- Ex-FDA head and Sanofi call for harmonized drug regulation (reuters.com)The need for global regulatory harmonization: A public health imperative (stm.sciencemag.org)
Drug regulation has failed to keep up with a globalized world and governments should harmonize oversight to improve patients' access to new and innovative medicines...That is the view of the former leader of the U.S. Food and Drug Administration, the world's top drug regulator, and the research head of French drugmaker Sanofi, who made a joint plea to governments for action...Margaret Hamburg, who led the FDA until 2015, and Elias Zerhouni said there was an urgent need to harmonize a "mosaic of regulations" in different countries, and they called for the issue to be taken up at the G8 or G20 groups of nations..."Essentially, it is a hidden bureaucratic inefficiency tax on the whole effort of finding new and valuable therapies," he said in a telephone interview...Drug development is global and we need to have safety and efficacy data globally, so we should have a global system, just like with airplanes...Sanofi was spending around 20 percent of its research and development budget on ensuring convergence between different systems, often involving duplication of efforts.
- Federal Jury Convicts Boise Doctor Charged with Controlled Substance Delivery (dea.gov)
A Boise jury...returned guilty verdicts against Michael Minas, 50...on charges that he unlawfully distributed controlled substances outside the usual course of professional practice and not for a legitimate medical purpose…Minas illegally distributed was primarily oxycodone 30 mg, but the jury also found Minas guilty of distributing fentanyl and hydromorphone. The jury heard evidence that Minas wrote prescriptions for extraordinary dosages, such as 240, 300, and even 420 oxycodone 30 mg. They also heard evidence that he often wrote prescriptions at intervals of two or three weeks, but wrote dosage instructions on the prescriptions indicating that it was a month’s supply…Minas faces a maximum prison sentence of up to 20 years in prison, a maximum fine of $1,000,00 ($1,000,000) and at least three years of supervised release on each count of conviction.
- A faster and cheaper way to produce new antibiotics (worldpharmanews.com)
A novel way of synthesising a promising new antibiotic has been identified by scientists at the University of Bristol. By expressing the genes involved in the production of pleuromutilin in a different type of fungus, the researchers were able to increase production by more than 2,000 per cent...With resistance growing to existing antibiotics, there is a vital and urgent need for the discovery and development of new antibiotics that are cost effective. Promising developments are derivatives of the antibiotic pleuromutilin, which are isolated from the mushroom Clitopilus passeckerianus...These new compounds are some of the only new class of antibiotics to join the market recently as human therapeutics...with their novel mode of action and lack of cross-resistance, pleuromutilins and their derivatives represent a class with further great potential, particularly for treating resistant strains such as methicillin-resistant Staphylococcus aureus and extensively drug resistant tuberculosis...
- API repackager in hot water with FDA (fiercepharma.com)
A U.S. drug repackager is in hot water with the FDA, earning itself a warning letter by taking liberties with the expiration dates on the drugs it repackages and a having quality unit that pretty much shirked its responsibilities, even though the problems had been noted during inspections 5 years earlier...The warning letter to Apotheca Supply, which does business as Apothecares, was posted this week...the FDA said the company had extended the expiration dates...by as much as two years beyond those listed by the manufacturers without any scientific justification to do it and no stability testing that would insure the APIs would still be effective...In addition, the agency was concerned about potential for cross contamination because of its procedures...On top of that the inspector found expired cleaning products and there had been no testing to ensure that surfaces were cleaned of one product before work began on another...
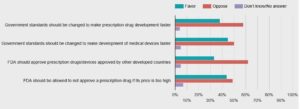
- Public wary of faster approvals of new drugs, STAT-Harvard poll finds (statnews.com)
A majority of Americans opposes federal regulatory changes to speed up the development and approval of new medical treatments, a new STAT-Harvard poll finds — suggesting the public has serious doubts about legislation now moving through Congress...both parties are pushing to change government regulatory standards that they blame for slowing the approval process to get new products to patients...nearly 6 out of 10 Americans said they oppose changing government safety and effectiveness standards to allow for faster approvals of new prescription drugs by the Food and Drug Administration, while 38 percent said they’re in favor of speedier FDA action...The poll sheds new light on how Americans balance their priorities between speed and safety in the approval of new medical treatments.
