- Viagra Price Drops 50% as Drug Faces Competition (247wallst.com)
Viagra, the wildly successful erectile dysfunction pill, faces challenges from a generic version that will launch next year, and direct competitors Levitra and Cialis. In the face of these, Viagra has started a promotion that offers 50% off a patient’s next three prescriptions...The offer does come with some fine print: home delivery ...In March, the mammoth generic drug company Teva Pharmaceutical Industries Ltd. got approval to market a generic version of Viagra. Teva already had FDA approval of its product. Due to a deal between Pfizer Inc. and Teva, the product will not come on market until the end of 2017. That leaves only 18 months for Pfizer to sell Viagra without generic competition. After that, it faces the Teva drug, which almost certainly will have a lower price.
- Drug maker loses battle over Canada’s right to impose a price cap (statnews.com)
A closely watched skirmish over the cost of prescription drugs has ended in defeat for a company that sought to challenge the right of a Canadian agency to impose a price cap on a pricey medicine...Canada’s Federal Court dismissed a constitutional challenge that Alexion Pharmaceuticals filed against the Patented Medicine Prices Review Board, which...sought to keep a lid on the cost of the company’s Soliris (eculizumab) medication...The...agency had asked Alexion to lower its price and repay sales generated by the drug from 2012 through the first half of 2014..."It is far from surprising," said Richard Gold, a professor at McGill University..."Alexion may still appeal in hopes that the Supreme Court of Canada will alter the law. I think the [possibility that the court will find] the provisions unconstitutional are remote. Parliament can create and limit patent rights as it wishes, but pharmaceutical companies have been known for being obstinate in the face of logic."
- Pfizer agrees to opioid marketing deal to escape a lawsuit by Chicago (statnews.com)
Pfizer reached an agreement with the city of Chicago to follow a code of conduct for marketing opioids that officials hope will become standard practice for other companies...The drug maker agreed last week to disclose in all promotional materials that opioids carry a risk of addiction and not to promote them for any unapproved uses...Pfizer must make clear that there is insufficient research about the effectiveness of opioids if used beyond 12 weeks…Some industry watchers, meanwhile, said that the four-year agreement, which does not pertain to any other city or state, largely requires Pfizer to adhere to existing laws and regulations governing drug marketing. For this reason, they said the agreement breaks little new ground in the battle against inappropriate or overprescribing of opioids..."This landmark agreement is a big step in the right direction to help protect and educate the public about the true risks and benefits of highly potent and highly addictive painkillers," said Chicago Mayor Rahm Emanuel in a statement...It’s in Pfizer’s interest to highlight the addictive properties of opioids because they have a competing product under development (tanezumab)...When a company comes out slamming a particular class of drugs, it’s generally because they have a competitor in the wings. Think of this as prelaunch marketing, which can start long before the launch...
- Gilead’s new price hikes on HIV drugs anger AIDS activists (statnews.com)Gilead hikes price of HIV therapies, other drugs by up to 10 percent (statnews.com)
As part of a strategy to switch patients to newer HIV treatments, Gilead Sciences...raised prices on a pair of older HIV medications that face patent expiration...triggering still more criticism by AIDS activists of its overall pricing strategies...the company raised the wholesale acquisition cost...for the two older medicines — Complera (emtricitabine, rilpivirine,tenofovir) and Stribild (cobicistat, elvitegravir, emtricitabine,tenofovir) — by 7 percent, to $2,508 and $3,469 a month, respectively. This follows price hikes of 7 percent and 5 percent last January...Meanwhile, Gilead left intact prices for two much newer versions of these drugs — Odefsey (emtricitabine, rilpivirine, tenofovir) and Genvoya (elvitegravir, cobicistat, emtricitabine, tenofovir alafenamide) – which remain priced at $2,346 and $2,578 per month...By boosting prices for the older HIV treatments twice in just six months, Gilead is clearly hoping that doctors will prescribe its newer drugs. And physicians have an added impetus for doing so: the newest Gilead medications are more potent and studies have indicated they are likely to cause fewer side effects...price hikes has angered AIDS activists, who say the higher prices reflect indifference to the ability of payers to absorb higher drug costs...AIDS Healthcare Foundation believes the price hike is a good reason to push to invalidate Gilead patents...The small savings from the cheaper new drugs pales in comparison to how cheap they would be if they went generic...
- Advocates hope shaming drugmakers discourages price spikes (finance.yahoo.com)California Senate Bill 1010 (openstates.org)
Frustrated by the rising cost of prescription drugs, California health advocates hope sunlight and a dose of shame will discourage drugmakers from raising their prices too quickly or introducing new medications at prices that break the bank...They're promoting legislation that would require drugmakers to provide advance notice before making big price increases. Pharmaceutical companies have come out in force against the measure, warning it would lead to dangerous drug shortages...California's SB1010 would require pharmaceutical companies to provide advance notice to drug purchasers before increasing the price of a drug by 10 percent or $10,000 a year. For generics, the threshold is $100 a month or 25 percent. Insurance companies would be required to report data on drug prices to state regulators, including the portion of premiums attributable to pharmaceuticals...Proponents hope the advance notice will give governments, insurers and pharmacy benefit managers a chance to negotiate...But drugmakers warn it could create regional shortages of some drugs if large pharmacy chains or distributors horde medications to beat the price increase. That would create an environment for speculators to drive prices up, not down.
