- Apple Likes the Patent ‘Death Squad.’ Allergan Pays to Avoid It (bloomberg.com)
Allergan Plc’s decision to pay a Native American tribe $15 million a year rather than let one of its blockbuster drugs be scrutinized by the U.S. Patent & Trademark Office is part of a backlash against an agency review panel that has been dubbed a “death squad.”...The drugmaker earlier this month transferred ownership of patents protecting a medicine with $1.49 billion in sales last year to the Saint Regis Mohawk Tribe of upstate New York. The tribe, which will receive royalties every year, says that as a sovereign entity it is immune from such civil patent challenges....The creative -- and untested -- maneuver is designed to circumvent the Patent Trial and Appeal Board...critics say the board has made it too easy for rivals to attack patents and they’re pressing Congress, the courts and the patent office for changes...companies such as Google or Apple Inc., which are among the biggest users of the review board to fend off what they consider nuisance lawsuits from companies looking for a quick payday...the Supreme Court agreed to take a case to determine if the reviews are constitutional -- critics of the reviews say a patent is a property right that only federal courts can revoke. But even those who want to see the system dismantled say that case is a long shot...The patent office has been considering changes to its procedures...
- Many drug companies fail to conduct timely safety checks on medicines after FDA approval (reuters.com)
In the rush to approve new medicines, the U.S. Food and Drug Administration often requires drug companies to study possible side effects and alternative doses for medicines once they hit the broader market...A new analysis in the New England Journal of Medicine concludes that, in many cases, that’s not being done...among the 614 studies mandated in 2009 and 2010, 20 percent were never started and 9 percent have been delayed...When drugs are approved, the trials are usually small and short-term, and some side effects may not emerge until the post-marketing phase...The problem is, the faster you get them on the market, the more open questions there are about their safety or the best dose...In some cases, the FDA has simply dropped a requirement for a postapproval study without giving a reason...
- FDA calls for industry input on continuous manufacturing guidelines (biopharmadive.com)
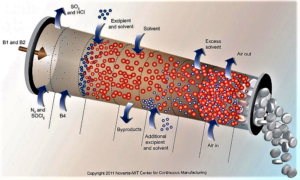
The Food and Drug Administration plans to develop clearer guidelines around the adoption of continuous manufacturing and has asked industry stakeholders for input on how best to design a regulatory framework...The regulator has advocated companies make the switch from traditional batch production to continuous manufacturing, which to a large extent hasn't been widely adopted by drugmakers despite the potential benefits to speed and reliability...continuous manufacturing can reduce the risk of manufacturing failures and potentially help prevent drug shortages from developing — areas that have been in focus as the agency steps up its oversight of pharmaceutical production facilities...Pharma has been loath to change over from batch production, which has served the industry for decades...Continuous manufacturing techniques — already adopted in many other industries — promise to shorten production times significantly, while reducing the amount of human intervention needed throughout...The FDA hopes providing a "framework of principles" will help drugmakers navigate CM adoption and implementation...
- Week in Review: September 15, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- This Week in Managed Care: September 22, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Injunction denied: Pharma argument fails to freeze Nevada drug pricing law (fiercepharma.com)
Drug pricing regulation hasn't gained much ground on a national level, but in Nevada, there's a fierce fight over pricing and transparency for diabetes medications. In the latest twist, the industry lost its bid to stall a new law that's designed to shine light on pricing practices...Industry groups PhRMA and BIO filed for a temporary restraining order and preliminary injunction against officials in Nevada, contending that the state's Senate Bill 539 will "impose irreparable injury" against drugmakers...The bill mandates that the state gather a list of essential diabetes medicines and as well as detailed pricing information from companies that sell those drugs. The reports would include costs, profits, rebates and other details, in addition to pricing numbers...And then that information would go public: Under the law, the state would publish the reported info online.
- Drug industry on tenterhooks as Maryland price-gouging law nears (reuters.com)
...state authorities and patient advocates in Maryland are preparing to enforce the nation’s first law designed to punish drugmaker price-gouging...The state Attorney General’s office said it will field complaints and investigate “unconscionable increases” in essential generic medicines when the closely watched law takes effect Oct. 1...Drugmakers fear the Maryland law will embolden other states and are seeking a court injunction. Both sides made their arguments...before a U.S. District Court judge in Baltimore, who could decide on an injunction in the coming days...But states, struggling to cover rising healthcare costs, are taking up the fight. At least 176 bills on pharmaceutical pricing and payment have been introduced this year in 36 states, according to the National Conference of State Legislatures...Maryland’s law is the most aggressive legislation to be passed so far, and allows the state to levy fines and order a reversal of price increases...
- Pharmacy Week in Review: September 22, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Exclusive: FDA plans new compounding pharmacy policy, agency head says (reuters.com)
The head of the U.S. Food and Drug Administration said...the agency is working on a new policy that would encourage more compounding pharmacies to register under...the Drug Quality and Security Act, which aimed to bring more compounding pharmacies...under the authority of the FDA rather than state pharmacy boards...The law created a category of “outsourcing facilities” that could register with the FDA, allowing them to sell products in bulk to hospitals and physician practices without prescriptions for individual patients...In exchange, those compounders would have to follow federal manufacturing standards and subject themselves to routine inspections...around 70 firms have registered as outsourcing facilities...compounders that did not register with the FDA would remain under state oversight, and...could only compound drugs based on prescriptions for specific patients...Gottlieb said that in order to encourage more compounders to register, the FDA would release draft guidance in the next two months reflecting its intention to adjust its enforcement priorities based on the size of registered compounders and the riskiness of their products...We’re looking at ways we can provide more of a gradation in our regulatory architecture so we don’t have a one-size-fits-all approach...
- This Week in Managed Care: September 15, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network