- Drug industry scrambles after rare loss in budget deal (thehill.com)
Pharmaceutical companies are pushing to repeal or roll back a provision in last week’s budget deal that delivered a rare loss to their industry...A provision included in the budget deal approved...raised the share of costs that drug companies have to pick up as part of closing the “donut hole,” a gap in drug coverage for Medicare Part D beneficiaries...Drug companies are quickly mobilizing to try and undo the change, or at least roll it back in some fashion. The most likely avenue is the long-term government funding bill that Congress is expected to pass in March, the lobbyists said...The budget deal raised the share of costs in the donut hole that drug companies have to pick up from 50 percent to 70 percent. The drug companies are pushing proposals that would put the share at 60 percent or even lower, sources say...The change in the budget deal would not go very far in reducing drug costs for patients, though it does impose some new costs on drug companies. The proposal was not championed by groups that want action to lower drug prices.
- Report: Opioid Manufacturers Gave Millions to Advocacy Groups (ptcommunity.com)
Senator’s investigation found a “lack of transparency” surrounding the donations...A new report from Senator Claire McCaskill found that five opioid manufacturers paid nearly $9 million to 14 outside groups between 2012 and 2017, alleging that the advocacy groups often “amplified messages favorable to increased opioid use.”...The groups—many of which work on chronic pain and other opioid-related issues—lobbied to defeat prescriber limits on opioids...and many criticized facets of 2016 guidelines from the Centers for Disease Control and Prevention that limited the prescribing of painkillers...“The financial relationships between these groups and opioid manufacturers should be clear to the general public,” McCaskill said. “We passed a law ensuring the public had information on payments to doctors by pharmaceutical companies, and I can’t imagine why the same shouldn’t be done in this space.”...
- Gilead’s freshly approved Biktarvy faces patent hurdle (drugstorenews.com)
Gilead Sciences’ new HIV treatment Biktarvy has been approved by the Food and Drug Administration — but its novel component is also the subject of patent infringement litigation from GSK...Biktarvy is a triple-therapy HIV treatment that brings together bictegravir’s unboosted integrase strand transfer inhibitor with the dual nucleoside transcriptase inhibitor backbone of emtricitabine, tenofovir alafenamide — which are the two components of Gilead’s Descovy...However, now bictegravir is at the center of GSK’s lawsuit, which was filed in the U.S. District Court for the District of Delaware, as well as in Canadian Federal Court in Toronto. GSK’s majority-owned Viiv Healthcare said it is looking to prove that Biktarvy’s use of bictegravir infringes on a patent covering its dolutegravir and other compounds that use dolutegravir’s chemical scaffold. The company said it would seek financial redress...
- Teva’s Turnarond: Is It Ever Or Never? (forbes.com)
No one doubts that Teva Pharmaceutical Industries CEO Kare Schultz has been making all the right moves to turn around the troubled drug maker’s dwindling fortunes...Burdened by more than $32 billion in debt from past acquisitions, the world’s largest generic drug maker is cutting costs at feverish pace. The company suspended dividend payments to investors, amended debt covenants and eliminated low-value research projects...hopes for 2018 just got dashed to pieces...Following the delivery of estimate-beating fourth quarter financial results early Thursday, Teva unveiled full-year sales and profit forecasts for 2018 that were a sliver of what had been expected by Wall Street analysts, due largely to falling prices for its U.S. drugs...Teva now sees 2018 revenue falling between 16% and 18% and expects to earn as little as $2.25 a share this year...
- Fake meds on trial: EU report weighs up Member State punishment (in-pharmatechnologist.com)
Diverging penalties in EU Member States for the falsification of medicines raise concerns for organised crime, says Medicines for Europe...The European Commission has submitted a report detailing the application of penalties for those involved in the production and circulation of falsified medicines to the European Parliament on the request of the Falsified Medicines Directive (2011/62/EU)...While the Commission said measures taken by Member States are “satisfactory”, it has called for their strict enforcement...“…penalties are only effective if they are well-enforced,”...“I urge all EU countries to make sure that criminals falsifying medicines are punished.”...A lack of data regarding illegal activity in Member States makes it challenging to determine the effectiveness of national penalties...According to the report, all Member States deem at least some activities relating to the falsification of medicinal products a criminal offence. However just 21 EU countries enforce criminal penalties for the manufacture, distribution, brokering, import, export and sale at a distance of falsified medicines...
