- July 5 Pharmacy Week in Review: Vitamin D Deficiency Linked to Elevated Blood Pressure in Children, Chronic Pain is the Most Common Long-Term Effect of Cancer Treatment (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
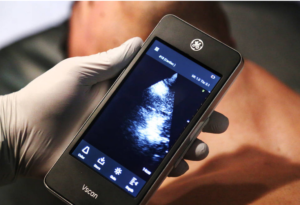
- CSN paramedic students 1st in state to train on portable ultrasound (reviewjournal.com)
College of Southern Nevada paramedic students are the first in the state to begin training on portable ultrasound machines that can help diagnose traumatic bleeding long before a patient arrives at an emergency room...Students in uniform practiced with the handheld devices for the first time...The devices, which will help first responders determine how to care for patients and where to send them, can also communicate patient information to emergency room doctors during transport, expediting the arrival process...“If we have a decision to make in the field, one hospital may not have the trauma surgery capabilities that are needed,” said Braiden Green, CSN’S program director of emergency medical services. “This allows us to decrease the amount of times…that a patient is moved, to get the patient to definitive care faster and improve patient outcomes.”...In rural Nevada, an on-the-spot diagnosis could determine whether a helicopter is needed, Green said, one big reason that CSN decided to pursue this technology...READ MORE
- Judge approves Teva’s $85 million opioid settlement with state of Oklahoma (cnbc.com)Suit: Generic drug makers used code to fix price increases (apnews.com)
An Oklahoma judge...approved Teva Pharmaceuticals’ revised $85 million settlement with the state over allegations the company helped fuel the nationwide opioid epidemic...decision came after state legislative leaders argued an earlier agreement did not comply with new state laws. Teva had reached an agreement with the state of Oklahoma last month, just two days before the drugmaker was set to face a trial alongside Johnson & Johnson...READ MORE
- New drug to boost women’s sex drive approved in US (apnews.com)AMAG wins FDA nod for Vyleesi. Could it be the ‘female Viagra’ that Addyi never was? (fiercepharma.com)
U.S. women will soon have another drug option designed to boost low sex drive: a shot they can give themselves in the thigh or abdomen that raises sexual interest for several hours...The medication OK’d...by the Food and Drug Administration is only the second approved to increase sexual desire in a women, a market drugmakers have been trying to cultivate since the blockbuster success of Viagra for men...The upside of the new drug “is that you only use it when you need it,”...“The downside is that it’s a shot — and some people are very squeamish.”...The FDA approved the new drug, Vyleesi (pronounced vie-LEE’-see), for premenopausal women with a disorder defined by a persistent lack of interest in sex, causing stress. The most common side effect in company studies was nausea...READ MORE
- Albertsons pharmacy technicians first in Idaho trained to prescribe naloxone (drugstorenews.com)
As a new Idaho law allowing pharmacy technicians to prescribe naloxone takes effect, Albertsons announced that its Boise-area pharmacy technicians are the first in the state who have been trained to do so...The law, signed in February, makes Idaho the first state in the nation to add pharmacy technicians to the ranks of pharmacists, physicians and nurse practitioners in being able to prescribe the opioid overdose reversal drug. Idaho Gov. Brad Little also issued an executive order creating an advisory group in the state to guide healthcare decisions and strategies related to opioid misuse...“Idaho is leading other states in the steps we are taking to expand access to this critical medication and in our efforts to address the opioid crisis,” Little said. “Training pharmacy technicians and all health professionals to prescribe naloxone further reaches our underserved and rural communities. Albertsons has been a key partner in increasing the scope of practice for both pharmacists and technicians to continue improving access to beneficial and lifesaving medications.”...READ MORE
- This Week in Managed Care: June 28, 2019 (ajmc.com)
Samantha DiGrande, Welcome to This Week in Managed Care from the Managed Markets News Network
- FDA’s overreach will harm compounding pharmacies and the patients they serve (statnews.com)
The deaths of 64 people and sickening of nearly 800 due to criminal negligence by employees of the New England Compounding Center in 2012 marked a profound failure of state and federal regulatory enforcement…led to…the Drug Quality and Security Act of 2013 provisions to create a more robust regulatory framework for compounding pharmacies…The legislation instructed the FDA to create regulations…that would assure patient safety while permitting local compounders to continue to meet patient needs by providing customized compounded medications using FDA-approved substances…But the FDA has overreached in implementing the provisions, all but halting common compounding practices that have been safely performed for years and on which patients with legitimate needs for compounded medications rely. Not only that, but the FDA has done so by circumventing the federal Administrative Procedure Act, issuing “guidance documents” to implement policies rather than following the statutory rule-making process that requires stakeholder input regarding proposed regulations…READ MORE
- Former FDA head Gottlieb joins Pfizer board (biopharmadive.com)Warren blasts Gottlieb's jump to Pfizer, but ex-FDA chief says he's 'proud' (fiercepharma.com)
Pharma giant Pfizer...named former Food and Drug Administration Commissioner Scott Gottlieb to its board of directors, less than three months after he left his post at the agency...Since departing the FDA, Gottlieb has also rejoined the American Enterprise Institute, a Washington, D.C. think tank, and New Enterprise Associates, a venture capital firm, both organizations he worked at before becoming FDA chief...Former commissioners frequently join industry boards following their tenure at the FDA. Robert Califf, Gottlieb's immediate predecessor, serves on the board of Cytokinetics, a small biotech developing drugs for neuromuscular and heart diseases...READ MORE
- June 28 Pharmacy Week in Review: PBM Reform in 1 State Could Have Implications on Federal Reform, Study Shows Medical Marijuana Does Not Reduce Fatal Opioid Overdoses (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- New 340B Health Reports Confirms the Program’s Size—But Double-Dips on Hospitals’ (drugchannels.net)EVALUATING 340B HOSPITAL SAVINGS AND THEIR USE IN SUPPORTING CARE FOR RURAL AND LOW-INCOME PATIENTS (340bhealth.org)
340B Health, which lobbies for hospitals that participate in the 340B Drug Pricing Program, recently issued an eye-opening member survey. It found that 340B Health’s member hospitals reported total 340B discounts of $15.3 billion...The 340B Health report also claims to show evidence that hospitals are properly using 340B funds...In reality, the report merely affirms that hospitals are spending money to meet their fundamental legal and statutory community benefit obligations. Any money from 340B discounts shouldn’t be double-counted to meet hospitals’ fundamental spending requirements...This latest report therefore again highlights the need for transparency and accountability to ensure that billions in 340B discounts are shared with the patients whose prescriptions generated those funds...READ MORE