- Seizure-Symptom App Is Latest Apple Health Research Tool (bloomberg.com)
...soon, Apple Inc. iPhones and watches may be able to recognize when someone’s having an epileptic seizure, and call for help…Researchers at Johns Hopkins University have developed an app that can track and measure seizure symptoms, and is the first research app to combine sensors on both Apple’s watch and phone. Eventually, it may run in the background and alert caregivers that a patient is having an event….the app measures heart rate, movement and other data as the seizure progresses. The watch will also display queries designed to test whether the wearer is alert…
- DNA4Life announces U.S. launch of its pharmacogenetics report (drugstorenews.com)
DNA4Life announced the…launch of its Pharmacogenetic Report, which will be available…to patients… DNA4Life’s test analyzes a person’s DNA to predict how they will respond to 120 commonly prescribed medication. One of the main goals of pharmacogenetics is avoiding adverse drug reaction…"Our differentiated and more comprehensive analysis will assist healthcare professionals in customizing a personalized treatment plan for their patients…particularly those being treated for chronic conditions, like high cholesterol, depression, ADHD or pain."
- U.S. prescription opioid misuse and deaths increase (reuters.com)
The proportion of people reporting use of prescription opioids for reasons other than medical necessity fell between 2003 and 2013, but use disorders and overdose deaths increased…rate of nonmedical use of opioids – not using a prescription as directed, or using a prescription that was written for someone else – fell from 5.4 to 4.9 percent…the rate of use disorders rose from 0.6 to 0.9 percent… more patients are experiencing…progression from initial opioid use to frequent use…Drug overdose deaths involving prescription opioids increased from 4.5 to 7.8 per 100,000 people.. These numbers could come down if we reduce inappropriate opioid prescribing and use and develop new treatments for pain that are safer…
- Sky-high price of new stem cell therapies is a growing concern (latimes.com)
…public uproar about high drug prices has focused on outlandish cases…Biotech companies have launched late-stage clinical trials that could lead to federal approval of two marketable treatments backed by CIRM (California Institute for Regenerative Medicine), the state's $6-billion stem cell program…they may also put CIRM smack in the middle of a burgeoning debate over how to ensure access for all patients to life-enhancing or life-saving cures…stem cell and other advanced biologic treatments will be among the most expensive therapies...Proposition 71, the 2004 ballot initiative…was pitched to the voting public as an economic bounty waiting to be reaped…healthcare cost savings to the state government of as much as $6.9 billion, and up to $18.4 billion in savings for private insurers and other payers. Such savings haven't materialized. CIRM also requires companies to share their income from drugs developed with the program's backing on a sliding scale based in part on the magnitude of the profits.
- The Merging of Medical Products: Enhancing review of therapeutic and diagnostic combination products (blogs.fda.gov)
Combination products – medical products that do not fit into the traditional categories of drugs, devices, or biological products – are a growing and important category of therapeutic and diagnostic products under FDA’s regulatory authority…These products, that combine drugs, devices, and/or biological product ("constituent parts") with one another… Products in this category range from familiar products such as prefilled syringes and surgical kits to novel and innovative products, which target and enhance therapies. Examples of groundbreaking combination products include antibodies combined with drugs for targeted cancer therapy and products that mimic or replace organs, such as an artificial pancreas….we’re taking a number of steps to clarify regulatory requirements and improve our internal processes and IT systems.
- Issuing more guidance for review of combination products (e.g., our pending draft guidance document on human factors);
- Enhancing and simplifying data access and sharing for internal staff;
- Making it easier for staff to request and monitor inter-center consults;
- Updating and maintaining our internal contact directory for experts to review a combination product; and
- Improving our internal standard operating procedures for premarket reviews and compliance activities.
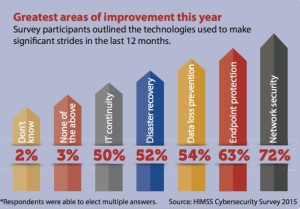
- Infographic: Greatest areas of improvement in cybersecurity (healthcareitnews.com)
This infographic from the 2015 HIMSS Cybersecurity Survey shows the greatest areas of improvement in cybersecurity that healthcare organizations were able to achieve this year.
- Patients’ social media posts may be the next big thing for big data (healthcareitnews.com)
There is a rich potential to identify health trends…Mining patients' social media data could provide enormous insight into overall health outcomes…By handling social media posts much like big data, it just may be possible to link social media posts to health outcomes…If patients consent to sharing this data, a research database could be created, comparable to that of genomic databases. This database could be used by researchers to better understand the relationship between certain patients and their health.
- 18k chemists down shutters in nat’l strike (timesofindia.indiatimes.com)Chemists’ strike hits patients (indianexpress.com)
Ranchi: Around 18,000 licensed chemists of Jharkhand participated in the nationwide strike of drug sellers on Wednesday to protest against the growing online sale of medicines in the country, posing problems for customers…Private drug stores in Ranchi and across 24 districts kept their shutters down while the Jharkhand Chemists and Druggists Association, the umbrella organization of drug sellers in the state, kept a handful of medicine shops operational to sell emergency medicines to consumers. They demanded the Union government check e-commerce in medicine.
- Indian drug retailers protest against e-pharmacies (reuters.com)Chemists all over India on strike today (timesofindia.indiatimes.com)
Indian drug retailers shut up shop for the day on Wednesday to protest against the country's growing online pharmacy industry, and threatened to close indefinitely if the federal government did not shut down e-pharmacies…nationwide protest was widely supported, with as many as 850,000 chemists closing their doors, leaving patients waiting in long queues at any pharmacies that were open…Drug retailers are worried…"It is a matter of our livelihoods, we must be prepared for a fight," said pharmacist Satish Vij, who traveled from northern Haryana state to take part in a protest in New Delhi, where about 1,000 people, mostly pharmacists, wore black arm bands, held placards and shouted slogans against e-pharmacies.
- QS/1 releases SharpRx pharmacy management software (chaindrugreview.com)
QS/1 this week launched SharpRx...pharmacy management software…brings touchscreen capability and an intuitive design and layout…SharpRx gives small chain and independent pharmacies a more efficient way to fill prescriptions and bill third-party insurance, in turn enabling faster customer service…allowing pharmacists to quickly navigate through the system…to do everyday tasks as quickly as possible…