- Pharmacy Week in Review: May 13, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Judge unseals records from Kentucky’s OxyContin lawsuit (cnbc.com)
A judge has unsealed records from a Kentucky lawsuit against the maker of the prescription painkiller OxyContin, including the secret testimony of a former company president...Pike Circuit Court Judge Steven Combs ordered the records be released in 32 days. But Combs said he would delay his order if Purdue Pharma appealed the decision...OxyContin is a powerfully addictive prescription painkiller that was marketed for its ability to slowly release its effects over a 12-hour period. The company suggested this long-acting formula made it less addictive and safer for patients. But users quickly found the pill lost its time-release qualities if it was crushed, resulting in an instant high...In 2006, the company agreed to plead guilty and pay more than $630 million to settle federal charges that it misled doctors and patients about the risks of its top-selling drug...The judge in the case agreed to seal any records Purdue Pharma marked as confidential. Statnews.com...sued to have the records released once the case was resolved. They are particularly interested in Sackler's testimony, as it could reveal how much the company knew about how addictive OxyContin truly is...Lawyers for Purdue Pharma said the company agreed to hand over documents and give Kentucky's attorneys access to high level company officials because it believed the documents would never be made public. They argued that releasing the documents now would betray that trust and bog down the state's civil court docket.
- Oncologists Want Medicare Drug Demo Proposal Gone (bna.com)
Oncologists and other doctors are urging the Medicare agency to rescind a proposed demonstration that would test six different approaches for paying for Part B drugs that are administered in physician offices and outpatient departments...The American Society of Clinical Oncology said...that the CMS should "withdraw an experiment of this magnitude without first understanding its potential impact on patient care."...The proposed regulation, published in the March 11 Federal Register, "is particularly ill-suited to the delivery and treatment of cancer care, which is complex and highly personalized to each patient," ASCO President Julie M. Vose said in a statement...The Federation of American Hospitals said the CMS needs to go back to the drawing board to develop policy proposals that more directly and effectively address the problem of unsustainable increases in drug costs.
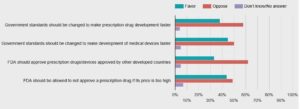
- Public wary of faster approvals of new drugs, STAT-Harvard poll finds (statnews.com)
A majority of Americans opposes federal regulatory changes to speed up the development and approval of new medical treatments, a new STAT-Harvard poll finds — suggesting the public has serious doubts about legislation now moving through Congress...both parties are pushing to change government regulatory standards that they blame for slowing the approval process to get new products to patients...nearly 6 out of 10 Americans said they oppose changing government safety and effectiveness standards to allow for faster approvals of new prescription drugs by the Food and Drug Administration, while 38 percent said they’re in favor of speedier FDA action...The poll sheds new light on how Americans balance their priorities between speed and safety in the approval of new medical treatments.
- FDA Advises Against Fluoroquinolone Antibiotic Use for Uncomplicated Infections (pharmacytimes.com)
Serious side effects associated with fluoroquinolones generally outweigh the antibiotics’ benefits for patients with uncomplicated infections like sinusitis and bronchitis…the FDA determined that systemic use of fluoroquinolones is associated with "disabling and potentially permanent serious side effects that can occur together" involving the tendons, muscles, joints, nerves, and central nervous system...The alert comes a year after the FDA convened an advisory committee to review the risk-benefit balance for fluoroquinolones...the risk wasn’t worth the potential benefit for patients with sinusitis, bronchitis, and uncomplicated urinary tract infections, for which other treatment options exist...Labels and medication guides for all fluoroquinolones will be updated to reflect the FDA’s findings...
