- 5 Ways Pharmacists Can Help Prevent Suicide (pharmacytimes.com)How Suicide Quietly Morphed Into a Public Health Crisis (nytimes.com)
With the recent deaths of Spade and Bourdain, health care professionals are also concerned about suicide contagion, a phenomenon in which high-profile suicides influence patients to attempt or committ suicide themselves...pharmacists can play a key role in preventing suicide...pharmacists are ideally situated to assist those in need because of their frequent interactions with patients and access to medical records...However, pharmacists are often unprepared to properly respond to signs of suicide risk, as very few pharmacy schools incorporate suicide prevention courses into their curricula...here are 5 ways pharmacists can help patients who are contemplating suicide...
- Identify at-risk patients.
- Monitor medication use and mental health.
- Collaborate with the health care team.
- Refer to suicide prevention resources.
- Be encouraging and empathetic.
- This Week in Managed Care: June 8, 2018 (ajmc.com)
Kelly Davio, welcome to This Week in Managed Care from the Managed Markets News Network
- U.S. drug prices hit by insurer tactic against copay assistance: analysis (reuters.com)
A recently adopted tactic by...health plans to limit the financial assistance drugmakers provide directly to consumers for prescription medicines is taking a toll on drug prices, according to a new analysis...Real...drug prices, including discounts and rebates, fell 5.6 percent in the first quarter of this year, compared to a 1.7 percent drop in the same period a year ago, according to...research analyst Richard Evans...He attributed most of the decline to ‘copay accumulator’ programs introduced by pharmacy benefits managers...drugmakers have increasingly offered so-called ‘copay assistance’ cards, similar to a debit card, that consumers can use at the pharmacy counter to reduce their out-of-pocket costs...these payments insulate consumers from the real costs of their drugs and can push them toward more expensive medications when a cheaper option is available...Beginning in January, Express and other pharmacy benefits managers introduced a new ‘copay accumulator’ approach, refusing to allow copay assistance payments to contribute toward a patient’s deductible before insurance kicks in...That has forced drugmakers to either keep paying out-of-pocket costs for a consumer, or risk them ditching a medicine because they can no longer pay for it....
- Women’s hormone drug prices have climbed for years, but controversy hasn’t ensued. Why? (fiercepharma.com)
Several drug pricing scandals have grabbed headlines in recent years in cases in which companies hiked the prices of old drugs or jacked up the cost of blockbuster drugs little by little year after year. Another case hasn't garnered the same attention—and maybe that's because people just don't like to talk about vaginas...insurers have shifted more costs to patients through higher deductibles and copays, leaving patients with a higher share of the bill, and that means women are paying more out of pocket...To avoid that, some resort to buying the remedy from overseas outlets...some insurers have placed the drugs on high formulary tiers, so patients must pay more to get them...
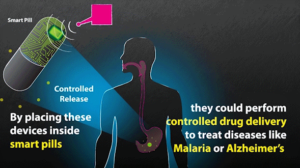
- Wireless system can power devices inside the body (news.mit.edu)
MIT researchers, working with scientists from Brigham and Women’s Hospital, have developed a new way to power and communicate with devices implanted deep within the human body. Such devices could be used to deliver drugs, monitor conditions inside the body, or treat disease by stimulating the brain with electricity or light...The implants are powered by radio frequency waves, which can safely pass through human tissues...in animals, the researchers showed that the waves can power devices located 10 centimeters deep in tissue, from a distance of 1 meter...Even though these tiny implantable devices have no batteries, we can now communicate with them from a distance outside the body. This opens up entirely new types of medical applications...An overarching aspiration is that regulators will provide input to the design and may incorporate framework elements and learnings into regulatory programs. .
- June 8 Pharmacy Week in Review: Drug Shortages, Folic Acid and Erectile Dysfunction, and Precision Medicine in Cancer Treatment (pharmacytimes.com)
Kelly Davio, host, the Pharmacy Times News Network's Pharmacy Week in Review.
- White House: Trump’s drug pricing plan will make the rest of the world pay its fair share (cnbc.com)
First, the president's plan would cut domestic drug prices by dismantling the burdensome government approval and reimbursement policies that inhibit healthy competition. Second, the plan would combat foreign government policies that devalue intellectual property rights and create unfair pricing systems that force drug manufacturers to sell to foreign buyers at unreasonably low prices...President Trump's plan would stop overpricing of drugs at home and underpricing abroad...Government policies that restrict competition...the lengthy drug approval process erects large entry barriers for both brand-name drugs and their cheaper generic counterparts. FDA's work to facilitate timely generic entry is estimated to have saved Americans billions of dollars...doctors are given precisely the wrong incentive when prescribing: the system reimburses them at higher rates when they prescribe more expensive drugs, ultimately funded by our tax dollars...There is no free lunch. If neither Americans nor foreigners pay for the R&D to develop new drugs, then soon nobody will receive new treatments...
- ISPE Field-Tests Quality Program (biopharminternational.com)
The International Society of Pharmaceutical Engineering announced...that it is field-testing the design principles of a comprehensive industry-led program of self-evaluation of pharmaceutical quality program with industry colleagues, senior leaders, and regulators. The vision for the program is to evolve the focus from submission of harmonized quality metrics as given in recent FDA guidance documents to establishing a framework for advancing the state of pharmaceutical quality, while at the same time continuing to align with the purpose outlined by FDA...ISPE believes this approach will deliver on the objectives outlined by FDA and will increase the value to industry...
- FDA sends warning letters to nine online marketers over opioids (reuters.com)
The...Food and Drug Administration...sent warning letters to nine online networks operating a total of 53 websites to stop illegally marketing unapproved versions of opioid medications...The regulator said... it is taking additional steps with these warning letters by going right to the source of the illegal supply of unapproved and misbranded versions of opioid drugs, including tramadol and oxycodone...The internet is virtually awash in illegal narcotics...Drug dealers and rogue website operators are using the internet to fuel the opioid crisis...The regulator has requested responses from the nine companies within 10 working days, or they may be subject to product seizure or injunction.
- The ‘cruel joke’ of compassionate use and right to try: Pharma companies don’t have to comply (statnews.com)
...the FDA has a compassionate use program to allow people access to experimental drugs, it can’t compel a company to provide those drugs. The newly signed “right-to-try” law doesn’t either...the Food and Drug Administration calls its expanded access program, also known as compassionate use. It governs the use of an investigational medicine that has not been approved by the FDA outside of a clinical trial...Here’s how it is supposed to work. A physician caring for a patient with a terminal illness who has exhausted all other treatment options and isn’t eligible for a clinical trial appeals to the pharmaceutical company to provide an investigational drug that has undergone at least a Phase 1 trial, which studies the safety of a drug. If the pharmaceutical company agrees, the treating physician applies to the FDA for approval for expanded access to the investigational drug...Large pharmaceutical companies are notoriously risk averse when it comes to expanding access to medications that are still in the testing phase. Many refuse to grant access to investigational drugs outside of clinical trials...One fear they have is that an adverse event, like an injury or death — even if it is not directly due to the medication — will derail a company’s ability to push a drug forward for FDA approval...Pharmaceutical companies also worry that if an experimental medication is given to one patient through compassionate use, it must be given to all patients who request it. In the case of rare diseases...this could mean that a company would have trouble enrolling enough patients when it eventually opens a clinical trial...