- Pozen and Tribute, spooked by tax rule changes, kill plans to put merged company in Ireland (fiercepharma.com)
The U.S. Treasury's latest stab at deflecting tax inversions deals has persuaded at least one player to give up plans for a move to Ireland, but not from looking outside the U.S. for some tax relief. Pozen, which was headed to Ireland as part of its merger plans with Tribute Pharmaceuticals, will set up shop instead in Tribute's home turf of Canada…The two drugmakers said recently they charted the change of course "after reviewing the recent guidance from the Treasury's Notice 2015-79 issued on November 19, 2015 and its potential impact on the proposed transaction."…Pozen and Tribute recommitted to their merger but said that they will go to Canada instead…The new company, which will be renamed Aralez Pharmaceuticals, has a $350 million commitment from a syndicate led by private equity firm Deerfield Investments.
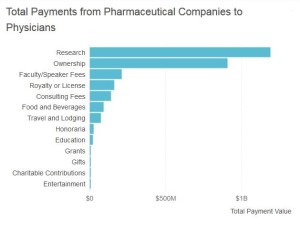
- Big Pharma Payments for High Prescribing Doctors (news.yahoo.com)
In an ideal world, doctors avoid outside influence and commit to ethical practice. However, they are also huge influencers in a for-profit pharmaceutical industry. This creates a breeding ground for potential conflicts of interest…In order to make relationships between health care providers and pharmaceutical companies more transparent, the Physician Payments Sunshine Act was enacted into law…Open Payments is the program that collects this information…Within this data, a pattern emerges: doctors who prescribe a lot of drugs tend to get more money from pharmaceutical companies…this data…identifies doctor and pharma relationships, it still does not determine causation. It only brings up certain questions: Are doctors prescribing specific drugs more because a pharmaceutical company is paying them? Or do pharma companies pay certain doctors because they prescribe a lot of their drugs? Further complicating the analysis is the knowledge that some physicians prescribe certain drugs in high volumes simply because they’re the best for their patients…While some argue that physicians should have nothing to do with drug marketing, others believe that collaboration between doctors and pharmaceutical companies is critical to improving the health and quality of life for patients. Not all doctors work simultaneously as drug prescribers and drug marketers, but those who do must strike a careful balance.
- How Stable Are the ACA Marketplaces? (commonwealthfund.org)
The news that UnitedHealth Group is considering leaving the new health insurance marketplaces established under the Affordable Care Act has prompted some concern about the their long-term viability...United Health Group’s possible departure is not really the issue. The insurer was a minor player in the individual market before the ACA passed, and currently covers only about 5 percent of marketplace enrollees…together with a few other developments, such as the failure of COOPs, the UnitedHealth Group announcement raised the fundamental question of whether the ACA marketplaces are enrolling sufficient numbers of people to ensure a well-balanced risk pool of healthy people and those with health problems. A pool where a majority of people have health problems could lead to higher premiums over time and destabilize marketplaces.
- FDA delays proposed rule on safety warnings for generic drugs again (statnews.com)Supplemental Applications Proposing Labeling Changes for Approved Drugs and Biological Products (federalregister.gov)
For the second time, the Food and Drug Administration has delayed a controversial rule for updating generic drug labeling that would allow generic drug makers to independently update safety warnings, something only brand-name drug makers can currently do before receiving regulatory permission…the agency said the rule will be published in July 2016…The delay came amid push-back from the pharmaceutical industry, as well as some congressional lawmakers who questioned whether the agency has the authority to issue such a rule…FDA said it wants to “create parity” between brand-name and generic drug makers…generic drug makers have not been required to run tests to prove their medicines are the same as brand- name drugs and, therefore, have not had to upgrade labels with safety information…the generic trade group has argued the FDA rule could add $4 billion to the nation’s health care bill because regulatory requirements and litigation costs would eventually force manufacturers to raise prices.
- 9 organizations urge Congress to oppose delay of Stage 3 (healthcareitnews.com)
Health IT Now, along with eight other organizations…are calling on Congress to stay the course on Stage 3 of the Meaningful Use EHR Incentive Program…Health IT Now describes itself as a broad-based coalition of patient groups, provider organizations, employers and payers…The coalition, along with eight other organizations made their pleas in a…letter…"We write to urge you to oppose any legislative changes to the meaningful use program, including delays in the timing of Stage 3, that do not also include reforms to improve the interoperable use of health information technology,"…"Delay without reform would rob taxpayers and patients of cost savings while doing absolutely nothing to make the program work well for overburdened doctors and hospitals."…The American Medical Association and 111 other national and state medical societies called for delay of Stage 3 in an earlier letter to leadership...
