- CBER Launches new Program for Early Interaction With Biologics Developers (raps.org)
The US Food and Drug Administration's Center for Biologics Evaluation and Research...announced it is launching a new program for early meetings with biologics developers...The new program, called INTERACT (Initial Targeted Engagement for Regulatory Advice on CBER ProducTs), replaces CBER's existing pre-investigational new drug meeting program...We know that academic investigators, small and medium enterprises, and sometimes even large companies can have questions regarding the path from preclinical to clinical development for biological products—particularly those that raise new regulatory questions," said CBER Director Peter Marks...the new program will be better tailored to providing input to biologics developers at early stages of development and will help sponsors plan their development programs to gather evidence needed for FDA approval more effectively.
- Opioid reclassification linked to increase in illegal online sales (reuters.com)Effect of restricting the legal supply of prescription opioids on buying through online illicit marketplaces: interrupted time series analysis (bmj.com)Tighter prescribing regulations drive illicit opioid sales (bmj.com)
Online black-market sales of opioid painkillers more than doubled in the United States during the two years after these drugs were “rescheduled,” putting tighter restrictions on legal prescriptions and sales, according to a recent study...In October 2014, hydrocodone combination products...were reclassified from...schedule III to schedule II, imposing stricter controls on prescriptions written by doctors and on patients’ ability to refill them...Almost immediately, the proportion of all drugs illicitly purchased in the U.S. from sellers on the “dark net” that were in the opioid category began rising, reaching 13.7 percent in 2016. Stronger, more dangerous opioids also gained in popularity in these so-called cryptomarkets...cutting off supply leads to people sourcing their drugs from illegal and uncontrolled sources, and they buy products of higher strength - with all the dangers that this type of supply carries...A better approach would be harm reduction, therapeutic intervention, reliable treatment options, dealing with over-prescription early on, and providing information on the danger of opioid use to reduce stigmas...policy measures like up-scheduling, an introduction of abuse-deterrent formulations, prescribing limits, and prosecuting health care providers created a powerful push that precipitated the changes in the overdose dynamics we are contending with today...
- This Week in Managed Care: June 22, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- 340B audits target hospitals more than pharma, sparking lawmaker criticism (biopharmadive.com)
The 340B Drug Pricing Program took center stage at the Senate's HELP panel for the third time in several months...this time turning the focus to audits of hospitals and drug manufacturers conducted by the Health Resources and Services Administration...HRSA Director Krista Pedley said that, in the past five years, 12 of 600 program manufacturers faced audits compared to 981 of 12,700 of hospitals...No findings of wrongdoing were discovered in any of the 12 audits of manufacturers, she noted, whereas 60% of the audits of covered entities resulted in repayment to manufacturers...
- Refusal to Fill Prescriptions Under Conscience Clauses (drugtopics.com)
A more difficult question is raised if a pharmacist refuses to fill based upon his or her religious beliefs...a University of Wisconsin student requested a refill on her birth control prescription at her local pharmacy. The pharmacist on duty, Neil Noesen, RPh, refused to fill it, citing his religious beliefs. He also would not transfer the prescription or tell her where she could get it filled...The student filed a complaint with the Wisconsin Board of Pharmacy.The board’s administrative law judge restricted Noesen’s license, finding that a pharmacist with a conscientious objection to dispensing a prescription must ensure there is an alternative mechanism for the patient to receive the medication, including informing the patient of their options to obtain their prescription.”...The Court on appeal ruled, “Noesen had a right to refuse to provide birth control pills but not to refuse to transfer a valid prescription to another pharmacy.”...The Trump administration is making it easier for medical providers to object to procedures on religious grounds...an HHS proposed rule...would allow HHS to enforce protections for religious medical providers...Pharmacists must be familiar with their state regulations and new federal guidelines...
- Study: With FDA Input, Compassionate Use Programs Appear to Work Well (ptcommunity.com)Availability of Investigational Medicines Through the US Food and Drug Administration’s Expanded Access and Compassionate Use Programs (jamanetwork.com)
When terminally ill Americans receive experimental medicines through so-called “compassionate use” programs, they typically only get these drugs after extensive tests for safety and effectiveness, a recent study suggests...This means that sufficient evidence of safety and effectiveness has been generated, ensuring that terminally ill patients are not being exposed to therapies that are unlikely to be beneficial or unsafe...this...suggests that companies and the FDA are providing expanded access to experimental therapies in a responsible way that protects patients and the public health...This system may change under The Right to Try Act of 2017, which would let companies decide whether to give patients experimental therapies without any input from the FDA...The chances of a drug not helping a patient or causing serious harm are higher when medicines are earlier in development, Paul Beninger, MD, MBA, of Tufts University School of Medicine...This is much more likely with Right to Try than with the current compassionate use system…
- June 22 The Week in Pharmacy: ADA Hosting 78th Scientific Sessions (pharmacytimes.com)
Ned Milenkovich, PharmD, JD, PTNN. , Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Canada Intends to Further Restrict Promotion of Opioids (raps.org)Notice of Intent to Restrict the Marketing and Advertising of Opioids (canada.ca)
Health Canada this week said it intends to restrict the marketing and advertising of opioid drugs...Evidence suggests that the marketing and advertising of opioids has contributed to increased prescription sales and availability of opioids...From presentations sponsored by opioid manufacturers at conferences to advertisements published in medical journals, these practices “can take many forms of direct and indirect activities and incentives.”...Manufacturers of opioids with a presence in the Canadian market are “strictly prohibited” from advertising to the public, but not to healthcare professionals...Health Canada...set forth additional authority for the Minister of Health to require implemented risk management plans from opioid manufacturers. These plans must include the preclearance of all materials related to opioids and provided to healthcare professionals to safeguard the compliance of their marketing clearances.
- Reno VA nursing home receives low rating (kolotv.com)
The Reno VA Hospital's nursing facility is under the microscope..."We have work to do, we know that and we are ready and willing to do it," said Glenna Smith, Hospital’s Public Affairs Officer...the nursing home received a one-star rating for quality between the months of July and September 2017. But overall, it received two stars...Smith explains the low rating can be attributed to a big change. That change took place a few years ago to better position with the Center for Medicare and Medicaid Services rating system, which is used for private sector nursing homes...But Smith says the VA hospital can't pick and choose who to care for, like its counterparts in the private sector. So that leads to admitting patients who are severely ill, which impacts the ratings...The most recent rating between January and March 2018 saw an increase. The overall rating went to three stars with quality going to two...Smith says paying attention to how to treat pain and also increasing veteran patients' mobility are two biggest factors to improving the ratings...
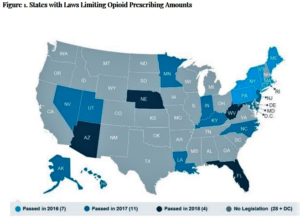
- Opioid supply levels decline in most states, report finds (biopharmadive.com)Opioid Supply Levels Decline in Most States (avalere.com)
A new report on opioids from DC-based healthcare consulting firm Avalere found supply levels were 11% lower in 2017 compared with 2016...The decline is seen in most states, and the greatest reductions are where laws have been passed to set limits on opioid prescriptions, with Maine seeing a fall of 24.8% after legislation was passed in 2016...The only state to see an increase was Idaho, with a 59.7% growth in opioid prescriptions. This included an almost three-fold rise between 2016 and 2017 in the amount of oxycodone sold...Avalere's analysis is based on opioid analgesics data from the Drug Enforcement Agency's Automation of Reports and Consolidated Orders System, which tracks distribution of most controlled substances from manufacture through distribution and point-of-sale...