- 340B Purchases Hit $12 Billion in 2015—and Almost Half of the Hospital Market (drugchannels.net)
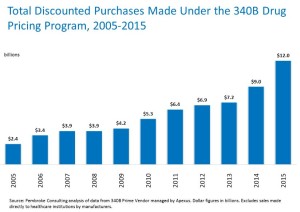
According to new data...discounted purchases made under the 340B Drug Pricing Program hit $12 billion in 2015. That’s a whopping 67% higher than the 2013 figure. I estimate that the undiscounted value of these purchases exceeds $17 billion...Most 340B purchases are made by hospitals. My...number-crunching below reveals that hospitals now receive 340B discounts on more than 44% of their drug purchases...two years ago, the 340B program is taking over the hospital market...How much of this money goes to uninsured and needy patients? No one knows, and the hospitals aren’t saying much...340B purchases have been growing much, much more quickly than have hospitals’ total drug purchases. From 2005 to 2015, total hospital drug purchases grew by 31%, compared with the 400%+ growth in the total 340B purchases...340B has infiltrated almost half of the hospital market...The 340B program is highly controversial, partly because the 340B legislation does not specify or restrict how covered entities should utilize funds generated by the program. Hence, it’s troubling to see that uncompensated care as a percentage of hospitals’ total expenses has remained at about 6% for many years, despite booming 340B purchases...Defenders of the 340B status quo argue that covered entities should be able to use discounts to reduce their cost of operations, without any transparency or accountability...The Office of Inspector General has documented how 340B-eligible hospital outpatient departments earn tremendous profits from the Medicare Part B program. Gross profit margins are about 60% compared with 3% to 4% for a non-340B outpatient program...
- Most drug makers report incomplete side effects to the FDA (statnews.com)
The regulatory system for reporting side effects caused by prescription drugs is producing its own kind of side effect — incomplete information about injuries that patients may have suffered. And the reason for the lack of data is that many drug makers are filing patchy reports...At issue is the Adverse Event Reporting System...The database is the key method for collecting side effect data. Both doctors and consumers can voluntarily report problems to the agency or a drug maker. But drug companies are also required to investigate and report side effects that may be attributable to their products...drug makers generally fail to include key data that the FDA might use to assess future warnings. For this reason...there are still wider implications...With increasing pressure for the FDA to approve drugs fast but with less clinical testing, it is a major concern that postmarket surveillance has major problems that are not being addressed…It is time for the FDA, the medical community, and industry to start work on a badly needed modernization" of this "critical tool" for monitoring safety…reports involving patient deaths offered the least amount of complete information for all of the key data points...the reporting system has not kept pace with changes in pharmaceutical marketing...interactions between patients and physicians, and patients and drug companies, have evolved, partly thanks to the Internet. This means a report may not be based on spontaneous information.
- ObamaCare May Be Growing The Number Of Unpaid Medical Bills (forbes.com)
A big hospital chain’s surprise decision to write off a slug of bad debt may be a signal of much deeper consumer healthcare strains being caused by ObamaCare...Community Health Systems surprised analysts this week...the company would take a $169 million provision for bad debt. The write off was a big part of Community’s dismal fourth quarter earnings report...The rising amount of uncollected co-pays and deductibles may be an early sign of consumer stress as the economy weakens. But more likely, it also reflects changes in the healthcare market that are saddling consumers with a much bigger share of their medical costs...ObamaCare is playing a big role...The structure of the insurance products offered under ObamaCare was deliberately skewed toward hollowed-out health plans. These plans sport large out-of-pocket limits and often skimpy or no co-insurance on drugs and doctors purchased outside a health plan’s increasingly narrow drug formularies and provider networks...Americans are now less insured for medical costs…The earnings report from Community Health was an early financial harbinger of this trend. Collecting on these rising out of pocket costs will get more difficult for healthcare providers as the ObamaCare insurance designs become the new market standard. This will be ObamaCare’s legacy – a new standard for hollow health coverage.
- Dreaded U.S. FDA Form 483s in the spotlight in China (fiercepharmaasia.com)
While much of the news in recent months on warnings issued by the Food and Drug Administration centered on alerts issued to Indian pharmaceutical manfacturers, active pharmaceutical ingredient and drug manufacturers in China have also received their own set of alerts under the FDA's Form 483 regime, according to a report by the Regulatory Affairs Professionals Society... 41 pharmaceutical manufacturing sites in China and 5 in Hong Kong are currently included on the agency's import alert list and are banned from shipping drugs to the United States, compared to the 42 sites listed as banned in India...RAPS found that in 2015 more than 80 Form 483s were issued to Chinese manufacturers after 132 inspections...the FDA conducted 203 inspections at Indian facilities...Some of the findings of the FDA reports include data manipulation at several companies where computer files were altered or deleted, missing logbooks, contamination issues and other serious failures in good manufacturing practices...
- Some doctors slow to sign up for electronic death certificate filing (reviewjournal.com)
Nevada's Office of Vital Statistics issued a call this month for relevant physicians to sign up as quickly as possible for a system that allows doctors to fill in required information for death certificates electronically. That system became mandatory statewide Jan. 1...Some doctors did not sign up after the state began notifying them of the change in fall because they have had issues registering or have outright refused to participate, vital records program officer Jason Lewis said...Karen Massey, chief administrative officer at Northern Nevada Emergency Physicians, said physicians have struggled with some requirements of the system, including its incompatibility with Windows 10 and Apple computers. The changeover has been time-consuming...I'm dismayed when it sounds like physicians haven't been responsive about it...Physicians have raised several concerns and have been working with the state to get them resolved...
