- Copay Maximizers Are Displacing Accumulators—But CMS Ignores How Payers Leverage Patient Support (drugchannels.net)
Last week, the Centers for Medicare and Medicaid Services released its final Notice of Benefit and Payment Parameters for the 2021 benefit year...This final rule permits insurers to exclude the value of a pharmaceutical manufacturer’s copay support program from a patient’s annual deductible and out-of-pocket maximum obligations...Translation: CMS has confirmed that insurers have the option to use copay accumulator adjustment for their pharmacy benefit programs...Patients on specialty drugs lose big from accumulators, while plans profit from the lower spending that results. Consequently, copay maximizers have emerged as a more patient-friendly alternative to accumulators...Plan sponsors are publicly denouncing copay support programs—while they’re privately embracing them. CMS’s final rule ignores the troubling reality behind maximizers and accumulators: They encourage plans to use pharmacy benefit deductibles as a scheme that allows payers—not patients—to reap the greatest benefits from a manufacturer’s patient support program...READ MORE
- The Latest CMS Outlook for Drug Spending—And How COVID-19 Will Change It (drugchannels.net)
...the Centers for Medicare & Medicaid Services recently released their new projections for U.S. National Health Expenditures. Unfortunately, the coronavirus almost immediately made these predictions obsolete...It’s still useful to analyze these forecasts for a pre-pandemic examination of U.S. healthcare spending. A few highlights of the 2024 outlook: READ MORE
- Total U.S. spending on healthcare was projected to grow, from $3.6 trillion in 2018 to $5.0 trillion in 2024.
- Spending on hospitals and professional services was expected to grow by a combined $800 billion—more than 60% of CMS’s projected $1.4 trillion increase in U.S. healthcare spending. That’s consistent with historical trends.
- Net spending on outpatient prescription drugs in 2024 was projected to shrink to less than 9% of total U.S. spending. That would be its lowest level since 2000.
- CMS releases new flexibility, waiver protections for providers to help handle coronavirus (fiercehealthcare.com)
The Trump administration has issued blanket waivers and new flexibilities to help hospitals and facilities cope with the coronavirus outbreak...“It is vital that federal requirements designed for periods of relative calm do not hinder measures needed in an emergency,” CMS Administrator Seema Verma said in a statement. “The nationwide waivers we are activating today will be a godsend for those on the frontlines of the fight against this new virus.”...Some of the waivers and flexibilities that CMS took on Friday include:
- Waiving requirements that critical access hospitals limit the number of beds to 25 and length of stay to 96 hours;
- Enabling acute care hospitals to house acute care patients in a separate unit;
- Waiving replacement requirements when durable medical equipment gets damaged or unusable. A contractor can waive requirements such as a new physician’s order, face-to-face requirement and other documentation; and
- Allowing long-term care hospitals to exclude patients stays from the 25-day average length requirement if treatment is required in conjunction with the emergency.
- CMS also sought to quickly approve waivers for states and territories for Medicare, Medicaid and the Children’s Health Insurance Program...READ MORE
- Proposed Medicare Changes Aim to Provide Improved Coverage, Access, Transparency (pharmacytimes.com)Proposed Medicare Changes Aim to Provide Improved Coverage, Access, Transparency (drugchannels.net)
The Centers for Medicare & Medicaid Services has issued a proposed rule and the Advance Notice Part II to further advance the agency’s efforts to strengthen and modernize the Medicare Advantage and Part D prescription drug programs. These changes would lower the beneficiary cost sharing on more expensive prescription drugs, promote the use of generic medications, and allow beneficiaries to know in advance and compare their out-of-pocket payments for different prescription drugs...The proposed rule would require Part D plans to offer real-time drug price comparison tools to beneficiaries starting January 1, 2022 so that consumers can buy lower-cost alternative therapies under their prescription drug benefit plan...READ MORE
- HHS files appeal to reinstate controversial MA overpayment rule (fiercehealthcare.com)
The Department of Health and Human Services has filed an appeal to reinstate a key rule that handles overpayments to Medicare Advantage plans, arguing a lower court's ruling was based on a flawed premise...The appeal, filed Thursday in the federal Court of Appeals for the District of Columbia Circuit, could decide how much money MA plans, an increasingly lucrative market for insurers, will have to give back to Medicare for diagnosis errors. HHS argues that an earlier ruling striking down the plan misunderstood how Medicare audits MA plans...The appeal is the latest salvo in a legal fight with insurers over a 2014 rule proposed by the Centers for Medicare & Medicaid Services...READ MORE
