- Indian API maker Pan Drugs is reconstructing its manufacturing suite after holes in the wall and rusty equipment landed the facility a US FDA warning. (in-pharmatechnologist.com)
Pan Drugs received the Warning Letter…with the US Food and Drug Administration citing a number of deviations from GMP (good manufacturing practices) standards during an inspection of the Nandesari, Vadodara facility (India)…failed to maintain, repair, and keep clean its API manufacturing units,…holes in the walls and roof which allowed pigeons access near production equipment…gaps and holes in outside walls for piping and air ducts which allow contaminants to enter the building…We have given the commitment to stop all production while we renovate the facility,…
- Quarantine All Sterile Drugs from Compounder Medistat (pharmacytimes.com)FDA Announces Medistat RX’s Nationwide Voluntary Recall of Sterile Drug Products (fda.gov)
All non-expired drugs produced for sterile use by Alabama-based compounder Medistat RX have been recalled due to potential contamination, the FDA is warning pharmacists… recalled products were distributed nationwide between Nov. 1, 2014, and Sept. 3, 2015… FDA investigators and Alabama inspectors observed significant deficiencies that raised concerns about the compounder’s ability to assure the sterility of the drugs it produced.
- The Promotion of Medical Products in the 21st Century – Off-label Marketing and First Amendment Concerns (jama.jamanetwork.com)
On August 7, 2014, Federal District Court …blocked the Food and Drug Administration from enforcing restrictions on the marketing and promotion of off-label use of the drug icosapent ethyl (Vascepa),..If the case heralds the future of jurisprudence, responsibility for the oversight of the truthfulness of pharmaceutical promotions may shift from the nation’s leading science-based regulatory agency, the FDA, to the courts. If it does, the market for medications in the 21st century may revert to a time of more claims and less evidence to guide clinical care… Judges should refrain from using the First Amendment to undermine core regulatory functions.
- EMA’s medical literature monitoring enters into full operation (worldpharmanews.com)Monitoring of medical literature and entry of adverse reaction reports into EudraVigilance (ema.europa.eu)
The European Medicines Agency has started its full medical literature monitoring service...Medical literature…source of information on suspected adverse reactions to medicines…European Union's pharmacovigilance legislation has given EMA responsibility for the monitoring of selected medical literature for a defined list of active substances used in medicines and for entering identified reports of suspected adverse reactions in EudraVigilance,..By reducing duplication of reporting from multiple pharmaceutical companies, the initiative will improve the safety monitoring of medicines by enhancing the quality and consistency of data reported...
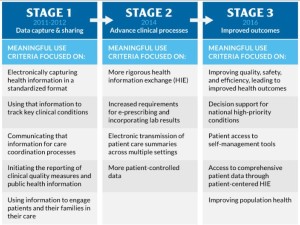
- HELP Committee chairman calls for flexibility in implementation of Stage 3 EHR rules (modernhealthcare.com)AMA urges hitting Stage 3 pause button (healthcareitnews.com)
Sen. Alexander, chairman of the Senate Health, Education, Labor and Pensions Committee,…called for a delay and a phased-in implementation of Stage 3 of the electronic health-records program…we should delay until Jan. 1, 2017, making of the final rules for Stage 3 of the federal government’s program to require doctors and hospitals to create electronic health-record systems…should be phased in at a rate that reflects how successfully the program is being implemented…about a quarter of a million physicians and about 200 hospitals face Medicare penalties if the current Stage 2 rules are not revised. Only about 12% of physicians and 40% of hospitals have been able to comply thus far …
- 3 ways pharmacies can build CMS Star Ratings (drugtopics.modernmedicine.com)
Pharmacists can adapt to the evolution of their industry by understanding treatment guidelines, learning about local physician practices, and improving patient adherence,...these actions contribute to quality healthcare...This matters to pharmacists because they’re increasingly facing pressure to join preferred networks that link them to the success or failure of health plans… Medicare Star Ratings… help…patients choose which plan to enroll in… Plans with low Star Ratings…risk losing their CMS contract...
- Will ICD-10 conversion be worth the trouble? (modernhealthcare.com)
The required Oct. 1 conversion to the ICD-10 coding system will be worth the costs and headaches, supporters say….With up to seven alphanumeric characters in ICD-10 compared with a maximum of four numeric digits in ICD-9, there will be lots more room for the codes to accommodate new medical conditions and procedures. With ICD-9, "we've just run out of space," … Advocates also say ICD-10 will allow providers to keep better track of patient care and aggregate data to perform quality-improvement analysis. It also should give hospital and physician leaders sharper tools for analyzing the impact of new procedures and managing population health. They'll be better able to track individual pracces and identify inappropriate practice variation.
- Pharmacy accidentally gave man chemo (radionz.co.nz)Patient given cancer drugs rather than immunosuppressant (mp3) (radionz.co.nz)Decision 14HDC00439 (hdc.org.nz)
Deputy Health and Disability Commissioner (New Zealand) has found a man wrongly took a chemotherapy drug (cyclophosphamide) for three weeks because a pharmacy dispensed the wrong medication…The 79-year old man was supposed to be taking an immuno-suppressant (cyclosporine), following a kidney transplant…He took the chemotherapy drug for about three weeks before asking the pharmacist why his tablets had changed…The pharmacist checked the records and found a technician had filled the prescription, but that he had signed it off... he did nothing further, he didn't complete an incident form, he didn't alert the owner of the pharmacy…the pharmacist was also remiss for not offering any counselling to the patient…pharmacist has been ordered to apologise to the man in writing, and further action could follow.
- Marijuana dispensary to reopen after running out of supply (reviewjournal.com)
Clark County's only medical marijuana dispensary was forced to close its doors for nearly a week after it ran out of supplies due to widespread failures in the safety tests performed on growers' crops…Euphoria Wellness plans to reopen Tuesday morning, having closed last week on Wednesday afternoon,… Any marijuana sold in Nevada must first be tested and approved by a state-licensed laboratory. The marijuana must meet strict standards approved by the state, including what amounts to a zero-tolerance rule for pesticides.
- U.S. task force narrows recommendation for aspirin use (reuters.com)U.S. Preventive Services Task Force (09/14/2015) (uspreventiveservicestaskforce.org)
People between the ages of 50 and 59 years at an increased risk of heart disease and stroke should take daily low-dose aspirin,…In addition to preventing heart attacks and strokes,…may reduce their risk of colon cancer if they take aspirin for at least 10 years, according to the U.S. Preventive Services Task Force,...Under the Affordable Care Act, the task force's recommendations are used to help set health insurance reimbursement policies.