- EMA and the Netherlands Finalize Seat Agreement (biopharminternational.com)
...the European Medicines Agency announced that the text for the Seat Agreement between the agency and the Netherlands has been finalized. The agreement describes how EMA will be treated by the Dutch government when EMA relocates to Amsterdam. The agency also announced that the Dutch Council of Ministers had agreed to sign the agreement...The agreement will be signed after the legislative process on the relocation is completed by the European Council, the European Parliament, and the European Commission...According to EMA, the Seat Agreement allows EMA to function independently in the Netherlands. Similar agreements apply to other EU agencies located in the Netherlands...As this transition needs to be supported by EMA staff in the Netherlands, a timely signing of the agreement would make sure that EMA staff and their families have clarity on their treatment and can settle in the Netherlands...
- In a blow for pharma, Supreme Court upholds the hated IPR patent challenge (fiercepharma.com)
Branded drugmakers have said "no fair" to the U.S. Patent and Trademark Office's inter partes review system almost since its inception seven years ago. One reason? "It's unconstitutional," the companies contend...The Supreme Court...disagreed...In a 7-2 vote, the U.S. High Court held that the system is constitutional. It doesn't violate Article III of the Constitution, which gives the judicial branch alone the power to decide legal arguments. Nor does it violate the Seventh Amendment, the right to a trial by jury...It's a limited ruling specific to the issues in this particular case, Oil States Energy Services v. Greene’s Energy Group...Justices John Roberts and Neil Gorsuch dissented, saying only an independent judge—not a political appointee at the PTO—should be able to revoke patents, which they equated with personal property such as a home or a farm...PhRMA...said the "narrowly tailored decision" found only that IPRs are constitutional, not "efficient or fair." The arguments and a...ruling in another case—SAS Institute v. Iancu—mean it's "clear there are problems with the IPR process that need to be addressed,"...
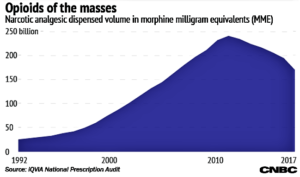
- How doctors in the US prescribe opioids — four charts (cnbc.com)Institute Reports Medicine Use and Spending in the US Review of 2017 Outlook to 2022 Medicine Use and Spending in the U.S. A Review of 2017 and Outlook to 2022 (iqvia.com)
The number of opioid pills prescribed peaked in 2011 and has since declined by 29 percent, according to a new report from the Iqvia Institute...The number of prescriptions accelerated its decline in 2017, with an even faster drop for high doses...More patients appear to be starting on medication-assisted therapies to treat opioid addiction...decline accelerated...helped by changes in regulation of opioid prescribing and in reimbursement policies from insurers...many of the programs that have been put into place in the past year or two seem to be having an impact...increased use of nonopioid pain treatments, like ibuprofen and other nonsteroidal anti-inflammatory drugs...as well as public awareness about overuse and misuse of opioids...
- FDA commissioner to drug middlemen: You’re part of the problem (cnbc.com)
Drugmakers may not be the only ones keeping less-expensive drugs off the market...The Food and Drug Administration has approved nine biosimilars, generic versions of biologic medicines, but only three are available...Manufacturers are using several schemes to "hamstring biosimilar competition," FDA Commissioner Scott Gottlieb said... he worries pharmacy benefit managers have been "complacent participants" in the schemes...PBMs and insurers may stick with branded biologics because they receive discounts from manufacturers on these treatments. That can leave consumers paying for costly treatments when less-expensive ones are available while PBMs make more money on these discounts, known as rebates...Many of these practices persist because high list prices enable lucrative returns across the drug supply chain as the spread between list and net price is carved up and shared among participants...he (Gottlieb) applauded insurers who have recently pledged to pass manufacturer's drug rebates directly on to some of their members...This is a bold action that will help create a fairer, more transparent market...I hope that other insurers, employers, and manufacturers follow their lead. I also hope that your industry will continue to innovate to make it more transparent to pass along these rebates...
- This Week in Managed Care: April 27, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Pharmacy Week in Review: April 27, 2018 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- This Week in Managed Care: April 20, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- CDER Publishes Drug Safety Report – FDA’s Center for Drug Evaluation and Research has published its second annual report on key safety programs and activities. (biopharminternational.com)
...FDA’s Center for Drug Evaluation and Research released its second annual Drug Safety Priorities report, which details drug safety initiatives carried out by CDER and FDA. The report highlights drug safety program milestones and gives an update on goals achieved in 2017. Efforts by FDA to ensure drug safety science, surveillance, and oversight are discussed...Detailed in the report are the agency’s efforts on pharmacovigilance, medication errors, and risk management...The report also goes into detail about the agency’s views on how real-world evidence can advance drug safety. An update on the agency’s efforts to combat the opioid crises is also provided...
- I-Team: Audit of UNR’s School of Medicine hidden from public (lasvegasnow.com)State's response to the I-Team requesting the audit report (media.lasvegasnow.com)Statement from University of Nevada, Reno School of Medicine Dean Thomas L. Schwenk, M.D. in response to KLAS-TV report (med.unr.edu)
An explosive but confidential audit which investigated reports of widespread billing errors within the University of Nevada medical school has been kept quiet for more than two years...It's only been known to a handful of officials in the higher education system. But the confidentiality is about to end...The audit uncovered incompetence on a massive scale, and quite possibly, fraudulent billing that continued for years within the University of Nevada's medical school practice...The I-Team has been asking for a copy of the report for weeks. Two days ago, the state said no because it is private and confidential...The audit by an independent firm, "Crowe Horwath," uncovered systemic problems within the University of Nevada, Reno Medical School's billing procedures: overbilling, underbilling, lack of oversight, and potential legal consequences...
- Pharmacy Week in Review: April 20, 2018 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.