- FDA wants drug makers to pay fees to review OTC medicines (statnews.com)
The US Food and Drug Administration is paid fees by the pharmaceutical industry to review brand-name and generic drugs. Now, the agency wants to collect money to review over-the-counter medicines...Specifically, the agency wants companies to pay for reviewing OTC monographs. Unlike brand-name and generic drugs, which involve individual companies seeking approval to market individual drugs, the OTC monographs refer to multiple medicines that share the same ingredient for the same use. Moreover, numerous companies can make these drugs, but do not require FDA approval prior to marketing...the pharmaceutical industry has largely supported so-called user fees...the fees helped transform the relationship between companies and regulators, who are under increasing pressure to meet benchmarks for speeding applications through the review process...Given the workload, the FDA argued that user fees will only bolster its ability to review OTC medicines…the large OTC companies interested in a modernized system who will support it in exchange for some concessions from FDA...The smaller, more fragmented (companies) will probably oppose it as (fees represent) more regulatory burden on them in an area that has worked for decades without fees
- India awards Gilead patent for Sovaldi, disappointing civic groups (fiercepharma.com)
Gilead Sciences has received Indian Patent Office approval for Sovaldi (sofosbuvir), with the U.S. drugmaker saying that the nod for a patent on the blockbuster hepatitis C medicine does not hinder access--even as civic groups such as Médecins Sans Frontières decried the ruling...Gilead has worked with several Indian drug manufacturers to produce licensed versions of the drug for defined emerging markets at a significantly lower cost for the therapy (than) in major reimbursed developed markets in the U.S., Europe and Japan...But the access program has not ended complaints from MSF, and other groups, that the particular decision sends a poor signal on access to a wide variety of generics made by Indian firms--some of which were approved under compulsory licenses in the country. At the same time, Indian officials have denied reports that patent policies are changing in favor of stronger intellectual property rules sought by multinational drug firms...according the Hindu, the drug may not be out of the woods yet in India, with a separate application before the Kolkata patent office with civic groups geared up to offer opposing views.
- The Impact of Precision Medicine and the 21st Century Cures Act on Modern Pharma (specialtypharmacytimes.com)
The 21st Century Cures Act will have a huge impact on the pharmaceutical industry nationally and globally...All of the major pharmaceutical companies target the United States...they will have to comply with the...Act (when passed)…Precision medicine, meanwhile, is a programme announced...last January...it is an emerging approach for disease treatment and prevention, which includes a patient’s variability in their genes, environment, and a person’s lifestyle...What has the precision medicine initiative got to do with the 21st Century Cures Act?.. Precision medicine would allow the inclusion of much more technology driven healthcare innovation and would permit the introduction of additional personal data and information into health science...The requirement to get drugs to market faster is evident. However, a lack of interoperability poses a huge challenge when it comes to getting patient data consolidated in time for clinical trials...A large part of the new legislation is aimed at trying to connect the research and development arms of life science companies to health IT or digital health specialists...all pharmaceutical companies will benefit from the Act as they will be able to get their drugs out faster. On the technology side, the major beneficiaries of the Act will be the big data analytics players, since this lies at the heart of speeding up approvals...The good news is most (if not all) of the major players are fully aware of the challenges and opportunities of the Act.
- Drug Price Control: How Some Government Programs Do It (commonwealthfund.org)
The federal government already has several tools for reducing the prices of drugs it buys...The VA and DOD pay drug prices that are roughly half as much as those paid by retail pharmacies...Drug pricing is having its moment. Proposed treatments for the drug pricing epidemic are varied, but some call for government to do more...While others resist the idea of government involvement, the federal government, in fact, already employs a number of effective tools for reducing the prices of drug purchased by certain public programs. These fall into two broad categories:
- Price controls, usually in the form of required discounts off of the average price paid by other purchasers.
- Negotiated pricing, in which the government wields its market power to bargain for favorable rates from pharmaceutical suppliers.
These strategies do not exhaust the available approaches, nor are they necessarily the most desirable...The Programs:
- Medicaid
- Veterans Health Administration and the Department of Defense (DOD)
- 340B Program
The bottom line is that the government can and does get better deals on drugs than private purchasers. The effects of these policies on quality and innovation need to be better understood—but were the government to expand them, it would be more of an evolution than a revolution.
- Ex-FDA head and Sanofi call for harmonized drug regulation (reuters.com)The need for global regulatory harmonization: A public health imperative (stm.sciencemag.org)
Drug regulation has failed to keep up with a globalized world and governments should harmonize oversight to improve patients' access to new and innovative medicines...That is the view of the former leader of the U.S. Food and Drug Administration, the world's top drug regulator, and the research head of French drugmaker Sanofi, who made a joint plea to governments for action...Margaret Hamburg, who led the FDA until 2015, and Elias Zerhouni said there was an urgent need to harmonize a "mosaic of regulations" in different countries, and they called for the issue to be taken up at the G8 or G20 groups of nations..."Essentially, it is a hidden bureaucratic inefficiency tax on the whole effort of finding new and valuable therapies," he said in a telephone interview...Drug development is global and we need to have safety and efficacy data globally, so we should have a global system, just like with airplanes...Sanofi was spending around 20 percent of its research and development budget on ensuring convergence between different systems, often involving duplication of efforts.