- This Week in Managed Care: March 17, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- How a Major Drugmaker Plans to Cure Disease… Without Drugs (fool.com)
Hacking into the body's nervous system may allow GlaxoSmithKline to commercialize an entirely new field of medicine...GlaxoSmithKline plc has the resources to pursue long-term bets in medicine that could eventually change the course of disease treatment and yield large payoffs...to literally eavesdrop on the body's electrical system and enter into the body's own internal conversations in order to heal disease...every organ has a nerve connection that regulates its function. Organs are controlled by patterns of electrical impulses transmitted through nerve fibers, but when organs dysfunction in chronic disease, the electrical patterns are different. That fact opens up the possibility that inserting patterns in nerves to certain organs can correct conditions that lead to disease...scientists are just beginning to realize the possibilities of using nerve signals to restore organs to normal function and actually treat disease...Bioelectronic medicines have the potential of doing to the pharmaceutical industry what biopharmaceuticals did to small molecules back in the 1980s…
- NCPA Asks U.S. Senate and House Leadership to Hold Hearings on Pharmacy DIR Fees Bills (ncpanet.org)
The National Community Pharmacists Association recently requested Congressional hearings on legislation, the Improving Transparency and Accuracy in Medicare Part D Spending Act, that would stop the unfair practice of pharmacy direct and indirect remuneration (DIR) fees being applied retroactively under Medicare Part D...Retroactive pharmacy DIR fees are creating a system of winners and losers...While pharmacy benefit managers profit, the unpredictable timing and amount of these clawbacks are wreaking financial havoc on pharmacies, seniors, and taxpayers...By requiring PBMs to divulge the costs of prescription drugs for Medicare beneficiaries at the point of sale, these bills fix a problem even the Centers for Medicare & Medicaid Services acknowledges has contributed to rising costs in Medicare Part D.
- DIR fees are pushing seniors into the donut hole coverage phase faster, where seniors absorb all of the costs for their prescriptions;
- DIR fees are also pushing seniors into the ensuing catastrophic coverage phase faster, where the government's costs have risen from $10 billion in 2010 to $33 billion in 2015;
- In both of those instances, the PBMs' co-pay burden disappears, which is not the case in the initial phase of coverage;
- Independent community pharmacies' ability to plan in advance is being undermined by how PBMs currently apply DIR fees, threatening patient access to these pharmacies, especially in underserved communities; and
- All of this is occurring while PBMs continue to operate in a non-transparent manner when it comes to DIR fees.
- 5 trends changing clinical trials (biopharmadive.com)
Clinical trials have become increasing costly ventures, adding to the overall cost of developing a drug and, ultimately, the price that patients pay for drugs. A 2016 estimate by the Tufts Center for the Study of Drug Development, for example, pegged average clinical trial costs across all three phases of development at roughly $340 million in out-of-pocket expenses. Big pharmas and small biotechs alike are looking for innovative ways to improve trial outcomes and, in turn, lower trial costs — this means increasing the efficiency in which they recruit patients, monitoring more closely how drugs are supplied and being more flexible about trial design. Here’s a look at some of the tools that are optimizing clinical trials today:
- Patient centricity
- Tapping into technology
- Data you can wear
- Flexibility
- Automated site supplies
- Trump’s first budget seeks to slash $6B from NIH, raise FDA user fees (fiercebiotech.com)
President Donald Trump’s first budget will take $5.8 billion away from the National Institutes of Health, around 20% of its total, with FDA user fees also set to rise as biopharmas should "pay for their share."...The budget cut to the NIH...had around $30 billion in funds last year...The NIH...got a funding boost just last year when the 21st Century Cures Act was passed, a law that allowed the Institute an extra $4.8 billion in funding over the next decade...There was no direct mention of FDA cuts, but use fees are set to potentially double..Recalibrates Food and Drug Administration medical product user fees to over $2 billion in 2018, approximately $1 billion over the 2017 annualized CR level, and replaces the need for new budget authority to cover pre-market review costs. To complement the increase in medical product user fees, the Budget includes a package of administrative actions designed to achieve regulatory efficiency and speed the development of safe and effective medical products...
- New UK drug cost rules leave companies fuming (reuters.com)
British drugmakers...accused Theresa May's Conservative government of breaking a manifesto commitment to improve access to new medicines, following approval of new cost rules...The angry response from both Big Pharma and biotech firms comes despite a concession by government in the latest version of the scheme, which increases the cost threshold for certain drugs for rare diseases from the previously planned level...The row comes at a sensitive time for the government, which is about to trigger proceedings to leave the European Union but wants to encourage investment by strategic industries, including the high-tech pharmaceuticals sector...Drug companies are already concerned that Brexit could make Britain a less attractive market, especially if the country ends up outside the current EU-wide system for drug licensing...The new system means that new drugs costing the National Health Service more than 20 million pounds ($24 million) a year will no longer be automatically funded, even if they are cost-effective. Instead, companies will have to enter negotiations to justify their use and work out funding...
- Indian Drugmakers Seek to Sway Trump Over Cheap Generic Imports (bloomberg.com)
India’s largest pharmaceutical firms are looking to convince U.S. President Donald Trump that his promise to lower drug costs should take priority over his vow to make sure the medicines are made in America...Savings from cheaper imported drugs will outweigh the jobs created by producing them in the U.S., the Indian Pharmaceutical Alliance...said in a report...One goal could be to win an exemption for generic drugs under any border-tax policy that is enacted in the U.S….Indian companies recommend their government highlight the firms’ investments in the U.S., where the largest companies have increased their presence in recent years in an attempt to diversify their manufacturing base...Closer collaboration with the FDA is also recommended to help the industry solve deficiencies in manufacturing practices that have prompted a wave of sanctions from the regulator in recent years, hurting the industry’s reputation and sales…..The approach should be to project India as a reliable and trustworthy source for access to safe and quality medicines at affordable prices.
- Pharmacy Week in Review: March 17, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharmacy among best-paying job categories in America (pharmacist.com)
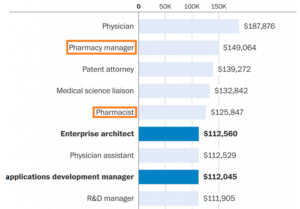
Pharmacy managers and pharmacists both landed in the top five of Glassdoor's annual list of the best-paying jobs in the United States. Glassdoor...noted health care jobs showed up in four of the top five spots. For the third year in a row, "physician" took the top slot, with a median base salary of $187,876...Pharmacy managers ($149,064) and pharmacists ($125,847) were listed at number two and number five, respectively. "Medical science liaisons" ($132,842)—specialists who work for pharmaceutical or biotech companies to establish relationships with medical experts—came in at number four.
- Express Scripts challenges Gilead in pricing blame game (biopharmadive.com)
Comments from Gilead Sciences’ head of worldwide commercial operations last week to Bloomberg have reignited a furious debate within the industry over who is to blame for rising drug costs...Gilead’s Jim Meyers told the publication that pharmacy benefit managers are to blame for keeping drug prices high...But PBM Express Scripts fired back this week with a letter to Gilead CEO John Milligan urging the company to cut the cost of its hepatitis C drugs and even pay back the difference retroactively...Meyers' comments insinuated PBMs keep drug prices high in order to inflate their own revenues through the cut they take from rebates and discounts given by manufacturers...Express Scripts...has taken particular offense to these accusations...The request from Express Scripts was a bold one and unlikely to materialize in Gilead paying back its profits. Yet, the public back-and-forth adds another dynamic to who may be to blame for high drug prices...