- California bans pharma’s infamous ‘pay-for-delay’ deals (fiercepharma.com)
When generic challengers come for a branded med’s patent, drugmakers have in the past chosen to pony up and stall their rivals with an anticompetitive pact better known as “pay for delay.” In an effort to keep drug prices down, California is looking to end the practice...a new bill...will make California the first state to ban pay-for-delay deals in pharma...AB 824, will make it unlawful for companies to exchange anything of value in return for a halt to patent challenges from generic drugmakers. That new measure could open the door to a range of civil suits against companies seeking to keep generic competitors off the market...READ MORE
- Virginia doctor who illegally prescribed 500,000 opioid pills sentenced to 40 years in prison (cnn.com)
A Virginia doctor convicted in May of illegally prescribing more than half a million opioid tablets was sentenced...to 40 years in a federal prison...Joel Smithers, who was convicted of 859 counts of illegally prescribing drugs... was sentenced to 480 months in prison in US District Court for the Western District of Virginia in Abingdon, where he was also given a supervised release of three years and ordered to pay a special assessment of $86,000...READ MORE
- 340B allies rally Congress to ensure Pelosi drug price plan doesn’t imperil discounts (fiercehealthcare.com)
A 340B advocacy group is imploring congressional allies to ensure House Speaker Nancy Pelosi’s drug prices plan won’t prevent hospitals from getting discounts under the program...The hospital industry-backed advocacy group 340B Health sent a letter to the House Energy & Commerce Committee...extolling the virtues of the program. The letter comes after the release of Pelosi’s Lower Drug Costs Act, which would call for the Department of Health and Human Services secretary to select at least 25 drugs a year to negotiate for a lower price...Currently, the legislative text says that for each year that a negotiated price is applied that drug shall “not be considered a covered outpatient drug subject to an agreement under … 340B.”...READ MORE
- CVS pulls Zantac and generics as recall spreads around the globe (fiercepharma.com)FDA says finds 'unacceptable level' of carcinogen in Zantac and its generics (reuters.com)
The Zantac recall is quickly turning into a landslide as CVS pulled Zantac and its generics from its shelves and countries from Korea to Bangladesh asked makers to remove ranitidine drugs from their markets...CVS Sunday said it was suspending sales of all Zantac brand and CVS Health brand ranitidine products until further notice. That came despite the fact the FDA has yet to seek the recall of the products or recommend that patients quit taking the heartburn med after it warned a suspected carcinogen has been found in them...FDA alerted the public Sept. 13 that it had been determined that NDMA could be produced during the manufacturing of ranitidine drugs. At the time, it said the amounts found in the drugs appeared to be less than would be found in common foods, and their risks seemed small...READ MORE
- This Week in Managed Care: October 4, 2019 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Nevada levies $17 million in fines on drug companies for noncompliance with diabetes drug transparency law (thenevadaindependent.com)
The state is imposing $17.4 million in fines on 21 diabetes drug manufacturers that have either failed to comply with or were many months late in complying with a drug pricing transparency law passed two years ago...The fines, which the state is allowed to assess at $5,000 a day, range from $735,000 for one company that submitted the required drug pricing data the same day it received a final notice from the state — but 147 days after the reporting deadline — to $910,000 for eight companies that still have yet to report the required information. The Department of Health and Human Services told the companies in letters sent this week that they have 30 days to either pay the fines in full or 10 days to request an informal dispute resolution meeting with the state...READ MORE
- Google parent Alphabet hires former FDA head Robert Califf to lead health strategy and policy (fiercehealthcare.com)
Google parent company Alphabet has hired former U.S. Food and Drug Administration Commissioner Robert Califf, M.D., to...serve as head of strategy and policy across the company's Google Health and Verily Life Sciences enterprises beginning Nov. 18...He served at the FDA during President Barack Obama’s administration, first as deputy commissioner for medical products and tobacco from 2015 to 2016 then as commissioner of food and drugs from 2016 to 2017...READ MORE
- October 4 Pharmacy Week in Review (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- FDA issues warning on medical devices that are vulnerable to takeover from hackers (cnbc.com)
The Food and Drug Administration issued a warning to consumers...about potentially serious cybersecurity flaws in some medical devices that could allow hackers to take control of them remotely...Medical devices that use third-party, decades-old software called IPnet are at risk, the FDA said. The regulator said it’s not sure how many or even which specific devices, such as insulin pumps or pacemakers, are vulnerable to getting hacked...Researchers have identified 11 vulnerabilities that may allow “anyone to remotely take control of the medical device and change its function, cause denial of service, or cause information leaks or logical flaws, which may prevent device function.”...READ MORE
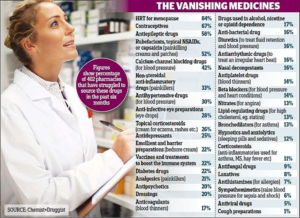
- Crisis at chemists: Pharmacies are ‘barely coping’ as dozens of popular medicines run short leaving hundreds of thousands of patients in the lurch (dailymail.co.uk)
Pharmacists have warned of shortages for every major type of medicine – including painkillers, contraceptives and diabetes pills...A poll of community pharmacists...revealed the dramatic extent of the drugs crisis that is hitting hundreds of thousands of patients in the UK...Pharmacists said they are living ‘on a knife-edge’ due to the mass shortages, which are caused by a perfect storm of manufacturing problems...READ MORE