- GSK CEO: Big pharma should keep investing in R&D (cnbc.com)
GlaxoSmithKline CEO Andrew Witty attributes the company's success to sticking to the tried-and-true business model of investing in research and development and not transitioning to an acquisition model...the key is to be patient and to see the value of investing in innovation...What we have to stay focused on is the fundamentals of the value of innovation and discovery of medicines and vaccines that make a real difference to patients...The alternative acquisition model in the health care industry has drawn criticism during this election year. In theory, the M&A strategy can save companies significant sums as they can purchase mature, fully developed products without having to invest in R&D...
- FDA Approves New Version of Oxycodone (painnewsnetwork.org)
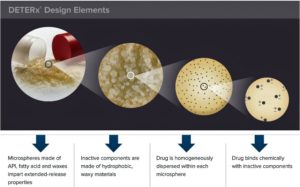
Food and Drug Administration has approved a new extended released version of the opioid painkiller oxycodone that has abuse deterrent properties unlike anything else on the market...Xtampza – can be ingested in capsule form, but users can also sprinkle the capsule contents on soft foods or into a cup, and then directly into the mouth...The medication, which can also be ingested through a feeding tube, is the sixth opioid pain medication with an abuse deterrent formula to be approved by the FDA...Xtampza is made by...Collegium Pharmaceutical with proprietary technology (DETERx technology platform) that combines oxycodone with fatty acid and waxes to form small spherical beads that are placed inside the capsule...The beads are designed to resist breaking, crushing, chewing, dissolving and melting, methods long used by drugs abusers to snort or inject opioids.
- Valeant annual report reveals new issues, company overhauls board (reuters.com)
Valeant Pharmaceuticals International Inc...issued a 2015 financial report that met an important deadline for creditors, revealing new details on a range of financial and legal issues, and it also announced changes to its board...Shares in Valeant fell 4.0 percent to $33.81...and are well down from their all time high of $263.70 in August...The publication of the report satisfies demands from Valeant's creditors, but also shows the difficult road ahead...We think investors should sell on this catalyst since the business is quite weak and faces a number of headwinds...The annual report does not answer some more basic questions about what strategy Valeant will adopt...They continue to dig out of a very troubled situation that will take time...The company...revealed it is being investigated by the U.S. Department of Justice in North Carolina...relating to the production, marketing, distribution, sale and pricing of three of its life-saving drugs...Massachusetts and New York are also investigating similar issues... Valeant said that seven current board members would not be standing for re-election at its June 14 shareholder meeting...
- In China’s tougher drug market, minnows open back door for ‘Big Pharma’ (reuters.com)
Armed with Beijing funds and friends in the right places, Chinese drug minnows are thriving, luring money from 'Big Pharma' majors struggling to restore the strong growth they once enjoyed in the world's second-largest medicine market...Chinese healthcare mergers and acquisitions nearly tripled last year to more than $50 billion, helped by giants like GlaxoSmithKline PLC and Eli Lilly and Co tapping small biotech and research innovators. The targets offer vital regulatory know-how as Beijing builds a domestic drug industry...For Big Pharma, acquisitions, licensing deals and joint ventures offer a back door into a market where Beijing expects healthcare spending to rise to $1.3 trillion by 2020. The majors need the opening: their China growth has stalled to low single-digit pace from over 20 percent just four years ago as branded generics have lost their shine...
- J&J Ordered to Pay $55 Million Over Cancer Linked to Talc (bloomberg.com)
Johnson & Johnson must pay $55 million to a 62-year-old South Dakota woman who blamed her ovarian cancer on the company’s talcum powder in the second such trial loss this year...State court jurors in St. Louis...awarded $5 million in compensation and $50 million in punitive damages to Gloria Ristesund, who was diagnosed with cancer in 2011 after using J&J’s talc-based feminine hygiene products for almost 40 years...J&J is accused in more than 1,000 lawsuits in state and federal courts of ignoring studies linking its Shower-to-Shower product and Johnson’s Baby Powder to ovarian cancer. Women contend the company knew the risk and failed to warn customers..."Unfortunately, the jury’s decision goes against 30 years of studies by medical experts around the word (world) that continue to support the safety of cosmetic talc,’’...Ristesund had several risk factors for ovarian cancer...That included a family history of cancer, having endometriosis and the fact she had no children...
