- UnitedHealth Says It Should Have Avoided Obamacare Longer (bloomberg.com)
UnitedHealth Group Inc. should have stayed out of Obamacare’s new individual markets longer, the chief executive officer of the biggest U.S. health insurer said…after announcing last month that it will take hundreds of millions of dollars in losses related to the business…While the company’s other lines of business are growing, instead of expanding into Obamacare next year, the company should have kept waiting, UnitedHealth CEO Stephen Hemsley said…“It was for us a bad decision,”…“I take accountability for sitting out the exchange market in year one so we could in theory observe, learn and see how the market experience would develop. This was a prudent going-in position. In retrospect, we should have stayed out longer.”…UnitedHealth is not alone in its Obamacare struggles. Other insurers, including competitors Anthem Inc. and Aetna Inc., have also either suffered losses in the markets or said they haven’t seen the margins they expected.
- Lahey Clinic computer theft leads to $850,000 HIPAA settlement (modernhealthcare.com)
Lahey Hospital and Medical Center has agreed to pay $850,000 in a settlement with HHS' Office for Civil Rights to resolve alleged privacy and security violations stemming from the theft of a laptop computer with unencrypted patient records…health system also entered into a corrective action plan to address other privacy and security issues raised during the breach investigation. Lahey “impermissibly disclosed” electronic medical records of 599 individuals “for a purpose not permitted by the privacy rule” under the Health Insurance Portability and Accountability Act…Lahey had failed to meet a number of other HIPAA requirements, including not conducting “an accurate and thorough” security risk analysis, failing to assign “a unique username for identifying and tracking user identity” on the stolen computer and failing to “implement a mechanism to record and examine activity” on the computer.
- New law restores lost time for makers of some drugs (statnews.com)
President Obama has signed a law that resets approval dates for some medicines that contain controlled substances. The goal is to streamline the process used by the Drug Enforcement Agency to place prescription drugs that are controlled substances on a list of medicines for which distribution is restricted…the new law ensures that a company offering such medications will not effectively lose time from a five-year period under which it can exclusively market the drugs…Drug makers have griped they are often at a competitive disadvantage, since controlled substances must first be placed on the DEA list before they can reach pharmacies. Companies have complained the process is often lengthy and hampers their ability to take advantage of a five-year marketing exclusivity period following Food and Drug Administration approval during which generic competition is delayed…the Improving Regulatory Transparency for New Medical Therapies Act, would require the DEA to list a drug within 90 days after receiving notice from the FDA...
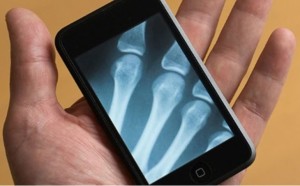
- Apple Watch used to study epileptic seizures (baltimoresun.com)Johns Hopkins EpiWatch: App and Research Study (hopkinsmedicine.org)
For the 2.5 million people living with epilepsy… medications can help control their seizures — most of the time. But some suffer unpleasant side effects from the drugs. And a few remain at risk of death…Researchers at the Johns Hopkins University hope to help those with the neurological condition by collecting information about their seizures through their…Apple Watches…"Physicians often ask patients to record their seizures, but that can be hard, especially when a patient loses consciousness," said Dr. Gregory Krauss…who is working on the program, named EpiWatch…collects data that help researchers better understand epilepsy, while helping patients keep a more complete history of their seizures," he said…The data could not only help doctors adjust an individual's treatment to make it more effective and less burdensome, but could also help...counter sudden unexpected death, a danger some face from seizures.
- Doctors’ use of computers during appointments leaves patients less satisfied (reuters.com)
Doctors who entered data into computerized health records during patients' appointments did less positive communicating, and patients rated their care excellent less often…Many clinicians worry that electronic health records keep them from connecting with their patients…doctors who used the computer more also spent more time correcting or disagreeing with patients…Doctors who spent more time using the computer spent less time making eye contact with patients and tended to engage in more “negative rapport building,” correcting patients about their medical history or drugs they’ve taken based on information in the electronic record…That’s not necessarily a bad thing…Doctors who spend most of the time looking at the computer may miss out on an emotional connection with the patient…When people are paying attention to the same thing at the same time, you get the best transmission of information… Electronic health records need to be more usable so clinicians with varying computer proficiency can use them without struggling and diverting focus from patients…
- Easy on those apps: Mobile medical apps gain support, but many lack clinical evidence (modernhealthcare.com)
Mobile medical applications increasingly are being used by patients and consumers. Now healthcare providers are evaluating whether and how to work with their patients in tapping these apps. But they're proceeding cautiously because of the dearth of clinical evidence for many consumer apps, and because some developers may be misleading consumers by overstating their products' capabilities…I think we will be seeing an increase in scrutiny and enforcement by the FDA and the FTC…medical devices treading in the areas of diagnosis and treatment, for those I think we'll see increased enforcement...healthcare providers, a major factor driving that adoption is the shift to value-based payment, which creates a powerful incentive to keep patients healthier and reduce costs by avoiding unnecessary hospitalizations and emergency department use…As with the introduction of any new clinical-care process, there are safety issues…We're going from a situation where we had no data to one where we probably have more data than we need…mobile apps can offer clinical benefits, particularly when used in a structured healthcare program…Healthcare lawyers caution that providers run a variety of legal risks in using mobile apps with patients. “If the patient brings in a bunch of stuff, if you rely on it and it's wrong, it's a problem”…“But if you ignore it and it's right, it's a problem. You're damned if you do and damned if you don't.”
