- The Startup Tracking ‘Valuable’ Doctors for Big Pharma (bloomberg.com)
Physicians are worth billions of dollars to drugmakers, who see the prescription pad as a path to profits. But it’s growing harder for Big Pharma to get doctors’ appointments. Since 2010, Obamacare has slowly curbed the mass travel junkets and fancy meals that drug companies once used to sway the doctors most valuable to their efforts to sell products...Pharmaceutical companies are now searching for ways to refine their marketing efforts, to target the doctors most compatible with the medications they’re pitching. "You’re desperate for data to make those key decisions,"..."But while there’s lots of data out there, it’s really challenging to bring it together."...Zephyr Health...promising to help drugmakers identify key medical personnel and find ways to approach them…Zephyr builds digital dossiers on individual doctors. It starts with basic information on prescription patterns from data clearinghouses...Then its software...scours the Web for more details...Zephyr generates profiles that score each doctor’s influence and ability to drive sales on a scale of 1 to 10. The software’s slick, mobile-friendly interface lets a drug company search in broad or specialized disciplines and ranks each person’s influence in the chosen field. It also specifies whether a doctor appears to influence colleagues or simply writes a lot of scripts..."There’s nothing private anymore,"...While doctors may not be exactly psyched about Zephyr tracking their every move...even they should appreciate the company’s ability to narrow marketing campaigns. For a physician, "working with Pharma is akin to getting pecked to death by a flock of ducks," he says. "Do you want nine salespeople queued up to call on you?"
- Boots changes prices after accusations of sexism (cnbc.com)
U.K. drug store Boots has bowed to pressure and cut the prices on some of its products after it emerged it was charging women more than men for similar products...An online petition started two weeks ago raised awareness of the price discrepancy between some of Boots' products. An eye cream for men cost £7.29 ($10.56) but the same product advertised to women cost £9.99 ($14.47). Meanwhile, a pack of 10 disposable razors for men cost £1.49, but a pack of eight razors for women costs £2.29...Boots said it conducted a review after the issue was brought to light and has taken action to correct the prices of its own-brand products. It also said it would speak to its suppliers and ask them to conduct a similar review of their brands..."At Boots UK, we have never operated a pricing system that discriminates against women so we were surprised and disappointed to see recent examples in the press that did not reflect our own standards,"...
- Aprecia completes $35M financing to support launch of the first 3-D printed medication (fiercedrugdelivery.com)
3-D printed medicine specialist Aprecia Pharmaceuticals announced that it has completed a $35 million financing round...The move should help...Aprecia commercialize the first FDA-approved 3-D printed drug, Spritam (levetiracetam), a reformulated, easy-to-swallow med for the treatment of epilepsy. The launch is expected to occur in the first half of this year...Aprecia has exclusive rights to utilize Powder-liquid 3DP, a 3-D printing technology developed by MIT in the 1980s. The technique enables the company's ZipDose delivery platform. By printing a tablet consisting of layers of powder, Aprecia drugs can achieve a high degree of dissolvability in liquid. That means dosages as high as 1,000 mg will disintegrate in liquid...The new manufacturing methodology could also facilitate decentralized drug manufacturing and customization of medications to the needs of individual patients.
- How Would Government Negotiation of Medicare Part D Drug Prices Work? (healthaffairs.org)
Medicare Part D premiums have been flat in recent years but will increase by 13 percent from 2015 to 2016...PDPs are also increasingly placing high-cost drugs on specialty tiers within formularies, which require patients to pay as much as 33 percent of the cost of these medications...Given these rising costs, a growing number of policymakers believe the government, through the Secretary of Health and Human Services, should be allowed to negotiate drug prices in Medicare Part D...federal law currently prohibits the government from negotiating with the pharmaceutical industry to lower the cost of drugs purchased under Medicare Part D. Instead, PDPs are required to negotiate directly with pharmaceutical manufacturers to obtain rebates and other discounts on drugs.
Proponents of government negotiation argue that HHS—because of its significant purchasing power—can more effectively negotiate drug prices than individual Part D plans.
Opponents argue that allowing the government to negotiate prices for Part D would inhibit innovation and limit beneficiary access to medications.
...what it means for the government to "negotiate" drug prices.
- Potential Scope Of Negotiations
- Which Drugs Would Be Negotiated?
