- J&J must pay $70 million to male teen who took Risperdal and developed large breasts (statnews.com)
Johnson & Johnson...was ordered...to pay $70 million to a male Tennessee teenager who claimed its Risperdal antipsychotic pill caused him to grow enlarged breasts. The finding by a Pennsylvania state court jury was not only the latest, but it is the biggest defeat to date in what has become another sprawling litigation over the drug....jury found that J&J failed to properly warn Risperdal could cause gynecomastia...also determined that the company "intentionally falsified, destroyed, or concealed records" that Risperdal could cause boys to develop breasts...J&J has a bad track record when it comes to marketing Risperdal...In 2013, the company paid more than $2.2 billion to resolve criminal and civil charges of illegally promoting the drug for unapproved uses...J&J reputation. The health care giant has portrayed itself as a trustworthy corporate brand, but has endured several episodes that have sullied its well-honed image...
- Anthem, Express Scripts Face Legal Challenge Over Prescription Drug Prices (realclearhealth.com)Anthem sues Express Scripts for $15 billion over drug pricing (modernhealthcare.com)
Anthem and its pharmacy manager Express Scripts overcharged patients with job-based insurance for prescription drugs, alleges a lawsuit that seeks class action status for what could be tens of thousands of Americans...the latest wrinkle in a battle that has already pitted the major national insurer and its pharmacy benefit manager against each other in dueling legal actions...The case alleges that insured workers paid too much because Express Scripts charged "above competitive pricing levels" and Anthem, in effect, allowed those higher prices...Those actions...violate the firms’ responsibilities under a 1974 federal benefits law called the Employee Retirement Income Security Act...This action seeks to recover losses suffered by the plaintiffs…who overpaid and continue to overpay for the portions of the costs of prescription drugs…they are responsible for paying as plan participants," says the lawsuit, filed as Burnett v. Express Scripts and Anthem.
- After Medivation, what’s next? Alexion, BioMarin, Incyte could be Big Pharma’s next M&A targets: Analysts (fiercepharma.com)
Megamergers could fall out of favor over the next year...small- to mid-sized deals up to $25 billion? Expect those aplenty given “the need many larger companies have to try and boost their growth prospects.”
Medivation’s prized prostate cancer pill Xtandi
Incyte, a $14.7 billion market-cap company that boasts myelofibrosis med Jakafi.
Epacadostat is entering Phase III combo studies...is being studied in a range of other tumor types
Alexion, a $27 billion company whose Soliris--the priciest med in the world--leads a portfolio of three approved rare-disease drugs.
BioMarin, the perennial subject of takeover buzz with 5 marketed meds and a host of pipeline prospects.
- Another big insurer will pay for key drugs based on patient outcomes (statnews.com)
Yet another health plan is aggressively pursuing deals with drug makers based on patient outcomes...Harvard Pilgrim Health Care...has reached agreements with Novartis and Eli Lilly to calibrate payments for two of their most important medicines...In one deal, the insurer will receive a discount from Novartis if its new Entresto (sacubitril/valsartan) treatment for congestive heart failure does not yield a specified drop in hospitalizations. In the other, Harvard Pilgrim agreed to accept a lower rebate from Lilly if its Trulicity (dulaglutide) diabetes drug lowers hemoglobin levels...Paying for value for drugs is an obvious way to go...The pharmaceutical industry is recognizing greater potential for regulation [on pricing], so I think they’re thinking more creatively and are more open-minded about how to develop contracts where cost is somehow related to outcome...The move toward so-called value-based contracting reflects accelerating costs for prescription drugs...It’s not rocket science to think about paying for value...Unfortunately, the pharmaceutical companies have been slow to adopt this...
- Court ruling on biosimilar launches could increase health care costs (statnews.com)
A federal appeals court...that biosimilar makers must always notify their brand-name rivals six months before launching expensive biologic medicines. The decision may have a significant impact on near-term health care costs, because it will effectively delay competition for these pricey drugs...The stipulation is designed to give a brand-name company time to determine what, if any, patent challenges can be pursued before a biosimilar is launched. In a closely watched case last year, the federal appeals court already ruled that biosimilar companies must wait until they actually receive Food and Drug Administration approval before giving 180-day notice to a brand-name rival...today’s decision will answer an important policy question and serve as a broader benchmark for the entire pharmaceutical industry going forward...A great deal is at stake for patients and payers because biosimilars are estimated to save as much as $44 billion in US health care costs over the next decade. Many insurers and analysts forecast that biosimilars will cost 10 percent to 30 percent less than brand-name biologics, although the number is a moving target, given that companies raise biologics prices to anticipate competition...
- FDA warnings slam Chinese drugmakers, including ViiV partner (fiercepharma.com)
...two Chinese companies with ties to Western drugmakers have been called on the mat to account for problems in their manufacturing. The FDA has issued warning letters to plants operated by Shanghai Desano Chemical Pharmaceutical and Chongqing Lummy Pharmaceutical, slamming them both for manipulating testing and turning in falsified batch test results on APIs...