- 5 things to know about CMS’ mandatory bundled payment program (healthcareitnews.com)
CMS announced the first mandatory test of shared-risk, outcomes-based payment model and the first initiative to make hospitals financially-responsible for patient recovery, 90-days after a knee or hip replacement surgery…The landmark program, Comprehensive Care for Joint Replacement, goes into effect April 1, 2016. Patient engagement and care coordination technology will be key to helping hospitals meet cost containment requirements…can lead to lower costs and increased healthcare quality…Healthcare institutions and leaders should know five items to move forward into the coordinated care initiative:
- 789 hospitals will be impacted
- 500,000 patients can participate, annually
- Quality matters. Hospitals must adhere to strict quality metrics and keep down care costs from admissions to 90 days after discharge
- Hospitals can donate $1,000 per Medicare beneficiary for patient engagement IT and services
- CMS will provide incentives for Patient Reported Outcomes
- Three types of marijuana to hit Uruguayan pharmacies in 2016 (reuters.com)
Uruguayans will be able to choose from three varieties of state-sanctioned cannabis when marijuana starts being sold in pharmacies in the small South American country next year…Each variety will have different levels of...tetrahydrocannabinol and cannabidiol..."There will be three options with indications about the effects of each that point beginners toward starting with the lowest level" of THC...Uruguay became the first country to legalize the cultivation and distribution of marijuana in late 2013, aiming to wrest control of the trade from gangs while regulating and taxing its consumption…Authorities have developed traceable, genetically-distinct plants to ensure they do not leave Uruguay's borders or end up on the black market…The government estimates registered marijuana users will be able to buy the drug from pharmacies in mid-2016, when the country's two licensed producers start selling their first commercial load of about four tonnes…Authorities are also working with companies interested in exploring the possibility of exporting medical marijuana to the United States and Europe...
- China toughens drug quality standards, rejects 13 applications (reuters.com)
China's food and drug regulator said late on Monday it had rejected applications for 13 new drugs, citing false or incomplete trial data, as the government toughens enforcement of quality standards…China Food and Drug Administration last month also rejected applications by eight Chinese companies for inadequate trial data related to generic drugs for heart problems, schizophrenia, pain, infections and other diseases…quality of locally made drugs is a priority for the government, which is pushing an ambitious program of healthcare reforms to reduce reliance on both generic and more innovative imported drugs.
- Medical board removes 3 Southern Nevada doctors from probation (reviewjournal.com)Local doctor threw drug, sex parties at his Henderson home (reviewjournal.com)
Three Southern Nevada physicians were taken off regulatory probation Friday, including a Las Vegas orthopedic surgeon accused of throwing drug-fueled sex parties at his Henderson home and at the Trump Hotel…Dr. Andrew Martin declined to answer any questions after his appearance before the Nevada State Board of Medical Examiners at the Westgate…The board also lifted probation for Dr. Sean Su, a family practitioner barred by the board from performing cosmetic procedures, and Dr. Arlyn Valencia, a neurologist who failed drug tests.
- State, Local Officials Push Manufacturers to Pay for Drug Disposal (realclearhealth.com)
...drug disposal is expensive — the Nebraska program spends $10 per pound to ship and destroy medication — and some states and municipalities want drug companies to pick up the tab…Six municipalities in California require drugmakers to pay for take-backs. Nine states...have considered similar measures...Proponents say these laws, which are similar to programs that require manufacturers to pay for electronics recycling, would make it easier for patients to dispose of prescriptions. But drugmakers oppose the local mandates and warn that disposal costs could be passed on to patients through higher drug prices…Alameda County in California was the first jurisdiction to pass a law requiring manufacturers to pay for take-backs, in 2012, though the industry was quick to file suit, arguing it violated the interstate commerce clause of the U.S. Constitution. The Alameda law was upheld this year when the Supreme Court refused to hear the industry’s appeal…Drugmakers would rather lead their own take-back programs than comply with local or state mandates, said Priscilla VanderVeer, of the Pharmaceutical Research and Manufacturers of America...Because drugmakers sell across the country, meeting the customized disposal requirements of local programs is complicated...There are also more effective ways to get rid of the medicines, she said, like at-home disposal or returning unwanted drugs using special envelopes with carbon lining…“That’s just a financial and logistical nightmare for something that’s not necessarily going to be effective, or secure or environmentally helpful,” she said.