- Access to Rx Drugs is Priority (realclearhealth.com)
Access to medicines and cures should be the priority – the focus – for America right now, not unnecessary complications over trade with the Chinese...If one nation shuts down the production of a good, it impacts the supply for the whole world. With drugs, the U.S. gets many ingredients from China and if the Chinese shut off access to these materials, American patients will suffer...The Chinese are using the threat of withholding necessary drug ingredients if trade obstacles with the U.S. supervene...The Chinese are trying to link the Huawei telecom controversy to the drug issue...if the U.S hurts China by continuing current restrictions on Huawei’s access to American technology, they would answer with the withholding of Rx drugs...READ MORE
- AstraZeneca Covid-19 vaccine study put on hold due to suspected adverse reaction in participant in the U.K. (statnews.com)
A large, Phase 3 study testing a Covid-19 vaccine being developed by AstraZeneca and the University of Oxford at dozens of sites across the U.S. has been put on hold due to a suspected serious adverse reaction in a participant in the United Kingdom...A spokesperson for AstraZeneca, a front-runner in the race for a Covid-19 vaccine, said in a statement that the company’s “standard review process triggered a pause to vaccination to allow review of safety data.”...READ MORE
- NIH panel says data doesn’t support plasma use for COVID-19 (biopharmadive.com)
A panel of advisers for the National Institutes of Health was not convinced convalescent plasma should be used to treat COVID-19, a recommendation that appears to conflict with a controversial decision by the Food and Drug Administration last week to issue an emergency authorization for the blood-derived treatment...The panel...reviewed the same data cited by the FDA, but concluded it to be "insufficient" to recommend "either for or against the use of convalescent plasma for the treatment of COVID-19."...the same panel cautioned against using hydroxychloroquine in treating coronavirus disease three weeks after the FDA cleared emergency use of the malaria pill...READ MORE
- Costa Rica researchers to trial coronavirus treatment from horse antibodies (reuters.com)Costa Rica researchers to trial coronavirus treatment from horse antibodies (scientificamerican.com)
Researchers in Costa Rica are due to begin trials of an inexpensive coronavirus treatment based on antibodies taken from horses injected with the SARS-Cov-2, the virus that causes COVID-19...Developed by University of Costa Rica’s Clodomiro Picado Institute, the equine antibodies medication is to be tested on 26 patients from mid-September...Costa Rican authorities hope to be able to begin applying the treatment more widely in hospitals if the results from the phase 2 study are encouraging. There are 471 hospitalized coronavirus patients in Costa Rica...Similar efforts are also underway in Argentina and Brazil, while scientists in Belgium are using llamas...READ MORE
- Generic Drug Shortage Solutions on the Horizon (drugtopics.com)
Shortages of vital generic drugs—particularly since the start of the coronavirus disease 2019 pandemic in the US—are well known by pharmacy teams in both health systems and community settings...although pharmacy associations appreciate the Trump administration’s efforts to move more pharmaceutical and active pharmaceutical ingredient manufacturing to the United States...they say government initiatives must include an overall plan for drug pricing, payment model, and supply chain transparency...READ MORE
- Amgen wriggles out of state bill on rat poisons, says it prefers its rodent-killing methods (fiercepharma.com)
Changing methods can be tough—that's why Amgen is sticking with its rat poison of choice...With a bill passed this week, California legislators and environmentalists cast a spotlight on the use of industrial rodent poisons that are killing off native wildlife. But local drugmaker Amgen wanted fewer restrictions on its rat-killing methods, leaving critics to suggest: Maybe try traps, instead?...Amgen finagled its way out of the bill—AB 1788, which passed the California Assembly earlier this week—which would have required the company to rethink its rat policy at a facility in Newbury Park. The complex backs up to a wildlife preserve frequented by cougars and other wildlife, according to the Acorn...In an emailed statement, Amgen said it worked to secure the exemption because it simply couldn't identify another method for sending rats to meet their maker—or one would meet the FDA's standard, at least...READ MORE
- FDA scolds Mylan for ‘repeated’ manufacturing problems (biopharmadive.com)
The Food and Drug Administration is concerned that generics giant Mylan has deep-rooted problems in its global manufacturing network, with the latest evidence coming from a site in India...The agency posted this week a warning letter about Mylan's Unit 7 facility, which makes dozens of active pharmaceutical ingredients, or APIs, used in blood pressure drugs, antifungals and central nervous system treatments. During a February inspection, regulators found the site did not have adequate procedures in place to prevent contamination. They noted that similar issues were seen last year at Unit 8..."These repeated failures at multiple sites manufacturing API demonstrate that your company's oversight and control over the manufacture of drugs is inadequate. You should immediately and comprehensively assess your company’s global manufacturing operations,"...READ MORE
- Researchers identify nanobody that may prevent COVID-19 infection (phys.org)An alpaca nanobody neutralizes SARS-CoV-2 byblocking receptor interaction (nature.com)
Researchers at Karolinska Institutet in Sweden have identified a small neutralizing antibody, a so-called nanobody, that has the capacity to block SARS-CoV-2 from entering human cells. The researchers believe this nanobody has the potential to be developed as an antiviral treatment against COVID-19...READ MORE
- House committee to subpoena AbbVie over drug pricing (biopharmadive.com)
A congressional oversight committee plans to subpoena AbbVie for documents as part of an investigation into drug pricing... The House Oversight and Reform Committee has been seeking documents related to the pricing of AbbVie's top two drugs, which generated more than $23 billion combined last year...Chairwoman Carolyn Maloney, D-N.Y., said the company's responses have been "particularly poor"...AbbVie is the only one to receive a subpoena thus far, with Maloney saying its "noncompliance stands out as particularly egregious." The drugmaker said it was "surprised and disappointed" by the committee's action...READ MORE
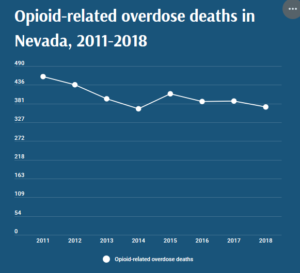
- With opioid-related overdoses on the rise, health care providers try preparing everyday Nevadans to respond to a crisis (thenevadaindependent.com)
With opioid-related overdoses on the rise, health care providers try preparing everyday Nevadans to respond to a crisis..From January to May 2020, Nevada saw 23 percent more opioid-related overdose deaths than during the same period in 2019, and similar trends are being seen across the country...Opioid-related overdose deaths peaked in Nevada in 2011 and have been on the decline since then, but around the U.S., rates have been rising throughout the COVID-19 pandemic. The American Medical Association released a report in mid-August citing news reports from 40 states and Washington, D.C. showing a rise in overdoses and illicit substance abuse since March...According to data from the Nevada Overdose Data to Action Program, there have been 197 opioid-involved drug overdose deaths in 2020 as of May 31, a 23 percent increase over the 160 counted in the first five months of 2019. April and May had the highest rates of overdose-related emergency room visits, with a 25 percent increase over the three months prior...READ MORE