- The Latest CMS Outlook for Drug Spending—And How COVID-19 Will Change It (drugchannels.net)
...the Centers for Medicare & Medicaid Services recently released their new projections for U.S. National Health Expenditures. Unfortunately, the coronavirus almost immediately made these predictions obsolete...It’s still useful to analyze these forecasts for a pre-pandemic examination of U.S. healthcare spending. A few highlights of the 2024 outlook: READ MORE
- Total U.S. spending on healthcare was projected to grow, from $3.6 trillion in 2018 to $5.0 trillion in 2024.
- Spending on hospitals and professional services was expected to grow by a combined $800 billion—more than 60% of CMS’s projected $1.4 trillion increase in U.S. healthcare spending. That’s consistent with historical trends.
- Net spending on outpatient prescription drugs in 2024 was projected to shrink to less than 9% of total U.S. spending. That would be its lowest level since 2000.
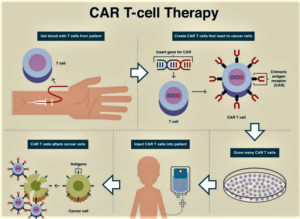
- CAR-T—the Future of Medical Progress Is Now (realclearhealth.com)
Personalized medicine is the future of medical progress...For instance, an immunotherapy treatment called chimeric antigen receptor T-cell therapy is accessible now, and the Trump administration has the opportunity to make it widely available to Medicare beneficiaries...over 70 Members of Congress—Democrats, Republicans, liberals, and conservatives—sent a letter to Seema Verma, administrator of the Centers for Medicare and Medicaid Services, commending the administration for “ensuring Medicare patients nationwide have access” to this life-saving treatment. The Congressional letter goes on to ask the administration to “ensure that hospitals are appropriately reimbursed so they may continue to provide” CAR-T therapy to America’s seniors...without appropriate reimbursement policy, a Medicare patient could be denied access to a treatment that would save his or her life. Without proper reimbursement by Medicare, providers simply will not be able to offer it as an option, especially in rural areas as patients must stay near a treatment center for four weeks to be monitored...READ MORE
- Nevada’s 2019 Hospital Safety Rankings Are Out
Leapfrog’s highest “A” rated hospitals for 2019 has remained unchanged from last year. These are Henderson Hospital, Mountainview Hospital, Northern Nevada Medical Center, Renown South Meadows Medical Center, and St. Mary's Regional Medical Center of Reno.
The Leapfrog Group hospital grading system is based on Centers for Medicare & Medicaid Services data collection and represents a single metric that evaluates a hospital’s overall safety performance. The nonprofit is dedicated to transparency and has created the Leapfrog Hospital Safety Grade as a quality standard for comparing health care institutions.
The “B” team consists of North Vista Hospital, Valley Hospital Medical Center, St. Rose Dominican Hospitals - Siena Campus, St. Rose Dominican Hospitals - San Martin Campus, and Renown Regional Medical Center.
The good news is two of these hospitals have shown a marked safety improvement by moving up from a “C” rating to a “B” rating; these are: Valley Hospital Medical Center, and Renown Regional Medical Center.
These hospitals are to be applauded for consistently maintaining high standards and striving to improve patient care.
The bad news is the University Medical Center of Southern Nevada continues to struggle with its’ “D” rating.
