- DOJ turns tables on Express Scripts, demands info on pharma deals (fiercepharma.com)
Express Scripts disclosed this week that the U.S. Attorney’s offices in New York and Massachusetts had demanded information about two different issues: financial ties with pharma companies, and relationships among drugmakers, patient assistance programs and the specialty pharmacies that fill prescriptions...Specifically, the federal prosecutors in New York want information about money changing hands between Express Scripts and pharma companies. That would include rebates that drugmakers pay to win favorable reimbursement deals for their products...The newly disclosed Express Scripts probes aren’t the first DOJ demands for information about PBM-pharma relationships. In a series of financial filings in May, it became clear that the DOJ was looking for information across the industry. Johnson & Johnson, Merck & Co. and Endo International all said they were being asked for info...secrecy could be coming to an end. Politicians and patient advocates are calling for more transparency from PBMs and drugmakers alike, and obviously, the DOJ is doing the same.
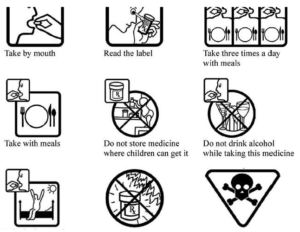
- New research reveals pictograms help seniors understand medication instructions (medicalxpress.com)
Nine different pharmaceutical pictograms that could help older people understand written medical information...Simple images designed to convey information about prescription drugs could help save lives and reduce the economic burden of non-adherence to treatment. New research...shows that including pictograms on written medication instructions helps seniors take their drugs correctly...Patients with multiple prescriptions can easily get confused and take the wrong medication, leading to hospitalization and even death…pictograms on a prescription drug label does help older people understand medical information and instructions. The pictograms provided information such as "take with meals" or "do not leave in direct sunlight" and warnings such as "poison" and "do not leave near children."...This not only prevents accidental overdose, it relieves some of the pressure that our aging population is putting on the health service by avoiding preventable tragedies...
- FDA issues guidelines for female libido pills after learning some hard lessons (statnews.com)
After a two-year wait, the US Food and Drug Administration finally issued new guidance for companies that want to develop drugs to bolster female libidos. But the details suggest the agency has belatedly learned some hard-fought lessons following complaints that the controversial Addyi pill did not warrant approval last year...The 15-page draft guidance...offers a typical how-to for companies, but also points to certain steps that Sprout Pharmaceuticals did not follow as part of its Addyi marketing application...The drug, which is now sold by Valeant Pharmaceuticals, was approved despite debate over its safety and effectiveness, and the extent to which medicines should be used to treat female sexual dysfunction...the FDA is locking the barn door after the horse got out. So now, the agency is telling other companies to do some things that Sprout didn’t do...they’ve made it harder to get a drug approved but they have extended helping hands in numerous places...unless [a company] really looks hard for subgroups [of patients], the whole effort is hopeless...
- DEA and Partners Hold Prescription Drug Take Back Day Saturday (dea.gov)
DEA reprises this weekend one of its most popular community programs: National Prescription Drug Take Back Day. On Saturday October 22 between 10 a.m. and 2 p.m., the public can dispose of their unused, unwanted prescription medications at one of 4,700 collection sites nationwide, operated by 3,800 local law enforcement agencies and other community partners. The service is free of charge, no questions asked.
- This Week in Managed Care: October 28, 2016 (ajmc.com)
Sara Belanger With The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Electronic prescriptions associated with less nonadherence to dermatologic Rx (medicalxpress.com)
Does how a prescription for dermatologic medicine is written - either on paper or electronically—matter when it comes to whether patients will fill it and pick it up?...A new study...used data from a large, urban county health system to measure primary nonadherence—defined as not filling and picking up all dermatologic prescriptions within one year of the prescription date—and to study whether electronic prescribing impacted primary nonadherence...electronic prescribing increases the coordination between pharmacists and clinicians, less is known about how electronic prescribing affects the rate at which patients will fill or won't fill new prescriptions...The risk of primary nonadherence was 16 percentage points lower among patients given electronic prescriptions than patients given paper prescriptions...As the health care system transitions from paper prescriptions to directly routed e-prescriptions, it will be important to understand how that experience affects patients, particularly their likelihood of filling the prescriptions. Primary nonadherence is a common and pervasive problem. Steps should be taken to better understand why primary nonadherence happens and how it can be improved...
- Inside big pharma’s fight to block recreational marijuana (theguardian.com)
Pharma and alcohol companies have been quietly bankrolling the opposition to legal marijuana, raising questions about threats to market share...Marijuana legalization will unleash misery on Arizona, according to a wave of television ads that started rolling out across the state last month...the surprise lies in who is backing them. In August, the pharmaceutical company Insys Therapeutics also cited concerns for child safety when, with a $500,000 contribution, it became the largest donor to Arizona’s anti-legalization drive. But their stated concerns have raised a few eyebrows across the state. Insys manufactures Subsys, a prescription painkiller derived from fentanyl…critics say, the Insys contribution in Arizona is a ploy to protect market share. And it mirrors other large donations to anti-marijuana campaigns by pharmaceutical and alcohol companies that fear the growing clout of legal marijuana...Research conducted...shows that medical cannabis patients are substituting cannabis for pharmaceuticals at a very high rate, and for alcohol at a pretty high rate as well...Opiate overdoses dropped by roughly 25% in states that have legalized medical marijuana compared to states that have prohibited sales of the plant, according to a 2014 study from the Journal of the American Medical Association. The study implies that people could be using medical marijuana to treat their pain rather than opioid painkillers, or they’re taking lower doses...
- Pharmacy Week in Review: October 28, 2016 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- How pharma is using Twitter to connect with patients (biopharmadive.com)
Pharma Twitter is not like your Twitter. Your Twitter probably isn't closely regulated by the FDA. Your Twitter presumably is not monitored by dozens of interest groups with competing agendas. Your Twitter probably existed well before Pharma Twitter, because pharma companies are notoriously slow to take up new technology. Your Twitter probably has a lot more interesting and relevant stuff. But if you are looking for a job or a clinical trial, then pharma's corporate Twitter feeds may be just the place for you...it's not easy for pharma to use Twitter. If a company is going to tweet about a drug, FDA regulations require the company to include the product name, indication, its most pressing risks and a link to an explanation of risks – all in the 140-character confines. For this reason, many companies shy away from product promotion on the platform...Here's a closer look at some of the best-known pharma names on Twitter:….
- Chicago’s proposal to license pharma sales reps is “bad morality, bad economics,” critic says (bizjournals.com)
Pharmaceutical sales representatives working in Chicago might soon have to carry a special license to operate within the city limits...city officials have proposed an ordinance that would require pharmaceutical reps to record and report to the city the number of health care professionals they've contacted, the types of drugs promoted, any samples provided, and if doctors were paid for their time...The ordinance might also require sales reps reps to keep a list of names of doctors they've contacted and potentially give those names to the city if requested. The new licensing requirements are part of a broader effort to fight prescription opioid abuse…The proposed pharmaceutical sales rep license would likely carry a $750 annual fee, the Tribune reported. In addition to the new reporting requirements, sales reps also would have to take city-certified training classes on prescription drug abuse, ethics and marketing standards...The...ordinance...would generate another stream in the flow of occupational licensing fees that go into city coffers...The city is looking for money, if I had to guess…