- FDA Publishes Guidance on CGMP Requirements During COVID-19 (pharmtech.com)
FDA published guidance on June 19, 2020 detailing the agency’s recommendations for current good manufacturing practices (CGMP) requirements for addressing COVID-19 infection in employees engaging in drug manufacturing. The guidance was issued to help mitigate and prevent effects on drug safety and quality by employees confirmed to be either infected with COVID-19 or potentially exposed to someone with COVID-19...READ MORE
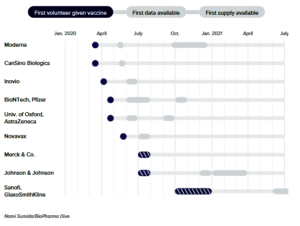
- The coronavirus vaccine frontrunners have emerged. Here’s where they stand
Fast progress by several companies has spurred hopes that a vaccine is coming soon, spurring jockeying among governments to secure supplies…Scientists, drugmakers and governments are moving with unprecedented speed to deliver a vaccine to protect against the new coronavirus…The fastest of them have already delivered preliminary data from human studies, and further results from others should come quickly as the year progresses…The goal, at least in the U.S., is to have a vaccine ready for use in some fashion by the end of the year, or early next. Doing so would be a scientific feat with few parallels. No vaccine has ever been developed so quickly, never mind manufactured for the world…Vaccine frontrunners plan for fast development…READ MORE
- Merck, Lilly and Amgen win again in lawsuit over drug prices in TV ads. Will it stick? (fiercepharma.com)
The Trump administration's quest for drug prices in TV ads just took another hit—and it might be a fatal blow. A U.S. appeals court agreed with a lower court ruling that found the U.S. Department of Health and Human Services didn't have the authority to require them...It was a big win for Merck & Co., Eli Lilly and Amgen along with the Association of National Advertisers, which sued last June to block the rule that would have forced drugmakers to include list prices in TV ads...Their argument? HHS has no statutory authority to create the rule in the first place, and even if it did, the rule violates the First Amendment...READ MORE
- FDA Releases Drug Interaction Warning for Remdesivir and Hydroxychloroquine (drugtopics.com)
The FDA released a warning to health care providers concerning an update on potential drug reactions for remdesivir, an antiviral drug that is being evaluated as a potential treatment for the novel coronavirus disease 2019 and has also been granted emergency use authorization status for treating hospitalized patients with severe COVID-19...The FDA is also revising their fact sheet for health care providers to include the warning that co-administration of remdesivir and chloroquine phosphate or hydroxychloroquine sulfate may result in less effective antiviral activity of remdesivir. The revised fact sheet also clarifies dosing and administration recommendations, and will provide additional safety data and updates from clinical trials from the National Institutes of Health and Gilead Sciences, Inc, the company that sponsors the drug and has donated 607,000 vials of remdesivir to the United States government...READ MORE
- It’s the end of road for hydroxychloroquine in COVID-19 as Novartis, NIH and WHO pull out of trials (fiercepharma.com)
The road for hydroxychloroquine against COVID-19 is coming to an end. Three major clinical programs have been terminated after a U.K. trial found “no clinical benefit” for the malaria drug...the World Health Organization, generic hydroxychloroquine maker Novartis and the U.S. National Institutes of Health have all ended their HCQ COVID-19 studies in hospitalized patients in quick succession...The WHO and NIH cited lack of benefits for patients, while Novartis blamed “acute enrollment challenges.”...Numerous investigator-sponsored trials may still be underway, but none of them has the scale of these three to yield any convincing results...READ MORE
- Pharmacy Groups Praise New York COVID-19 Pharmacist Vaccination Law (drugtopics.com)
New York pharmacists can provide the coronavirus disease 2019 vaccine when it becomes available, according to new legislation...The Community Pharmacy Association of New York State and NACDS praised the new law, based on legislation (S. 8182-A / A. 10508-A) that adds COVID-19 to the list of illnesses for which pharmacists can vaccinate...The legislation was...was signed into law by New York Governor Andrew Cuomo...“Now is the time to make sure that people will be able to get their COVID-19 vaccination as soon as it becomes available. It is essential that all states continue to remove barriers for pharmacies to help meet the needs of patients during this phase of the pandemic,”...READ MORE
- NACDS challenges WSJ on pharmacy value (chaindrugreview.com)Letting the Docs Dispense - Should patients have to make a trip to the drugstore to fill a prescription? (pay wall) (wsj.com)
In a letter-to-the-editor, National Association of Chain Drug Stores President and CEO Steven Anderson took extremely strong issue with a recent Wall Street Journal editorial that ignored the unique role of pharmacies and pharmacists in patient health, medication safety, healthcare cost reduction and crisis response...The newspaper referred to the pharmacy as a “needless middleman” – a point that Anderson confronted vigorously by detailing the trusted value that pharmacies deliver in the face of extreme barriers and often unfair and unsustainable reimbursement policies...READ MORE
- U.S. emergency visits due to heart attacks fell during early days of COVID-19 (reuters.com)Potential Indirect Effects of the COVID-19 Pandemic on Use of Emergency Departments for Acute Life-Threatening Conditions — United States, January–May 2020 (cdc.gov)
Fewer Americans were admitted to emergency departments with life-threatening conditions such as heart attacks during the initial months of the COVID-19 pandemic,..The study suggests that patients may be delaying or avoiding seeking care because of fear of COVID-19, researchers from the U.S. Centers for Disease Control and Prevention said...study...showed that the number of deaths in New York City from causes other than COVID-19 rose by more than 5,000 people above the seasonal norm during the first two months of the pandemic...Visits to the emergency department because of heart attacks fell 23%, ten weeks after the pandemic was declared a national emergency, compared with ten weeks before the emergency declaration...READ MORE
- Judge wipes out Biogen’s Tecfidera patent protections in suit against Mylan (fiercepharma.com)
In the high-stakes patent fight between Biogen and Mylan over Tecfidera’s main remaining patent, Mylan has scored a major win in federal court...U.S. District Judge...said Mylan “demonstrated by clear and convincing evidence” that certain claims of Biogen’s '514 patent are invalid for “lack of written description.”...The decision threatens Biogen’s bestselling medicine with early generics; Tecfidera, a multiple sclerosis drug, generated $3.3 billion in the U.S. last year. The company's '514 patent is set to expire in 2028, meaning the decision, if upheld, could wipe out years of monopoly sales...Biogen is also facing patent challenges in Delaware federal court. That court's decision will also factor into the ongoing efforts by generics companies to launch copycats...READ MORE
- OHSU’s COVID-19 Study Accused Of Racial Bias (opb.org)
Charges of racial bias in the design of an Oregon study of COVID-19’s spread are raising questions about whether it will do anything to help Black and Latino communities, which have been among those hardest hit by the pandemic...“All it will be able to say is if white people are fine. And then we open up counties and people of color will die,” said Andres Lopez, research director for the Coalition of Communities of Color, a Portland-based alliance of organizations representing a number of different communities of color...The Key to Oregon Study, which plans to enlist 100,000 Oregonians and monitor them for a year for COVID-19 symptoms, will include what its designers are calling “a focus on enrolling people who fully represent the state, including our diversity in geography, socioeconomic status and communities of color.”...critics doubt Key to Oregon will succeed in its goal. They say the study design is fundamentally flawed, and that those flaws could have been avoided if people of color had been brought to the table when the study was being created...READ MORE
-Responding with listening sessions
-Advocates say study design suppresses Black and Latino voices
-OHSU methodology overlooks lessons of the past
-The principle of ‘nothing about us without us’
-OHSU researchers respond