- This Week in Managed Care: June 21, 2019 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- EU industry loses €16.5bn in sales to counterfeit drugs (in-pharmatechnologist.com)2019 STATUS REPORT ON IPR INFRINGEMENT (euipo.europa.eu)
A report from the EUIPO demonstrated that a shift in the focus of counterfeited goods to include pharmaceutical products has caused significant loss of sales and employment...The European Union Intellectual Property Office has released a report this month regarding 11 analysed sectors in the region, including the pharmaceutical industry...The European Union organisation found that all analysed sectors lost sales as a result infringement of intellectual property rights for a total of €92 bn ($106bn) between 2013 and 2916...the pharma industry suffered the second biggest loss of sales, at €16bn…READ MORE
- Las Vegas Valley vets may benefit from new VA access program (reviewjournal.com)
A Department of Veterans Affairs program launched last week will offer military veterans new options in accessing health care...The Maintaining Internal Systems and Strengthening Integrated Outside Networks Act, or Mission Act, essentially expands on earlier legislation intended to cut down on wait times at VA medical facilities by allowing veterans to see private doctors in some circumstances...“It was a temporary fix at that time to deal with wait times and scheduling issues,” Chuck Ramey, spokesman for the the VA Southern Nevada Healthcare System in North Las Vegas...“The VA has been working with the community to provide community care since day one.”...Under the new program...veterans can work with their VA health care provider or other agency staffers to see if they are eligible to receive care from local non-VA doctors...Dr. Ramu Komanduri, chief of staff at the VA facility in North Las Vegas, said the new act “offers many more opportunities for the veteran to make a choice.”...READ MORE
- This Week in Managed Care: June 14, 2019 (ajmc.com)
Samantha DiGrande, Welcome to This Week in Managed Care from the Managed Markets News Network
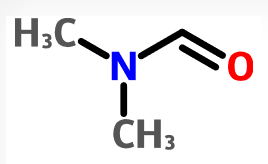
- Pharmacy takes FDA to task in citizen petition over tainted valsartan (fiercepharma.com)
The FDA has tied the contamination of blood pressure drugs by probable carcinogens to an approved switch in manufacturing to a process that uses certain solvents. Now an online pharmacy says it has discovered the solvents are as bad as the impurities they have been creating and has criticized the FDA for not taking steps to limit their use...In a citizen petition...Valisure says the FDA has established acceptable limits for impurities such as those commonly known as NDMA and NDEA after they were discovered in heart drugs last year, leading to massive global recalls...But it claims the FDA has not lowered the acceptable level for the solvent DMF, although it “has become apparent that the switch in the manufacturing industry to the use of the DMF solvent may be largely responsible” for the appearance of the impurities in the U.S. drug supply...The petition says the FDA needs to investigate the use of the solvent N,N-Dimethylformamide in drug manufacturing since it is classified as a Class 2 carcinogen...READ MORE
- NACDS applauds new Texas law to help curb opioid abuse (drugstorenews.com)
The National Association of Chain Drug Stores today praised Texas’ enactment of a bill (HB 2174) to address opioid abuse and addiction, while providing key safeguards to address the needs of those suffering from chronic pain...the new law will require electronic prescribing for controlled substances, to help prevent fraud and abuse. It also will limit the supply of a patient’s first opioid prescription to 10 days, when that prescription is for temporary, or acute, pain. It is important to note that this limit does not apply to prescriptions for ongoing, or chronic, pain. It also does not apply to cancer care, treatment of other illnesses, or end-of-life care...READ MORE
- Canadian panel calls for universal public drug coverage (reuters.com)
A Canadian advisory council studying prescription drug coverage said...the federal government should create a C$15.3 billion ($11.5 billion) universal, single-payer public pharmacare system, and warned that the current system requires a major overhaul...Canada is the only country with a universal health care system that does not include universal coverage for prescription drugs. Most prescriptions are paid for through employer-funded drug plans, while some are covered by government programs for the elderly, or people with low incomes or very high costs...If implemented in full, the plan would likely cut into profits of insurers and drugmakers in Canada, while saving employers and patients money...READ MORE
- June 21 Pharmacy Week in Review: Study Links BMI and Psoriatic Arthritis Severity, Survival Disparities Growing Among Young Patients with Cancer (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Nevada broadens lawsuit against opioid manufacturers, retailers (reviewjournal.com)
Nevada Attorney General Aaron Ford filed a lawsuit...in Clark County District Court accusing dozens of drug manufacturers and distributors of propagating the state’s deadly opioid epidemic...The 241-page complaint names more than 40 defendants, including prescription drug makers such as Purdue Pharma and Teva Pharmaceuticals, and distributors including CVS, Walgreens and retail giant Walmart...It accuses drug manufacturers of pushing doctors to prescribe the addictive medication while downplaying the side effects. It also alleges that distributors supplied many more opioids than justified for the Nevada market, ignoring data that allowed them to track overprescribing...READ MORE
- CMS approves Washington request for ‘Netflix’ model to pay for hepatitis C drugs (fiercehealthcare.com)
The Centers for Medicare & Medicaid Services approved a request...from Washington state to negotiate value-based drug rebate agreements with pharmaceutical manufacturers in its Medicaid program...The approval makes Washington the fourth state to test such an arrangement; similar policies have already been given the OK in Oklahoma, Michigan and Colorado, according to an announcement from CMS...Washington officials are aiming to test a “subscription” model for hepatitis C drugs...In this model, Medicaid would pay a fixed annual sum to a drug company for the hepatitis C medication, purchasing an unrestricted supply of the drug... CMS is committed to increasing states’ flexibility to develop policies that lower costs, increase the predictability of expenses and improve access for patients...READ MORE