- Bristol-Myers Squibb must face whistleblower suit claiming underpaid Medicaid rebates (fiercepharma.com)
Years after a former pharmacist sued Bristol-Myers Squibb and other companies for allegedly underpaying Medicaid rebates, a federal court rejected Bristol's attempt to escape the case...Pharmacist and lawyer Ronald Streck filed a False Claims Act whistleblower lawsuit against Bristol-Myers and other companies back in 2013, but later withdrew his claims against the other defendants. Now, his lawsuit alleges that BMS fraudulently manipulated its average manufacturer prices to underpay Medicaid rebates from 2007 to 2016...Pennsylvania federal judge Timothy Savage ruled last week that Streck "has alleged sufficient facts to state a false claims cause of action."...
- This Week in Managed Care: November 30, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Teva to recall certain blood pressure medicine in U.S. (reuters.com)
Teva Pharmaceutical Industries Ltd is recalling certain combinations of blood pressure drug valsartan in the United States following the detection of a probable cancer-causing impurity...The...drugmaker will recall all lots of amlodipine-valsartan and amlodipine-valsartan-hydrochlorothiazide combination tablets due to an impurity in an ingredient made by an India-based unit of Mylan, the U.S. Food and Drug Administration...The European Union last week effectively banned here sales of valsartan made by the Mylan India unit after some batches were found to contain the same impurity, N-nitrosodiethylamine...
- Florida’s opioid lawsuit against CVS and Walgreens takes aim at distributors with deep pockets (cnbc.com)
Florida Attorney General Pam Bondi has added Walgreens and CVS Health as defendants in the state's massive lawsuit against the opioid industry...Legal analysts say Florida and other plaintiffs are targeting the distributors and pharmacies, in part, because they have deep pockets...The...lawsuit accuses the drug stores and pharmaceutical distributors like Cardinal Health, Amerisource Bergen and McKesson of playing as big a role in the proliferation of opioid addiction as drug manufacturers like...Purdue Pharmaceuticals and Johnson & Johnson's Janssen Pharmaceuticals...
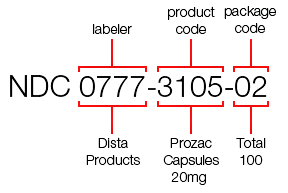
- The NDC Shortage: What the FDA Could (and Should) Do to Address It (drugchannels.net)An Open Letter To The FDA: New NDC Format Public Meeting (rxtrace.com)
...the U.S. Food & Drug Administration will run out of 5-digit National Drug Code codes within the next ten to fifteen years…The problem now facing U.S. healthcare is that, due to an explosion of new labelers entering the market, the FDA is running out of 5-digit labeler codes. The 5-digit format provides 90,000 potential combinations, and although that seems like a lot, the FDA anticipates running out of labeler codes within the next 10-15 years...the disruption and impact to the healthcare industry will be widespread and of a large magnitude, and will require retooling of major industry systems...
- November 30 Pharmacy Week in Review: Trial Evaluating Effectiveness and Safety of Drugs Used to Treat Patients with Ebola (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Medicine or vice? Socially screened funds struggle to define cannabis industry (reuters.com)
Is marijuana a medicine or a vice?..The $8 trillion U.S. socially responsible investment industry is grappling with that question as more states approve the recreational use of cannabis, pushing consumption closer to “sin” stocks like alcohol and tobacco that ethically focused investors avoid...No U.S. public companies are directly selling marijuana, but Canadian marijuana producers like Tilray Inc and Canopy Growth Corp are on U.S. exchanges...Marijuana is used to treat a range of conditions from epilepsy to migraines...There’s a lot of mixed feelings about cannabis, whereas with tobacco there’s a lot of consensus that tobacco is not safe in any amount...
- Walmart and Aurobindo sued as litigation mounts over contaminated blood pressure drugs (fiercepharma.com)
Litigation is growing over tainted blood pressure medications with a class-action lawsuit now filed in Florida against Walmart and three drug companies that span the supply chain...In addition to the retailer, the lawsuit names Indian drugmaker Aurobindo, U.S. drugmaker ScieGen Pharmaceuticals and distributor Westminster Pharmaceuticals. It was filed in federal court in Florida against the four over the sale of irbesartan because the drug contained the impurity, N-nitrosodiethylamine...Hauppauge...and its distributor Westminster initiated a nationwide voluntary recall to the consumer level of all lots of unexpired irbesartan tablets because they had been made with the Aurobindo API and then shipped to Walmart. The FDA learned this summer that the suspected carcinogen...can show up in “sartan”-based drugs as a result of certain manufacturing processes...
- CVS Health Completes Acquisition of Aetna, Marking the Start of Transforming the Consumer Health Experience (cvshealth.com)CVS Completes $70 Billion Acquisition of Aetna (wsj.com)CVS-Aetna merger approved by New York regulators (nypost.com)
CVS Health...today announced that it has completed its acquisition of Aetna...“Today marks the start of a new day in health care and a transformative moment for our company and our industry,” declared CVS Health President and Chief Executive Officer Larry J. Merlo. “By delivering the combined capabilities of our two leading organizations, we will transform the consumer health experience and build healthier communities through a new innovative health care model that is local, easier to use, less expensive and puts consumers at the center of their care.”
- UK vows to speed up drug review in return for 2% sales cap (in-pharmatechnologist.com)
UK government and ABPI have agreed a deal to limit branded drug sales growth to 2%, potentially reducing the cost of medicine by £930m ($1.18b)...The announcement was made after the UK government and the Association of the British Pharmaceutical Industry provisionally signed the Voluntary Scheme for Branded Medicines Pricing and Access...the two key details released are: branded medicines will be subject to a 2% cap on sales growth – with pharma companies repaying the NHS for sales over this limit – and appraisals of new technology applications by NICE will be completed up to six months faster than current timelines...