- ACA ‘Savings’: Paying Doctors And Hospitals Bonuses To Deny Care To Patients (forbes.com)
...one of the lesser-known provisions of the ACA (Affordable Care Act) calls for the federal government to pay physicians and hospitals bonuses if they deny health care to seniors and the disabled...Section 3022 of the ACA establishes the Medicare Shared Savings Program. The MSSP establishes the notion of Accountable Care Organizations. These are groups of health care providers (hospitals, physicians, other providers) who join together for purposes of a obtaining bonus payments based on their participation in the MSSP and Medicare fee-for-service incentive program...ACOs are paid to "reduce costs" for treating their patients...For patients assigned to ACOs, the ACO receives bonus payments based on the total amount Medicare pays for care of those patients. That total includes all care those patients receive from all providers – specifically, including providers who are not members of the ACO. Bonuses are based on reducing total costs relative to what would be expected risk-adjusted cost based on each patient’s health status...Because this program is part of Medicare’s fee-for-service system, "reduce costs" means "reduce services" – that is, "give patients less health care."...the ACO program encourages hospitals and physicians...to encourage patients to use less health care...It is highly ironic that a law proposed, in part, because of the allegation health insurance companies were increasing their profits by denying care to patients – is now the means by which the federal government pays physicians and hospitals bonuses to, in effect, deny care to patients...
- Teva Said to Seek Fast EU Approval for Allergan Unit Bid (bloomberg.com)
Teva Pharmaceutical Industries Ltd. will try to win an early European Union approval for its takeover of Allergan Plc’s generics unit by making concessions to regulators this week...Teva and Allergan will meet a deadline to propose remedies aimed at allaying any EU antitrust concerns...If EU regulators are satisfied with the proposals, they could clear the transaction without an extended probe that can last about 90 working days. The companies plan to close the deal by the end of March...Teva’s $40.5 billion cash-and-stock bid for Allergan’s generics drugs business will make it the world’s largest maker of generic medicines, giving it greater negotiating power with governments and private-health insurers. Allergan will receive a $1 billion termination fee from Teva if the deal fails to close due to regulatory issues.
- Senate confirms Dr. Robert Califf to lead FDA (reuters.com) New FDA head Robert Califf vows to use ‘bully pulpit,’ better explain agency decisions (washingtonpost.com)
The Senate voted overwhelmingly...to confirm Dr. Robert Califf as head of the Food and Drug Administration, an agency that regulates everything from food and drugs to tobacco, cosmetics and dietary supplements...Califf...a well-regarded cardiologist and researcher, takes the helm at the FDA when lawmakers are pressuring it to speed the approval process for drugs and medical devices and to finalize a proposed rule giving it authority to regulate e-cigarettes...He said one of his first priorities is to strengthen the workforce by reaching out to academic and other centers to attract new talent...Another priority...is improving surveillance systems to monitor for safety...We're not proposing to do away with the adverse event reporting system that currently exists...but we are acutely aware that it is not enough...Tools to monitor the safety of medical devices also need to be modernized...and though it will not happen overnight...we have to do the hard work of making it happen...
- Health exchange disputes federal report (reviewjournal.com)
Nevada's health insurance exchange disputes a federal report claiming it misallocated funds for creating its program and recommending it consider refunding $893,000 to the Centers Medicare and Medicaid Services...A refund is not needed, the Silver State Health Insurance Exchange stated in a written response to the report from the Office of Inspector General, Department of Health and Human Services...We believe that this conclusion is based upon an erroneous interpretation of federal guidance regarding cost allocation accounting principles and the timing of cost allocation adjustments...It also pointed to comments from CMS that not only back up the state agency's take on the matter but are also included in the OIG's own report...In its report, however, the OIG was not swayed, stating it believes "our first recommendation is valid." The OIG cited the Nevada agency for using outdated data when it was establishing its marketplace despite the availability of updated data, having no internal controls, allowing insufficient staff oversight, and having no written policy on the allocation process...The Nevada marketplace should have used the updated, better data to update its cost allocation methodology...
- Community pharmacy reforms (rpharms.com)
Changes to community pharmacy in 2016/17 and beyond...In a letter to the Pharmaceutical Services Negotiating Committee...the Department of Health and NHS England announced there would be a...reduction in funding through the community pharmacy contractual framework...this reduction in funding will have a substantial impact on pharmacy business owners, their employees and locums...it was announced that funding...would be cut by at least 6%, equating to £170 million ($242 million); lowering the funding available through the community pharmacy contractual framework from £2.8 billion ($4 billion) to £2.63 billion (3.75 billion). It is anticipated these changes will take effect in October 2016...Alongside the cut in funding...further changes that the Government would like to see, including:
- To better integrate pharmacy into the wider primary care and community health system – such as closer working with GP surgeries, care homes and urgent and emergency care departments - so that patients can benefit from pharmacists’ clinical skills through a "Pharmacy Integration Fund".
- A "Pharmacy Access Fund", which would provide more NHS funds to certain pharmacies compared to others, considering factors such as location and the health needs of the local population.
- The potential for automation and centralising dispensing to provide efficiencies. Also, in some areas, the distribution and number of pharmacies does not match local patients' needs.
- The view of the Government that patients would like to order prescriptions online and that "click and collect" and delivery options would be easier to arrange.