- Drug Take-Back Day Is April 30 (realclearhealth.com)Got Drugs? (deadiversion.usdoj.gov)
Have you ever wondered how to get rid of an unfinished bottle of prescription drugs?...Don't throw it in the trash or flush it down the toilet, advises the Drug Enforcement Administration. Those methods of dumping your pills can actually be a safety hazard...Instead, Americans with expired, unused and unwanted prescription drugs can bring them for disposal at drop-off centers nationwide during Drug Take-Back Day, which takes place this year on Saturday, April 30...Drop-off sites will be open from 10 a.m. to 2 p.m. You can find one near you by going to the DEA's Diversion Control website. The service is free and anonymous...Only pills and patches will be accepted. The DEA will not take liquids, needles or sharps...The 10 previous take-back events have collected more than 5.5 million pounds (more than 2,750 tons) of pills...
- Hawaii looks to allow psychologists to prescribe drugs (hosted.ap.org)
State lawmakers are poised to make Hawaii one of a handful of states that allow psychologists to prescribe medication in hopes of increasing access to mental health services...The...bill would allow psychologists to prescribe medication if they undergo special training. It sets requirements including 400 hours of training, supervision of 100 patients and passing an exam created by the American Psychological Association...So far, psychologists in the Department of Defense can prescribe medication, along with those in New Mexico, Louisiana and Illinois. Just this week, lawmakers in Iowa approved a similar bill. Supporters of the Hawaii bill say those states can serve as a model for increasing access to mental health care...Outreach workers say Hawaii is in the midst of a mental health crisis. More Hawaii residents die by suicide than in car accidents, according to the Hawaii Department of Health...But opponents including the American Psychiatric Association and the Hawaii Medical Association say the bill would put Hawaii residents with mental illness at serious risk. They say some psychologists might not have the proper medical training needed to safely prescribe drugs that can cause deadly reactions, especially when mixed with other medications...
- France gets G7 to discuss global regulation of medicine prices (reuters.com)
France will press its G7 partners this month to launch an "irreversible" process to control the prices of new medicines, part of a global drive to make life-saving drugs more affordable...President Francois Hollande said in March he would push for the international regulation of drugs prices when he meets other G7 leaders in Ise-Shima, Japan on May 26-27...We need to initiate this process with firmness, and the president wants it to be irreversible...G7 nations are home to most of the leading drug makers and while governments are keen to tackle rising health costs they may be reluctant to pitch themselves against their own pharmaceutical industries...G7 delegations have begun initial talks on the issue but no one expects a breakthrough in the near future...
- California Mulls Coverage of Comprehensive Medication Management (ashp.org)Medi-Cal: comprehensive medication management (leginfo.legislature.ca.gov)
A bill to have the nation's largest Medicaid program cover comprehensive medication management services by pharmacists and primary care physicians emerged from a committee hearing on April 5...In the past few years, we have added millions of Californians into Medi-Cal, making the effective management of the quality and cost of care an absolute necessity...CMM is a smart, important innovation to help meet these goals...CMM, according to California Assembly Bill 2084, is "the process of care that ensures each beneficiary's medications...are individually assessed to determine that each medication is appropriate for the beneficiary, effective for the medical condition, and safe given the comorbidities and other medications being taken, and [that] all medications are able to be taken by the patient as intended."...Absent from the bill, however, was any requirement for Medi-Cal to pay pharmacists for providing healthcare services or for Medi-Cal managed care plans to credential pharmacists as providers...pharmacists' strongest support comes from the rural areas of California, where access to healthcare is more difficult than in the urban centers of the state...
- FDA reconsiders training requirements for painkillers (hosted.ap.org)
Food and Drug Administration is reconsidering whether doctors who prescribe painkillers like OxyContin should be required to take safety training courses...The review comes as regulators disclosed that the number of doctors who completed voluntary training programs is less than half that targeted by the agency...Under the current risk-management programs, drugmakers fund voluntary training for physicians on how to safely prescribe their medications...many experts - including a previous panel of FDA advisers - said those measures don't go far enough and that physician training should be mandatory...The FDA's initial ideas to improve safety included mandatory certification for doctors and a national registry to track patients taking the drugs. But industry pushed back. Drugmakers and the pain specialty groups they fund argued that certification would be too burdensome for doctors, leaving many patients undertreated. And patient groups said that registries would unfairly stigmatize those who rely on painkillers to deal with long-term pain....