- Walgreens sending medical marijuana smoke signals? (drugtopics.modernmedicine.com)What is Medical Marijuana? Clarifying Clinical Cannabis (staywell.walgreens.com)
Walgreens’ posting of a seemingly sympathetic blog about the use of medical marijuana has created quite a buzz, with some online observers speculating the retail chain has its sight on that lucrative industry...But a Walgreens spokesperson insists the chain has not taken a stance on the use of medical marijuana and cautions people against reading too much into the blog posting. Pharmacies cannot legally dispense medical marijuana...The blog, entitled "What is Medical Marijuana? Clarifying Clinical Cannabis," was written by Dahlia Sultan, who is a resident pharmacist at Walgreens and associated with the University of Illinois at Chicago...Sultan...suggests "marijuana provides pain relief in ways traditional medicines don’t" and "medical marijuana can improve appetite and relieve nausea in those who have cancer and may help relieve symptoms such as muscle stiffness in people who have multiple sclerosis."...Not surprisingly, the blog has attracted the attention of medical marijuana advocates. Some believe that pressure from giant retailers such as Walgreens could eventually convince the federal government to reclassify marijuana so that it could be dispensed by pharmacies...Jim Cohn, spokesperson for Walgreens, said that people shouldn’t read too much into the blog. "The content [of the blog] is strictly informative, and nowhere do we take any stance on the issue,"...
- Onscreen doctors to write scrip at China’s Jo-Jo Drugstores (fiercepharma.com)
With a regulator nod for full online prescription drug sales in China on hold for now...Jo-Jo Drugstores plans a TV loop direct to doctors who will listen to your ailments and write up scripts...The Zhejiang province-based pharmacy chain won China FDA approval for the plan to install the virtual doctor screens at 6 stores, allowing them to consult and write prescriptions if needed that the pharmacist can fill on the spot...China FDA has grown increasingly cautious about the sale of online drugs. But it is willing to experiment with models that may bring down costs and provide better services...Access to doctors in China has been difficult historically in many rural areas of China...Not only does our program rectify this problem, relieving hospitals of patient overflow; we are also able to save consumers between 10% and 30% in prescriptions costs as compared to the exact same service rendered at area hospitals...
- U.S. probes contracts between drugmakers, pharmacy benefit managers (reuters.com)
The U.S. Attorney's Office...is investigating contracts between drugmakers and companies that manage prescription benefits...Federal prosecutors have approached at least three companies, including Johnson & Johnson, Merck & Co and Endo International Plc, demanding information about their contracts with pharmacy benefit managers...Pharmacy benefit managers...which administer drug benefits for employers and health plans and also run large mail-order pharmacies, have been challenging the rising cost of new medications...When drugs are knocked off their formularies, patients may have to pay full price for them. PBMs often keep or dump a product depending on whether they can obtain favorable pricing.
- FDA generics backlog improves, although criticism continues (drugtopics.modernmedicine.com)
While many groups are criticizing the FDA's backlog of generic drug approvals, the agency said the situation has improved...In December 2015 alone, OGD (Office of Generic Drugs) issued 99 approvals and tentative approvals...the most approvals and tentative approvals granted in a single month since the start of the generic drug program...FDA’s Office of Generic Drugs awarded 580 generic drug approvals and 146 tentative approvals in 2015...the Campaign for Sustainable Rx Pricing...is pressing legislators to grant FDA more resources, to allow quicker processing of generic drug applications. The group's members include AARP, ASHP, numerous health plans, providers, and Walmart...The FDA faces a backlog of nearly 4,000 generic drug applications, yet approval times can be three or more years...The FDA should be provided necessary resources to clear this backlog and prioritize generic drug approval applications...
- UPDATED: Merck KGaA expands viral, cell therapy production capacity in U.S. (fiercepharma.com)
Merck KGaA wants to be a go-to company to help others develop and manufacture gene-related therapies and will expand its campus in Carlsbad, CA, by nearly 50% as part of that effort...The German company’s MilliporeSigma unit said...it will add about 21,000 square feet to the facility...The expansion...will leave it with 16 modular viral bulk manufacturing cleanroom suites, two fill/finish suites and twice the warehouse capacity. The expansion will incorporate single-use equipment in a flexible, scalable format for clinical and commercial bulk drug production...Merck considers the Carlsbad site its flagship for viral and gene therapy work...