions or other preexisting conditions...The combination pairs Lilly's bamlanivimab...with another antibody called etesevimab that the drugmaker developed in partnership with China's Junshi Biosciences. Each antibody targets a separate section of the "spike" protein used by the SARS-CoV-2 virus to breach the body's cells — a feature designed to preserve the drug's effectiveness even as the virus mutates...READ MORE
- Pfizer, Johnson & Johnson balk at shareholders’ push for COVID-19 vaccine pricing info (fiercepharma.com)
What's the rationale behind COVID-19 vaccine and drug prices? You don’t have a need to know—or so say a couple of the leading contenders...Two major players in the pandemic fight, Pfizer and Johnson & Johnson, are urging the Securities and Exchange Commission to forestall shareholder resolutions that would require them to disclose how they set prices on their COVID-19 vaccines...Several not-for-profit groups are pushing the two companies—along with fellow pharmas Eli Lilly, Gilead, Merck & Co. and Regeneron—for information on their drug and vaccine pricing decisions, citing the federal money all have received, either for supplies, R&D or manufacturing scale-up. Or all three...READ MORE