- Pharmacy Week in Review: April 27, 2018 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharmacy Week in Review: April 20, 2018 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Industry fears disruption as EU excludes UK from drug approvals (reuters.com)
A European decision to exclude Britain from the EU’s drug approval system from March 30 2019 - the day after Brexit - has raised alarm among drugmakers, who fear the abrupt change could disrupt medicine supplies to patients...The move confounds hopes for continued joint cooperation via the European Medicines Agency, at least during a transition or implementation period until the end of 2020 when the UK will remain closely tied to the European Union...the EMA has appointed experts from other European countries to take over work currently undertaken by Britain’s Medicines and Healthcare products Regulatory Authority from next March...Since the MHRA assesses around a fifth of EU medicines, drug industry leaders fear this sudden handover will cause disruption...The Association of the British Pharmaceutical Industry said it was clearly in the EMA’s interest to continue to draw on the expertise of the MHRA and it urged London and Brussels to come to an early agreement...
- U.S. appeals court strikes down Maryland drug price-gouging law (reuters.com)
A federal appeals court...declared unconstitutional a 2017 Maryland law that lets the state attorney general sue generic drugmakers who sharply raise prices on medications...The 4th U.S. Circuit Court of Appeals sided with the main trade group for generic pharmaceutical companies in holding that the law violated the U.S. Constitution by regulating the price of transactions that occur outside of Maryland...the law violated the Constitution’s bar against states interfering with interstate commerce, by targeting wholesale rather than retail pricing in transactions that occur largely outside of Maryland...
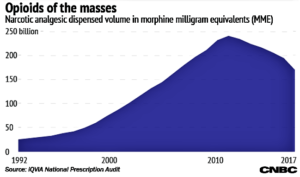
- How doctors in the US prescribe opioids — four charts (cnbc.com)Institute Reports Medicine Use and Spending in the US Review of 2017 Outlook to 2022 Medicine Use and Spending in the U.S. A Review of 2017 and Outlook to 2022 (iqvia.com)
The number of opioid pills prescribed peaked in 2011 and has since declined by 29 percent, according to a new report from the Iqvia Institute...The number of prescriptions accelerated its decline in 2017, with an even faster drop for high doses...More patients appear to be starting on medication-assisted therapies to treat opioid addiction...decline accelerated...helped by changes in regulation of opioid prescribing and in reimbursement policies from insurers...many of the programs that have been put into place in the past year or two seem to be having an impact...increased use of nonopioid pain treatments, like ibuprofen and other nonsteroidal anti-inflammatory drugs...as well as public awareness about overuse and misuse of opioids...
- FDA commissioner to drug middlemen: You’re part of the problem (cnbc.com)
Drugmakers may not be the only ones keeping less-expensive drugs off the market...The Food and Drug Administration has approved nine biosimilars, generic versions of biologic medicines, but only three are available...Manufacturers are using several schemes to "hamstring biosimilar competition," FDA Commissioner Scott Gottlieb said... he worries pharmacy benefit managers have been "complacent participants" in the schemes...PBMs and insurers may stick with branded biologics because they receive discounts from manufacturers on these treatments. That can leave consumers paying for costly treatments when less-expensive ones are available while PBMs make more money on these discounts, known as rebates...Many of these practices persist because high list prices enable lucrative returns across the drug supply chain as the spread between list and net price is carved up and shared among participants...he (Gottlieb) applauded insurers who have recently pledged to pass manufacturer's drug rebates directly on to some of their members...This is a bold action that will help create a fairer, more transparent market...I hope that other insurers, employers, and manufacturers follow their lead. I also hope that your industry will continue to innovate to make it more transparent to pass along these rebates...
- DEA to share prescription drug data with 50 attorneys general, crack down on drugmakers (fiercehealthcare.com)
The Drug Enforcement Agency has reached an agreement with 50 attorneys general to share prescription drug data with one another to support ongoing investigations...from its Automation of Reports and Consolidated Orders System, which collects 80 million prescription drug transactions from manufacturers and distributors each year...Attorney General Jeff Sessions...said the data-sharing pact will “make both the DEA and our state partners more effective at finding evidence of crime.”...Exactly how that data sharing agreement would operate remains fuzzy...
- This Week in Managed Care: April 20, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Amazon finds it’s not so easy to sell drugs—and nixes one plan entirely: CNBC (fiercepharma.com)
Amazon is running into challenges in its drug distribution push...Amazon's potential leap into pharmaceuticals has weighed on drug distributors and pharmacies for months, but those companies are getting relief from news that the online retail giant has put at least one of its plans on ice...a unit that sells bulk products to companies—hasn't been able to convince hospitals to get on board with its plan. One reason: The company doesn't have a proper cold-chain logistics network, which is crucial to distributing many drugs...Amazon has now backed away from the effort, the publication reported...The company is pressing ahead with other healthcare projects, though it's unknown whether they involve pharmaceutical sales...
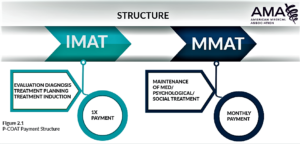
- Backed by medical groups, value-based payments for opioid addiction treatment could be imminent (fiercehealthcare.com)PATIENT-CENTERED OPIOID ADDICTION TREATMENT (P-COAT) (asam.org)
Two medical associations have announced a collaborative value-based payment model for opioid addiction treatment...The payment model, announced by the American Society of Addiction Medicine and the American Medical Association, is aimed at improving care coordination and lowering healthcare spending by reducing costly emergency department visits and hospitalizations...The model increases utilization of and access to medications for opioid treatment, combining them with the appropriate level of medical, psychological and social support services which can be delivered at multiple providers…The model is for outpatient care only...a basic version of the model is already in operation with Optum…the groups have discussed the model with officials at the Centers for Medicare & Medicaid Services, as well as its innovation center, which is responsible for new demonstration programs.