By Chase
12.30.2019
- Regulatory agenda lays out timetable for major rules on drug pricing, interoperability (fiercehealthcare.com)
The Trump administration estimates it will release rules on drug prices and information blocking this month but punted finalizing a rule on interoperability until as late as 2022..The White House Office of Management and Budget released...the unified regulatory agenda for fall 2019...The agenda said that a major rule that would outlaw information blocking among providers is estimated to be released this month...The unified agenda also gives an update on key regulations addressing high drug prices...One such proposal is a demonstration model that would tie the price of physician-administered drugs paid by Medicare Part B to an index made up of cheaper prices paid by other countries...The agenda also provides an estimated release date of January 2020 for a proposed rule to allow the importation of drugs from Canada. The proposed rule would create a pilot project to allow states, wholesalers and pharmacists to import drugs from Canada at a cheaper price than in the U.S...READ MORE
- DOJ sues Anthem over Medicare Advantage fraud claims (fiercehealthcare.com)
Anthem faces a federal lawsuit for submitting inaccurate diagnostic data to get a higher Medicare reimbursement...The U.S. Attorney for the Southern District of New York said in the lawsuit...that Anthem failed to find and delete inaccurate diagnosis codes from 2014 to early 2018 through its “chart review program,”...“By ignoring its duty to delete thousands of inaccurate diagnoses, Anthem unlawfully obtained and retained from [Centers for Medicare & Medicaid Services] millions of dollars in payments under the risk adjustment payment system for Medicare Part C,” the lawsuit said...READ MORE
- Sanofi, Justice Department ink $11.85M charity kickbacks settlement (fiercepharma.com)
A federal probe of pharma’s charity contributions has secured settlements from drugmakers large and small, and Sanofi is the latest to ink a deal with the government over allegations its payments were kickbacks...Sanofi agreed to pay $11.85 million to resolve allegations that it used The Assistance Fund as a “conduit” to help multiple sclerosis patients pay their Medicare co-pays for Lemtrada...The drug costs nearly $100,000 per year, and patient co-pays can run thousands of dollars per year...“Sanofi sought to undermine the Medicare program through its use of kickbacks disguised as routine charitable donations aimed at helping patients battling multiple sclerosis and who were struggling with costly copays,”...READ MORE
- U.S. sues CVS for fraudulently billing Medicare, Medicaid for invalid prescriptions (reuters.com)
CVS Health Corp and its Omnicare unit were sued...by the U.S. government, which accused them of fraudulently billing Medicare and other programs for drugs for older and disabled people without valid prescriptions...The Department of Justice joined whistleblower litigation accusing Omnicare of violating the federal False Claims Act for illegally dispensing drugs to tens of thousands of patients in assisted living facilities, group homes for people with special needs, and other long-term care facilities...According to a civil complaint filed in Manhattan federal court, Omnicare would often assign new numbers to prescriptions after the original prescriptions expired or ran out of refills...The government said this enabled Omnicare to bill Medicare Medicaid, and Tricare...for hundreds of thousands of drugs, under what the company internally called “rollover” prescriptions, from 2010 to 2018...READ MORE
- CMS releases new flexibility, waiver protections for providers to help handle coronavirus (fiercehealthcare.com)
The Trump administration has issued blanket waivers and new flexibilities to help hospitals and facilities cope with the coronavirus outbreak...“It is vital that federal requirements designed for periods of relative calm do not hinder measures needed in an emergency,” CMS Administrator Seema Verma said in a statement. “The nationwide waivers we are activating today will be a godsend for those on the frontlines of the fight against this new virus.”...Some of the waivers and flexibilities that CMS took on Friday include:
- Waiving requirements that critical access hospitals limit the number of beds to 25 and length of stay to 96 hours;
- Enabling acute care hospitals to house acute care patients in a separate unit;
- Waiving replacement requirements when durable medical equipment gets damaged or unusable. A contractor can waive requirements such as a new physician’s order, face-to-face requirement and other documentation; and
- Allowing long-term care hospitals to exclude patients stays from the 25-day average length requirement if treatment is required in conjunction with the emergency.
- CMS also sought to quickly approve waivers for states and territories for Medicare, Medicaid and the Children’s Health Insurance Program...READ MORE
- NACDS Voices Support for CMS Pharmacy Quality Proposal (drugtopics.com)
NACDS is welcoming “the strongest signal yet” by the Centers for Medicare & Medicaid Services that a standardized pharmacy quality program should be created...In a new proposed rule, CMS proposed taking action to increase transparency in Medicare Part D plan sponsors’ pharmacy performance measures, NACDS said in a press release Plan sponsors would have to disclose these measures and CMS would publish them, according to the proposal...The CMS rule also recommends principles to guide pharmacy performance measures, urges the industry to work together toward appropriate measures, and indicates the possibility that CMS may incent plans to use appropriate quality measures, NACDS said...READ MORE
- Part B Update: Hospitals Displacing Physicians, Amid Slow Growth in Drug Prices (drugchannels.net)
The Medicare Payment Advisory Commission (MedPAC), the independent agency that advises Congress on the Medicare program, recently released its June 2019 Data Book: Health Care Spending and the Medicare Program...This year’s report provides the latest details about the ongoing disruption of the buy-and-bill market in Medicare Part B. As you will see below:
- Physician offices account for a diminishing share of Part B spending, though absolute spending at these sites continues to grow.
- Hospital outpatient settings have been crowding out physician offices. Hospital outpatient departments now account for more than 40% of Part B spending—and an even greater share for oncology products...READ MORE