- Trump Says Elderly to Get $200 for Drugs in Bid for Senior Vote (msn.com)Trump Health Policies Again Thrust Drug Pricing and Access into the Spotlight (biopharminternational.com)
President Donald Trump said that Americans in the Medicare program for the elderly and disabled will be sent $200 discount cards for prescription drugs within weeks, potentially putting cash in their pockets ahead of his November re-election...Assuming they are sent to 33 million Medicare beneficiaries, the figure Trump used, the cards would cost about $6.6 billion...Money for the cards will be drawn from a demonstration program Medicare uses to test new payment systems and other projects, and the cost will be offset by future savings generated from new price cuts Trump has ordered for drugs bought by Medicare, according to a White House official. The cards can be used for prescription drugs co-pays, the official said, but didn’t elaborate...READ MORE
- Tech startups developing rapid COVID-19 tests to be taken completely at home (fiercehealthcare.com)
Computer vision company Gauss teamed up with biotechnology company Cellex to develop a rapid, at-home and point-of-care COVID-19 antigen test...While there are other at-home COVID-19 diagnostic tests available, at least two companies are working on rapid antigen tests that can be performed by people at home without involving a laboratory...The Gauss and Cellex test has not yet gained Food and Drug Administration approval. The companies are pushing for FDA Emergency Use Authorization some time this fall...READ MORE
- Hospitals cheer demise of Medicaid Fiscal Accountability Rule (fiercehealthcare.com)
Hospital groups praised the Trump administration's decision Monday to pull a Medicaid rule aimed at fiscal accountability, which providers warned could lead to massive cuts in reimbursement..."up to $50 billion in annual funding for the Medicaid program was on the line" if the rule had been finalized..."We appreciate CMS for acknowledging the harmful consequences this rule would have for patients,"..."Hospitals and health systems will be greatly relieved when the proposed rule is formally withdrawn."...The Centers for Medicare & Medicaid Services first unveiled the Medicaid Fiscal Accountability Rule in November, with the goal of tamping down on schemes used by states to boost federal matching funds in the program...READ MORE
- CMS: ACOs save Medicare $1.2B under ‘Pathways to Success’ program (fiercehealthcare.com)
A federal program aimed at lowering Medicare costs by incentivizing providers to move from fee-for-service to value-based care models generated $1.2 billion in net savings to Medicare last year, the Centers for Medicare & Medicaid Services announced...CMS Administrator Seema Verma credited savings seen under the Medicare Shared Savings Program to recent rules that shortened the pathway for financial risk...These results are additional evidence that physician-led practices can be successful in generating savings through value-based care..."Once again, physician-led ACOs out-performed hospital ACOs,"..."What we need now is to help more practices participate in these models of care."...READ MORE
- Trump says may block stricter FDA guidelines for COVID-19 vaccine (reuters.com)
U.S. President Donald Trump said...he may or may not approve any new, more stringent FDA standards for an emergency authorization of a COVID-19 vaccine, saying such a proposal would appear political...The Washington Post reported on Tuesday the U.S. Food and Drug Administration would issue the guidance to boost transparency and public trust as health experts have become increasingly concerned the Trump administration might be interfering in the approval process to rush out a vaccine...READ MORE
- CDC Head and Trump Spar Over COVID-19 Vaccine Timeline (biospace.com)
Robert Redfield, director of the U.S. Centers for Disease Control and Prevention, testified...before the U.S. Senate...He noted that although a vaccine against COVID-19 will likely be available and to begin dosing in November or December of this year, it will be limited. Getting the entire U.S. population vaccinated will likely take “six to nine months.”...He also said that face masks are “the most important, powerful public health tool we have.” He added, “I might even go so far as to say that this face mask is more guaranteed to protect me against COVID than when I take a COVID vaccine.”...READ MORE
- Biosimilars May Lead to Improved Competition and Lower Costs (drugtopics.com)
Biosimilars, which offer the potential for more treatment options and less expensive alternatives, are having a good year in 2020 and may have a significant impact on the prescription product market in 2021. With the approval of more biosimilar medications, experts hope that greater competition may occur and help lower overall costs. “As intellectual property protections for biological products continue to expire in the US, we can expect many more applications for potential biosimilar and interchangeable products, and increased uptake of approved products too,”...Some of the key issues affecting biosimilar uptake involve reimbursement factors and insurance coverage...However...business and intellectual property concerns also have contributed to the delayed launch of some FDA-approved biosimilars. ...READ MORE
- BREAKING: FDA will make it HARDER to get a coronavirus vaccine approved in a move that could block Trump’s plan to have a shot before Election Day (dailymail.co.uk)U.S. FDA to tighten coronavirus vaccine authorization standards ahead of election - paper (reuters.com)
The US Food and Drug Administration... is expected to issue new, tougher requirements for its approval of a coronavirus vaccine, a move that could obliterate the chances of a shot getting emergency use authorization before Election Day...Regulators could publish the new approval standards as early as this week, and will do so publicly in an effort to bolster Americans' eroded trust in the US to ensure the safety a COVID-19 vaccine...President Trump and his Operation Warp Speed initiative have been pushing for months to have a coronavirus vaccine approved ahead of the November 3...READ MORE
- A vaccine alone won’t stop Covid-19. We also need a trusted plan for it (statnews.com)
Safe and effective vaccines represent the most effective way to restore the health and economic security disrupted by the Covid-19 pandemic. To help achieve that goal, the U.S. government launched Operation Warp Speed...to accelerate development and manufacturing of several Covid-19 vaccines, with a goal of having 300 million doses available to the U.S. population by January 2021...Operation Warp Speed is expediting vaccine development primarily by moving clinical trials forward without pauses between phases, and by scaling up manufacturing capacity before knowing if a candidate works...Covid-19 vaccines can help stop the pandemic only if people trust them and want to be vaccinated. To earn and keep the trust of the American people, our government needs to ensure three key needs are met before launching any immunization campaign...READ MORE
- Ensure transparency and confidence in FDA decisions
- Ensure robust active safety monitoring as Covid-19 vaccines are rolled out
- Ensure the distribution and administration of Covid-19 vaccines are equitable and well-executed
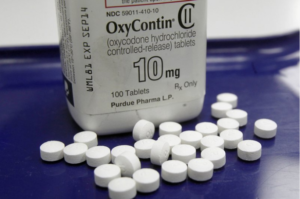
- Experts: Revamped OxyContin hasn’t curbed abuse, overdoses (apnews.com)
A panel of government health advisers said...there’s no clear evidence that a harder-to-crush version of the painkiller OxyContin designed to discourage abuse actually resulted in fewer overdoses or deaths...The conclusion from the Food and Drug Administration advisory panel comes more than a decade after Purdue Pharma revamped its blockbuster opioid, which has long been blamed for sparking a surge in painkiller abuse beginning in the 1990s...The long-acting OxyContin tablets can still be misused by simply swallowing them, which remains the most frequent route among those with opioid addiction...READ MORE