- DOJ sues Anthem over Medicare Advantage fraud claims (fiercehealthcare.com)
Anthem faces a federal lawsuit for submitting inaccurate diagnostic data to get a higher Medicare reimbursement...The U.S. Attorney for the Southern District of New York said in the lawsuit...that Anthem failed to find and delete inaccurate diagnosis codes from 2014 to early 2018 through its “chart review program,”...“By ignoring its duty to delete thousands of inaccurate diagnoses, Anthem unlawfully obtained and retained from [Centers for Medicare & Medicaid Services] millions of dollars in payments under the risk adjustment payment system for Medicare Part C,” the lawsuit said...READ MORE
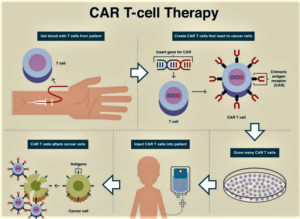
- CAR-T—the Future of Medical Progress Is Now (realclearhealth.com)
Personalized medicine is the future of medical progress...For instance, an immunotherapy treatment called chimeric antigen receptor T-cell therapy is accessible now, and the Trump administration has the opportunity to make it widely available to Medicare beneficiaries...over 70 Members of Congress—Democrats, Republicans, liberals, and conservatives—sent a letter to Seema Verma, administrator of the Centers for Medicare and Medicaid Services, commending the administration for “ensuring Medicare patients nationwide have access” to this life-saving treatment. The Congressional letter goes on to ask the administration to “ensure that hospitals are appropriately reimbursed so they may continue to provide” CAR-T therapy to America’s seniors...without appropriate reimbursement policy, a Medicare patient could be denied access to a treatment that would save his or her life. Without proper reimbursement by Medicare, providers simply will not be able to offer it as an option, especially in rural areas as patients must stay near a treatment center for four weeks to be monitored...READ MORE
- PhRMA submits comments on CMS’s Part D specialty tier proposal (catalyst.phrma.org)PhRMA Comments on Part D Rule, April 2020 [CMS-4190-P] (phrma.org)
...PhRMA submitted comments on a Centers for Medicare & Medicaid Services proposed rule on policy and technical changes to Medicare Part D and Medicare Advantage...part of the proposed rule, CMS is seeking to allow Part D plan sponsors to establish formularies with up to two specialty tiers. If there are two specialty tiers, one must be a “preferred” tier that offers lower cost-sharing than the proposed maximum allowable specialty tier cost sharing. Consistent with CMS’s current policy, beneficiaries would not be able to appeal to access medicines from either specialty tier at preferred tier cost-sharing rates...PhRMA has expressed concern with the specialty tier and the impacts the policy has on beneficiaries’ ability to access and afford needed medicines that happen to meet the specialty tier criteria. The new proposal for a second specialty tier is a step in the wrong direction for the Part D program...READ MORE
- CMS Approves More Medicaid Section 1135 Waivers, Bringing Total to 34 States (pharmacytimes.com)
Following the first approved Medicaid section 1135 waiver for Florida, the Centers for Medicare and Medicaid Services has approved 33 more waiver requests in the last 2 weeks....The waivers are intended to provide the states with relief during the coronavirus disease 2019 pandemic by providing states the flexibility to focus resources on managing the outbreak. Some waivers available under Section 1135 of the Social Security Act include temporary suspension of prior authorization requirements; extension of existing authorizations; modified timeline requirements for state fair hearings and appeals; and relaxed provider enrollment requirements to allow states to quickly enroll out-of-state or other new providers...READ MORE
- NACDS Voices Support for CMS Pharmacy Quality Proposal (drugtopics.com)
NACDS is welcoming “the strongest signal yet” by the Centers for Medicare & Medicaid Services that a standardized pharmacy quality program should be created...In a new proposed rule, CMS proposed taking action to increase transparency in Medicare Part D plan sponsors’ pharmacy performance measures, NACDS said in a press release Plan sponsors would have to disclose these measures and CMS would publish them, according to the proposal...The CMS rule also recommends principles to guide pharmacy performance measures, urges the industry to work together toward appropriate measures, and indicates the possibility that CMS may incent plans to use appropriate quality measures, NACDS said...READ MORE