- Bristol-Myers Squibb must face whistleblower suit claiming underpaid Medicaid rebates (fiercepharma.com)
Years after a former pharmacist sued Bristol-Myers Squibb and other companies for allegedly underpaying Medicaid rebates, a federal court rejected Bristol's attempt to escape the case...Pharmacist and lawyer Ronald Streck filed a False Claims Act whistleblower lawsuit against Bristol-Myers and other companies back in 2013, but later withdrew his claims against the other defendants. Now, his lawsuit alleges that BMS fraudulently manipulated its average manufacturer prices to underpay Medicaid rebates from 2007 to 2016...Pennsylvania federal judge Timothy Savage ruled last week that Streck "has alleged sufficient facts to state a false claims cause of action."...
- As state awaits data from diabetes drug manufacturers, initial report highlights price increases (thenevadaindependent.com)
Pharmaceutical companies are preparing to submit their initial reports detailing why rising prices of some diabetes drugs have outpaced medical inflation, giving state officials the first detailed look into the costs associated with a disease that affects about a tenth of Nevadans...Despite a protracted legal battle...pharmaceutical manufacturers are required to submit reports...to comply with a new diabetes drug transparency law… the state released a list of 175 so-called national drug codes...manufacturers...are required to submit reports to the state detailing the factors that contributed to the price increases...
- UK vows to speed up drug review in return for 2% sales cap (in-pharmatechnologist.com)
UK government and ABPI have agreed a deal to limit branded drug sales growth to 2%, potentially reducing the cost of medicine by £930m ($1.18b)...The announcement was made after the UK government and the Association of the British Pharmaceutical Industry provisionally signed the Voluntary Scheme for Branded Medicines Pricing and Access...the two key details released are: branded medicines will be subject to a 2% cap on sales growth – with pharma companies repaying the NHS for sales over this limit – and appraisals of new technology applications by NICE will be completed up to six months faster than current timelines...
- Florida’s opioid lawsuit against CVS and Walgreens takes aim at distributors with deep pockets (cnbc.com)
Florida Attorney General Pam Bondi has added Walgreens and CVS Health as defendants in the state's massive lawsuit against the opioid industry...Legal analysts say Florida and other plaintiffs are targeting the distributors and pharmacies, in part, because they have deep pockets...The...lawsuit accuses the drug stores and pharmaceutical distributors like Cardinal Health, Amerisource Bergen and McKesson of playing as big a role in the proliferation of opioid addiction as drug manufacturers like...Purdue Pharmaceuticals and Johnson & Johnson's Janssen Pharmaceuticals...
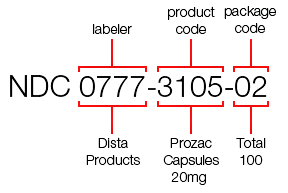
- The NDC Shortage: What the FDA Could (and Should) Do to Address It (drugchannels.net)An Open Letter To The FDA: New NDC Format Public Meeting (rxtrace.com)
...the U.S. Food & Drug Administration will run out of 5-digit National Drug Code codes within the next ten to fifteen years…The problem now facing U.S. healthcare is that, due to an explosion of new labelers entering the market, the FDA is running out of 5-digit labeler codes. The 5-digit format provides 90,000 potential combinations, and although that seems like a lot, the FDA anticipates running out of labeler codes within the next 10-15 years...the disruption and impact to the healthcare industry will be widespread and of a large magnitude, and will require retooling of major industry systems...
- This Week in Managed Care: November 30, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Elko cardiologist pleads guilty to opioid charges (kolotv.com)
An Elko cardiologist has pleaded guilty to distributing highly-addictive prescription drugs...to patients without a medical purpose...59-year-old Dr. Devendra I. Patel...who owns and operates Northeastern Nevada Cardiology, was indicted by a grand jury in December 2017...The investigation showed Patel illegally prescribed opioids and other prescription narcotics to patients for financial gain. As part of his plea, Patel admitted that, between September 2015 and February 2016, he prescribed Oxycodone and Hydrocodone to patients without a legitimate medical purpose and outside the usual course of professional practice...The statutory maximum penalty is 20 years in prison and a $1,000,000 fine.
- Walmart and Aurobindo sued as litigation mounts over contaminated blood pressure drugs (fiercepharma.com)
Litigation is growing over tainted blood pressure medications with a class-action lawsuit now filed in Florida against Walmart and three drug companies that span the supply chain...In addition to the retailer, the lawsuit names Indian drugmaker Aurobindo, U.S. drugmaker ScieGen Pharmaceuticals and distributor Westminster Pharmaceuticals. It was filed in federal court in Florida against the four over the sale of irbesartan because the drug contained the impurity, N-nitrosodiethylamine...Hauppauge...and its distributor Westminster initiated a nationwide voluntary recall to the consumer level of all lots of unexpired irbesartan tablets because they had been made with the Aurobindo API and then shipped to Walmart. The FDA learned this summer that the suspected carcinogen...can show up in “sartan”-based drugs as a result of certain manufacturing processes...
- November 30 Pharmacy Week in Review: Trial Evaluating Effectiveness and Safety of Drugs Used to Treat Patients with Ebola (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Nevada Medicaid to pause enrollment of mental health providers (reviewjournal.com)How Nevada Medicaid struggles with mental health care fraud (reviewjournal.com)
Nevada Medicaid officials are putting a temporary stop on the enrollment of some mental health providers while new certification requirements are considered...the largely federally funded state agency will put a six-month pause on enrolling new qualified behavioral aides and qualified mental health associates...State mental health groups had brought up concerns that some enrolled as either provider type were performing services without proper qualification…Nevada Medicaid acting Administrator Cody Phinney also expressed concern over potential improper billing for codes including medication training and support and crisis intervention...