- March 6 Week in Review (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
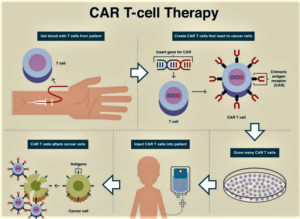
- CAR-T—the Future of Medical Progress Is Now (realclearhealth.com)
Personalized medicine is the future of medical progress...For instance, an immunotherapy treatment called chimeric antigen receptor T-cell therapy is accessible now, and the Trump administration has the opportunity to make it widely available to Medicare beneficiaries...over 70 Members of Congress—Democrats, Republicans, liberals, and conservatives—sent a letter to Seema Verma, administrator of the Centers for Medicare and Medicaid Services, commending the administration for “ensuring Medicare patients nationwide have access” to this life-saving treatment. The Congressional letter goes on to ask the administration to “ensure that hospitals are appropriately reimbursed so they may continue to provide” CAR-T therapy to America’s seniors...without appropriate reimbursement policy, a Medicare patient could be denied access to a treatment that would save his or her life. Without proper reimbursement by Medicare, providers simply will not be able to offer it as an option, especially in rural areas as patients must stay near a treatment center for four weeks to be monitored...READ MORE
- Sanofi, Justice Department ink $11.85M charity kickbacks settlement (fiercepharma.com)
A federal probe of pharma’s charity contributions has secured settlements from drugmakers large and small, and Sanofi is the latest to ink a deal with the government over allegations its payments were kickbacks...Sanofi agreed to pay $11.85 million to resolve allegations that it used The Assistance Fund as a “conduit” to help multiple sclerosis patients pay their Medicare co-pays for Lemtrada...The drug costs nearly $100,000 per year, and patient co-pays can run thousands of dollars per year...“Sanofi sought to undermine the Medicare program through its use of kickbacks disguised as routine charitable donations aimed at helping patients battling multiple sclerosis and who were struggling with costly copays,”...READ MORE
- Bristol-Myers Squibb reaches tentative deal to end long-running Medicaid rebate lawsuit (fiercepharma.com)
Lawsuits over Medicaid rebates are a fact of life in pharma, and in one particularly long-running case, Bristol-Myers Squibb stands accused of fiddling with its prices to lower payments. Now, after seven years in litigation, that lawsuit could be nearing an end...Bristol reached an "agreement in principle" in January to settle a 2013 whistleblower lawsuit accusing the company of manipulating the average manufacture price of its drugs in order to underpay Medicaid rebates between 2007 and 2016, according to an SEC filing...READ MORE
- First patient in Nevada tests presumptively positive for novel coronavirus, is in ‘airborne isolation’ (thenevadaindependent.com)
A Clark County man who is in his 50s and has an underlying health condition is the first patient in Nevada who has tested presumptively positive for the novel coronavirus...The determination made Wednesday evening stems from a test the health authority performed on the man, who is currently hospitalized and in “airborne isolation,” and a sample is being sent to the Centers for Disease Control and Prevention for official confirmation...VA spokesman Charles Ramey said in an email that the man who tested presumptively positive for the disease is a veteran inpatient at the VA Southern Nevada Healthcare System...Health authorities said the patient reported traveling recently to Washington State, where the virus has been widely spreading...READ MORE
- Novartis shells out $195M to settle criminal charges in generics price-fixing probe (fiercepharma.com)
When federal prosecutors nabbed a guilty plea from a former exec at Novartis' Sandoz in an expansive price-fixing probe, it seemed to spell trouble for the massive generics player. Now, the other shoe has dropped for Novartis—to the tune of $195 million and an extraordinary confession—and more could be on the way...Novartis agreed Tuesday to pay $195 million and enter deferred prosecution to settle federal claims it colluded in an industrywide price-fixing scheme between 2013 and 2015...The criminal settlement is the single largest domestic antitrust deal ever signed....and it comes two weeks after a former Sandoz unit exec pleaded guilty to federal conspiracy charges in the scheme...READ MORE
- Cardinal Health pays SEC $8.8 million to settle China FCPA offenses (fcpablog.com)
Cardinal Health paid the SEC $8.8 million Friday to settle FCPA (Foreign Corrupt Practices Act) offenses related to a Chinese subsidiary that provided marketing services...In an internal administrative order, the SEC charged Cardinal Health with violating the FCPA’s books and records and internal accounting controls provisions...Dublin, Ohio-based Cardinal Health agreed to disgorged $5.4 million to the SEC, plus prejudgment interest of $916,887, and pay a civil penalty of $2.5 million...In one instance, a marketing account it administered for a UK- based pharmaceutical manufacturer was terminated in 2013 after Cardinal’s CEO received an internal report alleging that Cardinal China employees were using the account to “bribe employees of China’s Center for Disease Control.”...READ MORE
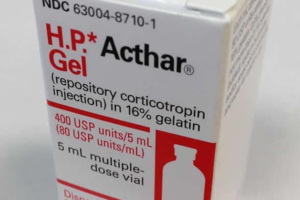
- Feds target Mallinckrodt after joining Medicaid rebate suit worth ‘hundreds of millions’ (fiercepharma.com)
Amid efforts to close a $1.6 billion opioid settlement, Mallinckrodt has fought tooth and nail to escape a massive Medicaid rebate bill for its controversial H.P. Acthar Gel. Instead of ceding ground, the U.S. government opted to take the fight right back to Mallinckrodt––and it could create even more trouble for the embattled drugmaker...The federal government has joined a False Claims Act whistleblower suit filed in Boston accusing Mallinckrodt of underpaying Medicaid rebates for Acthar by "hundreds of millions of dollars,"...READ MORE
- Coronavirus spurs India to restrict exports of 2 dozen drugs (fiercepharma.com)India's restrictions on API exports only temporary, official says: report (fiercepharma.com)
While eyes have been on China for signs that COVID-19 might result in drug shortages, India has come up with a surprise of its own. The country, which accounts for about 40% of U.S. generic drugs, has halted exports of more than two dozen APIs and drugs...India’s Directorate General of Foreign Trade today announced it was restricting 26 APIs and formulations until further notice...The government gave no further explanation, but Dinesh Dua, chairman of the Pharmaceuticals Export Promotion Council of India, told Reuters, “Irrespective of the ban, some of these molecules may face shortages for the next couple of months.” If interruptions from the virus get worse, he said, some shortages may become “acute.”...READ MORE
- Sisolak: Don’t panic, but practice basic hygiene to prepare for coronavirus (thenevadaindependent.com)No coronavirus cases confirmed in Nevada, but state officials prepare for possibility (thenevadaindependent.com)
Gov. Steve Sisolak...delivered a message of unity and calm Friday afternoon as Nevada prepares for possible coronavirus cases...There have been no confirmed cases of coronavirus...in Nevada, Sisolak said, before urging residents to continue practicing basic hygiene methods, such as washing hands with soap and water, coughing into your elbow and staying home from work or school if sick...“We’re going to prepare, not panic,” he said at a press conference in Las Vegas. “We’re going to choose collaboration over chaos.”...READ MORE