- Generic Drug Pricing Trends (drugtopics.com)
According to the Association for Accessible Medicines 2019 report with data from IQVIA, generic drug savings totaled $293 billion in 2018, which resulted in a 10-year savings of almost $2 trillion. Other key findings include that 4 billion generic prescriptions were filled across the U.S. in 2018, which is 90% of all dispensed prescriptions. Additionally, 95% of generic prescriptions were filled at $20 or less with an average copay of $5.63 whereas the average brand copay is $40.65. In 2017, patients who were prescribed more expensive brand products were 2-3 times more likely to leave the pharmacy without their prescriptions...There were 68,353 pharmacy stores that provided cash prices for the study; results revealed that independent pharmacies and small chains had the highest cash prices for generic drugs, while big box pharmacies had the lowest prices compared with large chains...READ MORE
- Mexico’s drug gangs churning out deadly fentanyl-laced pills: DEA (reuters.com)
Mexican drug cartels are making “mass quantities” of fake prescription pills containing the synthetic opioid fentanyl with the intention of selling them to users throughout North America...Mexico’s cartels have for years diversified into a wide variety of illicit activity, helped by porous domestic law enforcement agencies as well as long-standing trafficking routes into the United States...READ MORE
- Industry Voices—Industry reaction to shortages of lifesaving drugs underscores urgency (fiercehealthcare.com)Identifying the Root Causes of Drug Shortages and Finding Enduring Solutions (healthpolicy.duke.edu)U.S. Blames Drug Shortages on Low Prices and a ‘Broken Marketplace’ (nytimes.com)
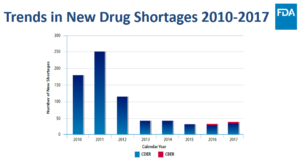
Drug shortages are a major challenge in healthcare. According to the U.S. Food and Drug Administration, the number of drug shortages is rising, with most shortage events lasting longer and resulting in a more adverse impact on public health, including added costs, delayed treatments or even death...Ultimately, drug shortages create uncertainty for a range of stakeholders: patients, hospitals, manufacturers, pharmacists and others. Especially in cases of shortages for lifesaving drugs, doctors must implement conservative efforts to ensure the patients that need it the most get their medication...Today, more than 100 lifesaving drugs are in shortage, and 70% of all hospital pharmacists report at least 50 shortages a year...READ MORE
- FDA task force offers suggestions to reduce drug shortages (fiercepharma.com)Report | Drug Shortages: Root Causes and Potential Solutions (fda.gov)
The FDA Tuesday dropped its report Drug Shortages: Root Causes and Potential Solutions with the explanation that includes suggestions that might help relieve chronic shortages but acknowledging that there has never really been any “private- or public-sector effort to collect and analyze comprehensive information to characterize shortages, quantify their effects, or closely observe the contracting practices that may be driving them.”...While there are many reasons for drug shortages, it had three recommendations to get at the root causes...READ MORE
- GOP, Dems agree on international reference price policy, but the devil is in the details (mmm-online.com)
There is one drug-price policy that Republicans and Democrats can agree on: international reference pricing...Leaders from both parties have proposed the model, but with some differences...It’s an idea that works around the world. Nearly every member of the European Union uses some kind of international reference pricing model. Countries such as Australia, Brazil, Canada, Japan, New Zealand and South Africa also employ it to set prices for their government healthcare systems...a drug’s price would be based on the average cost in several countries. That price would then become the benchmark for U.S. prices...READ MORE
- CMS retains 340B, site-neutral payment cuts in final hospital payment rule (fiercehealthcare.com)CMS finalizes payment rules to spur greater use of innovative kidney drugs, home infusion (fiercehealthcare.com)
The Trump administration finalized a hospital payment rule...that retains proposed cuts to off-campus clinics and the 340B drug discount program...The changes outlined in the hospital Outpatient Prospective Payment System rule come despite both cuts being struck down in legal challenges and amid major pushback from providers...The goal is to bring payments to off-campus clinics in line with standalone physicians' offices...“With the completion of the two-year phase-in, the cost sharing will be reduced to $9, saving beneficiaries an average of $14 each time they visit an off-campus department for a clinic visit in [calendar year] 2020,” the Centers for Medicare & Medicaid Services said...CMS also finalized a proposed cut for the 340B program that cuts payments by 22.5% in 2020...READ MORE
- This Week in Managed Care: November 1, 2019 (ajmc.com)
Christina Mattina, welcome to This Week in Managed Care from the Managed Markets News Network
- FDA blames market failures for drug shortages (biopharmadive.com)Drug Shortages: Root Causes and Potential Solutions, A Report by the Drug Shortages Task Force 2019 (fda.gov)
An inter-agency U.S. task force looking into drug shortages found "a broken marketplace" and issued a set of recommendations that includes creating a rating system for manufacturers...Medicines that fall into shortage are often relatively inexpensive and can become unprofitable for manufacturers...Creating a manufacturer ratings system might be one way to incentivize higher prices... the rating system for manufacturers would address one key aspect of the broken marketplace...Manufacturers who have gone beyond basic requirements and have a "mature" quality management system are more likely to have reliable production, and purchasers who can identify these manufacturers through ratings may be willing to pay more for their drugs...The task force also advocated new approaches to contracts that might include financial incentives to make steady production of critical drugs worthwhile for drugmakers. Purchasers could pay higher prices for medications made at top-rated facilities or require a certain rating before entering a contract...READ MORE
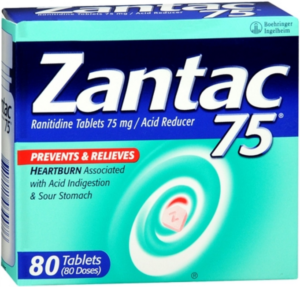
- FDA downplays risks from already recalled heartburn drug Zantac (reuters.com)
...Food and Drug Administration...downplayed the risk caused by contamination of the already recalled popular heartburn drug Zantac, and said its tests suggest it does not cause carcinogens to form after ingestion by patients...Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said...that the levels of the probable carcinogen N-nitrosodimethylamine it found in ranitidine...“are similar to the levels you would expect to be exposed to if you ate common foods like grilled or smoked meats.”...Still, the FDA released results of its testing here on branded and generic ranitidine products that showed some versions of the over-the-counter medicine had as much as nine times the regulator's "acceptable limit" for NDMA...READ MORE
- November 1 Week in Review (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.