- HIPAA breach for hospital after worker swiped patient data (healthcareitnews.com)
A 12-hospital health system is notifying hundreds of its current and former patients that their protected health information has been compromised after discovering an employee was involved in identity theft…Merit Health system based in Jackson, Miss., only learned of the breach after local law enforcement notified them that one of their employees…was under investigation for identity theft,..The employee was allegedly swiping patient files for more than a year undetected,..swiped records containing patient names, Social Security numbers, medical diagnoses data, health plan data and also payment information.
- Millions in the red, a Nevada Obamacare insurer has failed (reviewjournal.com)
Nevada's health insurance exchange is losing its only locally based carrier. Nevada Health CO-OP, a nonprofit insurer created by the Affordable Care Act and federally funded to offer health coverage through the Nevada Health Link marketplace, said Wednesday that it cannot make enough money to stay in business after Jan. 1…second year of high claims costs and limited growth projections for enrollment made it "clear" the insurer would have a hard time providing "quality care at reasonable rates" in 2016.
- Generic Drug Sponsors Will Get Status Reports– But Only For Old ANDAs (pharmamedtechbi.com)
FDA is ready to restore some informal communications for generic drugs, but only for applications not covered by the newly implemented user fee action dates…applicants with pending ANDAs (abbreviated new drug application) that are not covered by user-fee goals will have the right to a status report under a new policy …governing communications with ANDA applicants in response to widespread criticism from the generic industry about the loss of interaction that followed enactment of the Generic Drug User Fee law..
- Pharma junkets for U.K. docs trigger tough new Sunshine rules, penalties (fiercepharma.com)NHS bosses paid by drug firms (telegraph.co.uk)
Health Secretary says officials influenced by pharma payments could face jail time...England may have been behind the U.S. in shining the light on financial ties between pharma and physicians, but that's changing..National Health Service doctors be required to declare all gifts they receive from drugmakers. They'll also face losing their jobs--or even jail--if pharma payments influence their work..
- CMS appoints ICD-10 ombudsman (healthcareitnews.com)
William Rogers, MD, director of the Physicians Regulatory Issues Team at the Centers for Medicare and Medicaid Services, will act as ICD-10 ombudsman to the federal agency, He will work out of an ICD-10 coordination center in Baltimore.. Implementation of ICD-10 is only 34 days away… CMS has said it will not deny claims as long as the right code family is used, the agency will not send back a message if the wrong code is used.
- The FDA has spoken on biosimilar names. But will its hybrid proposal work? (fiercepharma.com)Naming and Biological Products (blogs.fda.gov)
After much industry lobbying and public debate, the FDA has proposed a system for naming biosimilar drugs. It's a sort of hybrid of the generic name on one hand and the unique brand name on the other. ..it would allow the FDA to make some biosims easy to substitute for the brand-name original--and others not so easy… both branded meds and biosims would share a "core drug substance" name… Each product would also carry a four-letter suffix….individualized product-by-product,..
- Safety net hospitals say new rating system doesn’t consider socio-economic factors (modernhealthcare.com)Medicare.gov Hospital Compare (medicare.gov)
...new federal tool aimed at helping patients determine the best hospital for their care is raising questions over whether it provides an accurate assessment of quality.. CMS is developing a hospital quality star-rating system for the Hospital Compare website… We are not confident that the measures currently available…to create a single, methodologically sound rating of all aspects of hospital quality,".. should incorporate risk adjustment for socio-economic factors so results reflect differences in treatment across hospitals.
- EDITORIAL: Finally, a new kind of pharmacy (reviewjournal.com)
Clark County's first marijuana dispensary opened for business Monday. Legal retail sales of pot commenced… Euphoria Wellness…the first medical marijuana… was commercially grown…More than 6,000 registered medical marijuana patients live in Clark County. There still isn't enough supply for all of them…considering Nevada dispensaries are allowed to sell to registered marijuana patients from other states, buyers can expect a limited supply for the near future.
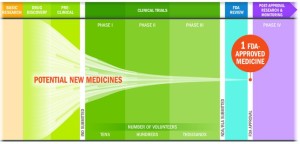
- Nine Explanations For Why The FDA Is Approving Almost Every New Drug Application (forbes.com)The drug development and approval process is about much more than the final “okay” (catalyst.phrma.org)
..Food and Drug Administration, which once approved as few as 40% of new drugs submitted by industry, has been on a green-light-almost-everything jag, approving 89% of drug applications. What’s more, a closer look showed an even higher approval rate. This year so far, 96% of new molecular entities.. – that have been submitted to the FDA have reached the market. For anyone who was watching the FDA a decade ago, that’s just shocking. Good or bad, it’s a radical change...there are a lot of factors that explain why the FDA approval rate is suddenly so high,..
- The approval rate is much lower, because only 12% of drugs that enter clinical trials reach the market.
- Drug companies are better at research, and they are simply producing better drugs.
- Drug companies are picking areas where the chances of approval are higher.
- The FDA is doing a better job communicating with companies.
- The FDA has more power to restrict the use of an approved drug than it used to.
- The FDA is taking a risk by taking strong stands against drug approvals right now.
- The FDA is without a permanent commissioner.
- It’s just random chance.
- In the current political environment, the agency is approving drugs it shouldn’t.
- Bass failed at challenging Acorda’s patents. Can pharma rest easy now? (fiercepharma.com)
Kyle Bass struck out. A U.S. review board (Patent and Trademark Office) nixed the hedge funder's challenge to two Acorda Therapeutics patents,.. showing Bass that his short-selling crusade against "low quality" pharma patents won't be an easy home run…Bass' very public challenges could work against him--particularly given the fact that he's made no secret of his plans to make money off the IPR process (inter partes review), by shorting the shares of companies with so-called "weak" patents.