- FDA Commissioner Gottlieb, who raised alarms about teen vaping, resigns (washingtonpost.com)
Food and Drug Commissioner Scott Gottlieb...resigned...effective in about a month...The 46-year-old physician, millionaire and cancer survivor known for a self-assured, sometimes brash, manner lives in Westport, with his wife and three daughters – 9-year-old twins and a 5-year-old...“It was a very hard decision,” Gottlieb said in an interview. “This is the best job I will ever have. I’m leaving because I need to spend time with my family. I get home late Friday, work on weekends and come back to Washington on Sunday. I did the job 100 percent."...The resignation took some senior FDA officials by surprise, and came as Gottlieb’s signature issue – youth vaping – is being reviewed by the White House Office of Management and Budget. The plan, detailed by Gottlieb last fall, would sharply restrict the sale of flavored e-cigarettes to curb a surge in underage vaping, which he argues could lead to a whole new generation addicted to nicotine...READ MORE
- NACDS, NCPA brief Congressional Doctors Caucus on DIR fee reform (chaindrugreview.com)
NACDS and the National Community Pharmacists Association held a briefing this week for the Congressional Doctors Caucus...chaired by Rep. Phil Roe...comprised of 16 medical providers in Congress who use their medical expertise to develop patient-centered healthcare policy...NACDS and NCPA discussed the Centers for Medicare & Medicaid Services’ recently released proposed rule—Modernizing Part D and Medicare Advantage to Lower Drug Prices and Reduce Out-of-Pocket Expenses—and explained the serious repercussions of direct and indirect remuneration (DIR) fees and the impact they can have on patient access to care and on pharmacies, who are struggling to maintain their businesses amid the uncertainty caused by the retroactive fees. The groups noted that they have advocated strongly for DIR reform in the CMS proposal because it will benefit pharmacies and the patients they serve.
- Executives from seven major drug companies will be grilled about the high cost of their medications at a Senate panel on Tuesday morning. (ksat.com)

Richard Gonzalez, Pascal Soriot, Giovanni Caforio, Jennifer Taubert, Kenneth Frazier, Albert Bourla, and Olivier Brandicourt
The hearing is the latest volley in Washington's battle against soaring drug prices, which has become a top priority for the Trump administration, as well as many Democratic and some Republican lawmakers...Executives from companies including Pfizer, Johnson & Johnson and AbbVie, will appear before the Senate Finance Committee and likely face tough questions from the panel's new chairman, Republican Sen. Chuck Grassley of Iowa...
- Congressman aims to break pharma’s insulin ‘pricing monopoly’ by legalizing Canadian imports (fiercepharma.com)
U.S. legislators have been introducing bills left and right proposing a variety of methods for bringing down drug prices. Now, Vermont's congressman is zeroing in on a specific class of diabetes medicines that he says have become far too expensive: insulin...Rep. Peter Welch introduced a bill on Wednesday that would make it legal for patients, wholesalers and pharmacists to import insulin from Canada, and eventually from other countries with acceptable safety standards...Welch clearly wants to make an end run around Lilly and its fellow insulin makers. If his bill were to become law, patients with valid prescriptions would be able to import low-cost insulin and have it covered by their insurance plans...
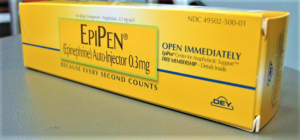
- Pfizer unit Meridian under civil investigation by U.S. Attorney (reuters.com)
Pfizer Inc said...it received a request for documents as part of a U.S. investigation related to quality issues involving the manufacture of auto-injectors at its Meridian Medical Technologies site...Meridian, a unit of Pfizer that manufactures EpiPen injectors...has been hit by a series of manufacturing problems in recent years...In 2017, Meridian had received a warning letter from the Food and Drug Administration. The FDA said Meridian had failed to thoroughly investigate product failures, including EpiPen products that were associated with patient deaths and severe illnesses. It said the company failed to take corrective actions until FDA’s inspection...
- This Week in Managed Care: March 1, 2019 (ajmc.com)
, welcome to This Week in Managed Care from the Managed Markets News Network
Jaime Rosenberg - HHS’s Multi-Pronged Approach To Lower Drug Prices: Will It Work? (forbes.com)
...the Department of Health and Human Services has put forward a series of initiatives aimed at lowering prescription drug prices, Medicare expenditures, and patient cost-sharing. The proposals run the gamut from increasing transparency of net pricing, to allowing the use of certain formulary management tools previously off limits to Medicare plans, to direct price controls...But, will they work? The federal government appears to think so. Several weeks ago, Secretary of HHS Alex Azar stated that "the models being announced create new incentives for plans, patients, and providers to choose drugs with lower list prices...[advancing] our priority of using HHS programs to build a value-driven healthcare system."...it's debatable whether HHS will achieve the twin objectives of lower drug spending and decreased patient cost-sharing as a consequence of implementing its series of initiatives. Some may not get implemented as planned, or not at all given the opposition they evoke. Others may result in tempering the growth in drug costs overall, but raising the amount patients must spend out-of-pocket, whether on premiums or patient cost-sharing...
- China slashes tax to stimulate domestic orphan drug market (pharmaceutical-technology.com)
China has announced a plan to slash the Value Added Tax on 21 rare disease therapies and four active pharmaceutical ingredients, dropping it by 80% in a bid to stimulate the domestic rare disease therapy market and drop the prices of the therapies...China is currently suffering from a shortage of rare disease therapies; of the 44 globally available rare disease therapies, only 19 are currently marketed within China, and of these 19, all are priced out of reach for the average Chinese patient. And with an estimated population of nearly 20 million people suffering from a rare disease within China, this is a huge area of unmet need...
- March 1 Pharmacy Week in Review: Thousands of Lives Saved Due to Improvements in Breast Cancer Treatment, Sesame Allergies Are Increasing (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- FDA oversight of the prescribing of fentanyl products is inadequate, report finds (healthcarefinancenews.com)Assessment of the FDA Risk Evaluation and Mitigation Strategy for Transmucosal Immediate-Release Fentanyl Products (jamanetwork.com)
The Food and Drug Administration and manufacturers did not take action when evidence emerged that potentially lethal fentanyl products were being inappropriately prescribed to patients, new research...shows...even as evidence emerged that as many as half of patients were taking dangerous medications known as TIRFs that should never have been prescribed to them, the FDA and fentanyl makers did not review prescribing records of even a single physician to consider disqualifying them from the program, which would have prevented them from prescribing the products...The study focused on Transmucosal Immediate-Release Fentanyls, or TIRFs, which are more dangerous than most prescription opioids on the market due to their very high potency and rapid onset. TIRFs are designed to get into the bloodstream within seconds, and because of their risks, were approved by FDA only for adult cancer patients "who are already receiving and who are tolerant to opioid therapy for their underlying persistent cancer pain."