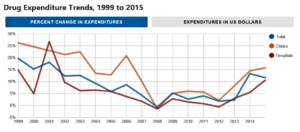
- Pharmacy Journal Examines National Trends in Prescription Drug Spending (ashp.org)National trends in prescription drug expenditures and projections for 2016 - Abstract (ajhp.org)
A sharp rise in prescription medication prices — driven by the introduction of new, expensive specialty medications and huge increases in the costs of older drug products with few competitors — drove an 11.7 percent increase in spending on medications in 2015, according to a new report published in AJHP (American Journal of Health-System Pharmacy)...The report projects an 11 to 13 percent increase in total drug expenditures in hospitals and health systems in 2016, which includes a 15 to 17 percent increase in clinic spending and a 10 to 12 percent increase in hospital spending...pharmacists need to be diligent about understanding what factors influence drug spending within their own organizations. "By understanding the key cost drivers, pharmacy leaders can implement programs to target them," he noted, adding that such strategies could include working with prescribers to use equally effective but less expensive medications or developing programs such as antibiotic stewardship to reduce unnecessary use of drugs...
- Drug maker is warned over missing study about child opioids use (statnews.com)
...one drug maker has failed to provide the Food and Drug Administration with a required study about the effect its powerful painkiller might have on teenagers...At issue is Xartemis XR (oxycodone/acetaminophen), which Mallinckrodt Pharmaceuticals received regulatory approval to sell two years ago. At the time, the company was also required to conduct a so-called post marketing study to determine its safety in youngsters between 12 and 17 years old…as of June 24, the company blew past a March 31 deadline for submitting its pediatric assessment. The drug maker also failed to respond to an April 25 "noncompliance" letter for which the FDA provided a new 45-day deadline to either submit the data or request an extension...The assessment was required under the Pediatric Research Equity Act...What happens if the company fails to comply and submit the data to the FDA? According to federal law, the agency could determine that Xartemis XR is misbranded. That would not lead to a product withdrawal, but the FDA could pursue an injunction or seizure proceedings...
- Painkiller panel drops experts linked to pharmaceutical industry (financialexpress.com)
One of the experts, Dr. Gregory Terman, said he was dismissed Tuesday afternoon by phone. He said he was told the decision was made because his nonprofit group, the American Pain Society, receives funding from drugmakers...A group advising the Food and Drug Administration on medical issues abruptly dropped four experts from a panel on prescription painkillers after concerns emerged about apparent ties to the pharmaceutical industry...Federal advisers are supposed to be vetted for financial ties that can influence their judgment. Senator Ron Wyden of Oregon sent a letter Friday to the academies’ leadership noting that two of the panel nominees had also served in professional societies that receive funding from drugmakers. Wyden has protested industry influence on federal expert panels before.
- Alibaba Health Expands Into New Consumer Business After Setbacks (bloomberg.com)
Alibaba Health Information Technology Ltd…the subsidiary of China’s biggest e-commerce operator is attempting to revive its fortunes by tapping new areas of growth -- like consumer safety...China...scrapped plans to implement a compulsory drug coding system intended to identify counterfeit medicines that would have used a platform developed by Ali Health. The Alibaba Group Holding Ltd. subsidiary says it is now using the same technology to build a commercial tracking platform that will have wider applications beyond medicines...Ali Health’s new system could be used to track the origins of products such as baby formula, farm produce and even wine for Chinese consumers concerned about food safety…Ali Health is currently talking to partners to expand use of the technology, and continues to run the existing drug tracking system for the government while awaiting a final decision. The China Food & Drug Administration suspended the electronic coding system earlier this year for use in the drug industry as it made draft amendments to existing rules, allowing the use of other methods to track medicines back to their origin...
- Q&A: Why the World Health Organization plans a fair pricing model for drugs (statnews.com)
...World Health Organization wants to develop a fair pricing model for pharmaceuticals...the agency plans to convene governments, patient groups, and drug makers to fashion the notion into something realistic. The trick is to find the right balance between access to affordable medicines and enticing companies to develop new and improved medicines, while also ensuring lower-cost generics remain available. We spoke with Suzanne Hill, the secretary to the WHO Expert Committee on the Selection and Use of Essential Medicines, about how the agency hopes to get this done…
Pharmalot: This seems rather lofty or at least complicated. How is this supposed to get done?
Hill: We want to get evidence together that looks at the price drivers — how manufacturers set prices and what we know and don’t know about that. And we need to understand the questions surrounding R&D costs. We also want to understand the strategies that countries, payers, and health systems are using to manage prices and which ones may be effective and which aren’t. We also want to understand the return on investment that manufacturers need to ensure that supplies of medicines are continued...