- Shanghai Desano Chemical Pharmaceutical - FDA...criticized the facility for conducting "unofficial" tests of drug batches that it kept out of its official record...also disturbed by finding many electronic logs of production deviations in a folder titled "GMP Anomalies" that had never been investigated…
- Chongqing Lummy Pharmaceutical...FDA...warning letter slams the drugmaker for widespread and serious data manipulation of batch analyses...In one egregious case, the FDA...an analyst set the gas chromatography personal computer clock back to make it appear as if testing had been done...7 months earlier. The analyst then performed 5 injections to produce falsified results for long-term stability for a finished API lot, deleted four and reported only the results of the final injection as passing in the quality-control...
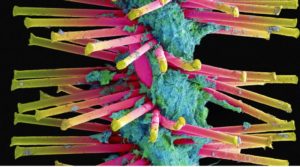
- Slimy clumps of bacteria kill thousands. Scientists are fighting back (statnews.com)
It’s a battle that seems ripped from a sci-fi film: Scientists are racing to develop new weapons to destroy the slimy colonies of bacteria, known as biofilms, that cause tens of thousands of deaths across the US each year...Biofilms are the leading cause of infections acquired in hospitals. They grow on medical devices such as heart valves, pacemakers, and catheters. They take root inside wounds, pulsing and rippling as they spread...Encased in gooey protective sheaths, biofilms are exceptionally hard to stop. Many are impervious to antibiotics. They also cost the health care system billions each year, as patients often require surgery to remove and replace contaminated implants...researchers and biotech startups are testing new methods of attack, from coating medical devices with spiky coverings to blasting bacteria with electrical fields to interrupting the chemicals that cells inside biofilm colonies use to send messages to each other...researchers at Ohio State announced they’d invented a way to coat the surfaces of medical devices with Y-shaped nanoparticles of quartz in a bid to block biofilms from latching on tight...
- Here’s a look at the battle:
- What are biofilms?
- Is this a new menace?
- Why are biofilms so hard to kill?
- So, what’s being done?
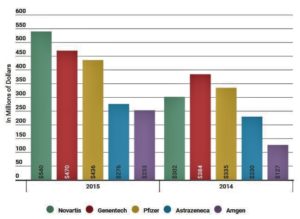
- These companies paid the most to docs last year (medcitynews.com)
The Physician Payment Sunshine Act was meant to shed light on the financial relationship between drugmakers, biotech, and medical device companies whether those payments be for general or research purposes...The Centers for Medicare and Medicaid Services has recorded these payments now for two complete years in a row with partial data available for 2013...The infographic below, created by MedCity News, based on data from CMS’s Open Payments website...shows the companies that spent the most money in 2015 in comparison with the amount they spent the year before and overall payments.
- Pfizer building modular biologics plant in China (fiercepharma.com)
Pfizer will build a biologics plant in China, where it will make biosimilars for the Chinese market but also for sale throughout the world. The New York drugmaker has turned to GE for construction of the facility, which has developed a modular construction process that will cut the cost and allow the plant to be operational in about 18 months, instead of three years...The $350 million facility, which it is building at the Hangzhou Economic Development Area in China, will be Pfizer’s third biologics production facility and its first in Asia. In addition to manufacturing, the facility will house Pfizer China’s Biosimilars and Biologics Quality, Technical Service, Logistics and Engineering divisions, and serve as a site for process development and clinical supplies. It will have about 150 employees when it is complete in 2018…The new center will be built using GE’s single-use technology…likened assembly to working with LEGO blocks...
- AstraZeneca sues FDA to prevent generic versions of Crestor (statnews.com)
Drug makers generally don’t complain when regulators widen the market for their medicines. But AstraZeneca filed a lawsuit...claiming the Food and Drug Administration is on the verge of illegally broadening the indication for its best-selling Crestor cholesterol pill, and the move would unfairly allow generic competition...The argument, which the company also made late last month in a citizen’s petition, hinges on the interpretation of federal law governing product labeling. Depending upon the outcome, AstraZeneca may either maintain a monopoly on Crestor for another seven years or face lower-cost rivals to a key revenue stream when the Crestor patent expires on July 8...the drug maker won FDA approval to sell Crestor to treat children with a rare genetic disorder called homozygous familial hypercholesterolemia...Under the Orphan Drug Act, the company was awarded an additional seven years of marketing exclusivity for Crestor, but only for treating this particular rare...disease...Several generic companies are lined up to sell a version of Crestor. AstraZeneca argues that a generic must include all pediatric labeling information approved for the corresponding brand-name drug. The company filed its lawsuit over concerns that the FDA will, instead, rely on a decision it made last year allowing generic companies to exclude certain information, so long as a safety risk is not created...AstraZeneca...believes federal law entitles the company to an additional exclusivity period of seven years for Crestor in the US...the American Journal of Clinical Oncology, a team of researchers argued that drug makers are exploiting loopholes in the Orphan Drug Act that allow them to widen the market for such drugs and distorting the original purpose of the law.