- Law for rare disease drugs needs revamping, researchers say (statnews.com)The Orphan Drug Act: Restoring the Mission to Rare Diseases.(req sub) (ncbi.nlm.nih.gov)
…the Orphan Drug Act was passed to give drug makers incentives to create medicines for rare diseases, which are defined as maladies that affect fewer than 200,000 people. The incentives include tax credits and seven years of marketing exclusivity. Since then, more than 400 orphan drugs have been approved by the Food and Drug Administration. Last year, though, 41 percent of all FDA approvals were for orphan drugs. And sales of orphan medicines, which carry high price tags, are forecast this year to total $107 billion…team of researchers argues that drug makers are exploiting loopholes that allow them to widen the market for such drugs and distorting the original purpose of the law. We spoke with Martin Makary, a cancer surgeon and professor of health policy at Johns Hopkins School of Medicine…
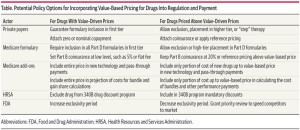
- Payer and Policy Maker Steps to Support Value-Based Pricing for Drugs (jama.jamanetwork.com)
Prescription drugs is the only major category of health care services for which the producer is able to exercise relatively unrestrained pricing power. By law, drug manufacturers can set the price that Medicare and Medicaid programs pay for new drugs, and they also benefit from significant negotiating advantages over private insurers, who are required to cover most new drugs and are unable to obtain significant price concessions from manufacturers, particularly for drugs that offer some clinical advantage or use alternative mechanisms of action compared with available treatment options. As a result, drug prices in the United States are generally 2 to 6 times higher than prices for the same drugs in other major industrialized nations…Now that value-based prices can be determined for drugs, insurers and policy makers can consider steps toward a value-driven pricing system that does not require Medicare negotiation, price setting, or mandates. What will be needed, however, is a shift toward providing an array of incentives to bolster the business case for drugmakers to sell their products at a price in alignment with the benefits to patients and sensitive to the potential budget effect on the entire health care system.
- Billion-dollar babies (economist.com)
The high cost of R&D is used to explain why drugs giants merge, and why they must charge high prices. The reality is somewhat different…Pfizer’s boss, put it, the merger will create “a leading global pharmaceutical company with the strength to research, discover and deliver more medicines and therapies to more people around the world.”…the common suggestion that size is needed to create a research-driven powerhouse does not stack up…the industry is…moving away from a model in which giant firms throw huge sums at in-house research in a quest for ground-breaking new treatments… rather than spending heavily in many different areas of cutting-edge research, the largest firms are increasingly buying in drugs that are already in the course of development. In some cases they do so by buying other established firms…the drugs giants are buying smaller, younger biotechnology firms which focus on a single-treatment approach…in the past 20 years those drug companies that consistently did well in various therapeutic areas were earning more than 70% of their sales from products developed elsewhere.
- Annual Specialty Therapies Conference; Jan. 28-29 | Planet Hollywood | Las Vegas, NV (drugchannels.net)Effectively Managing Specialty Therapies a Forum for Payers 2016 (cbinet.com)
CBI’s Specialty Therapies Forum, the conference that brings together healthcare and pharmaceutical thought leaders to share best practices for managing specialty therapies…an opportunity to discuss not only strategies for managing the cost of specialty therapies, but also to provide the latest information and case studies dedicated to ensuring access and quality care in specialty benefit management. New to the agenda this year is a workshop dedicated to Specialty Pipeline Management.
Dialogue led by key thought leaders:
Avella Specialty Pharmacy | Blue Cross Blue Shield of Massachusetts | Blue Cross and Blue Shield of Minnesota | Blue Cross and Blue Shield of North Carolina | Denver Health Medical Plan | Fairview Specialty Pharmacy | Florida Blue | Gorman Health Group | Horizon Blue Cross Blue Shield New Jersey | Independent Health | NSHOA Cancer Centers in NY | Rutgers RWJ Medical School | Walgreens | Walmart | Widener University