- Nordic project takes ‘manufacturing-on-demand’ approach to future drugs (in-pharmatechnologist.com)
Nordic Universities will investigate 3D printing, electrospraying, and microfluidics in an industry supported collaboration aimed at revolutionising production in an age of personalised medicine.
The collaboration, known as Nordic POP (Patient-Oriented Products), will use 35m DKK ($6m) of funding from NordForsk...to create flexible and translational approaches to personalised medicine manufacturing…Jukka Rantanen...University of Copenhagen...said that new patient-oriented and personalised drug products require a “totally new mindset” in the drug development process...“The project team is aiming to create new innovative drug products, where the dose, release mechanism, and size/shape of the product could be easily personalised based on the patient needs...“Instead of one-size-fits-all medication, the potential of new patient oriented products considering gender, age, lifestyle, genetic profile, metabolic capacity, and microbiota will be explored,”... - US OKs medical isotope system that isn’t based on bomb-grade uranium (cnbc.com)
The federal government...approved a device made by a private company...that will allow the first domestic production of a medical imaging isotope...a move the government said would enhance national security by reducing the need to transport weapons-grade uranium...The Food and Drug Administration granted the approval to NorthStar Medical Radioisotopes, which said it would begin delivering systems to make technetium-99...the most common isotope in medicine and is used in 40,000 procedures a day in the United States...consumers have long had to depend on a complicated and risky supply chain for the materials...The current process involves shipping weapons-grade, or highly enriched, uranium from the United States to research reactors in Australia, South Africa and Europe where it is irradiated to make molybdenum-99, which decays into technetium-99..."This is a win for our national security," said Peter Hanlon, an official with the National Nuclear Security Administration office of material management and minimization...
- Minnesota renews push for tax on prescription opioids (reuters.com)
Minnesota Governor Mark Dayton...announced a renewed legislative proposal to tax prescription opioid pills to help fund treatment...Minnesota is one of at least 13 states to have considered an opioid tax in recent years to help pay for the fallout from the United States’ opioid epidemic, although none have passed, according to the National Conference of State Legislatures...Dayton’s proposal would levy a one-cent tax on drugmakers for each milligram of active ingredient in a prescription pain pill, generating an estimated $20 million a year for prevention, policing, emergency response and treatment...
- Purdue Pharma to stop promoting oxycodone to U.S. doctors (pharmacist.com)
Purdue Pharma says it will stop promoting oxycodone (OxyContin) and other opioids to U.S. doctors. The company will continue selling the products, but Purdue's sales force "will no longer be visiting offices to engage in discussions about opioid products," the company says. Doctors and other prescribers who have questions about the drugs will have to contact Purdue's medical affairs department, the company says. Purdue is also cutting its U.S. sales force by more than 50%, to about 200 people. The remaining sales representatives will market non-opioid products. Purdue's halting of its opioid marketing comes as the company faces growing legal scrutiny. More than a dozen states and about 400 cities and counties in the U.S. have sued Purdue or other opioid analgesic makers, accusing them of fueling addiction by misrepresenting the risks of their drugs. In response to the suits, Purdue has said it is "dedicated to being part of the solution" to the opioid crisis.
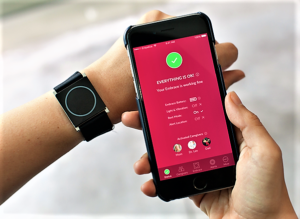
- FDA Clears the First Smart Watch for Use in Neurology (ptcommunity.com)
Wearable device identifies convulsive epileptic seizures and sends alerts to caregivers...The FDA has cleared the Embrace smart watch (Empatica, Inc.) for use by patients with epilepsy. Embrace uses advanced machine learning to monitor for the most dangerous kinds of seizures, known as “grand mal” or “generalized tonic-clonic” seizures, and sends an alert to summon caregivers’ help...The smart watch stands apart from any seizure detection system in that it measures multiple indicators of a seizure. Its unique property is its use of electrodermal activity, a signal used by stress researchers to quantify physiological changes related to sympathetic nervous system activity, also known as the "fight-or-flight" response. Embrace has been approved in Europe as a medical device for seizure monitoring and alert since April 2017.