- What Is Being Negotiated?
- Would HHS Negotiate For All PDPs?
- The Negotiation Process
- Additional Details Needed
- Estimated Cost Savings Of HHS Price Negotiations
- Next Steps
- The Challenge of High Cost Drugs in Specialty Pharmacy (specialtypharmacytimes.com)
Michael Zeglinski, vice president of Specialty Pharmacy Operations at BriovaRx, discusses how expensive hepatitis C drugs have impacted the specialty landscape. (video)
- Big price hikes widespread in pharma, with dozens doubling last year: Bloomberg (fiercepharma.com)Shkreli Was Right: Everyone's Hiking Drug Prices (bloomberg.com)
Lately, the spotlight has turned on Valeant and Turing Pharmaceuticals when it comes to the drug-pricing debate. But the price-hiking strategy goes far, far beyond that pair of drugmakers...A survey of roughly 3,000 brand-name prescription treatments has found that prices more than doubled on 60 of them since December 2014...And they've more than quadrupled on 20 of the medications over that same timeframe...The data shows that price increases are an integral part of the business plan... The Wall Street Journal pointed to a slew of Big Pharma moneymakers that owed part of their success to hefty price increases...And while Valeant has pledged to nix most of its planned price increases this year, recent political pushback and public scrutiny haven't convinced some of its peers to do away with the practice...As drugmakers have argued, boosting their list prices doesn't mean necessarily boosting their bottom lines all that much. Insurers and health plans negotiate discounts, meaning pharma companies don't reap the entire increase. Still...a higher starting point for negotiations helps them end up at a higher finishing point...Even if they don't get all of that price increase, they will get some of it...
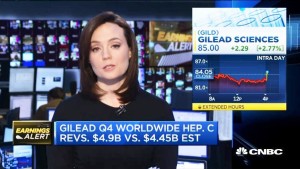
- Gilead shares pop 2% on earnings beat (video.cnbc.com)
Gilead Sciences Q4 EPS beat analysts' expectations, reports CNBC's Meg Tirrell. (video)
- Pharmacies Won’t Compound Lethal Injection for Alabama Executions (pharmacytimes.com)
Alabama recently executed its first inmate using a 3-drug cocktail containing midazolam in place of the more commonly used pentobarbital...This is because compounding pharmacies in Alabama were not willing to make pentobarbital for lethal injection...Four other states’ pharmacies also declined to compound pentobarbital for the Alabama Department of Corrections...Both the International Academy of Compounding Pharmacists and the American Pharmacists Association have issued statements discouraging pharmacists from participation in executions...IACP said compounding lethal injection drugs goes against pharmacists’ focus on healing and care. It also pointed out that state boards of pharmacy could potentially seek ramifications against a pharmacy that knowingly prepares a drug that will cause harm...With manufacturers and compounding pharmacies’ resistance, state departments of corrections may have to turn to alternative methods for executions...in April 2015, Oklahoma approved the use of nitrogen hypoxia in a gas chamber if the state could not obtain lethal injection drugs...In Utah, Governor Gary Herbert signed a law in March 2015 that allows for the use of a firing squad as a plan B.
- Alibaba health initiative takes it on the chin with CFDA move, lawsuit (fiercepharmaasia.com)
Chinese Internet giant Alibaba suffered two blows this past week: the China Food and Drug Administration announced that it will no longer work with the company on an initiative to track medicines to ensure their authenticity, and a pharmacy chain has filed a lawsuit against the company saying the initiative gave it an unfair advantage over other drug sellers...CFDA Vice Director Sun Xianze held a confab with 20 pharmacy and drug companies and wholesalers on Jan. 27 at which the decision was announced to curtail the deal with Ali Health, a subsidiary of Alibaba...The original plan with Alibaba had its detractors early on who complained about the costs involved and said the plan would do little to stop fake drugs from getting into the hands of unsuspecting patients...CFDA officials said they would continue to push for the bar code tracking system, but would instead work with a nonprofit institution and not a commercial entity...Meanwhile, Alibaba found itself in the middle of a lawsuit filed...by Yangtianhe Pharmacy...The suit says the CFDA broke the law by partnering with a commercial entity to create the system, which is called the Product Identification, Authentication and Tracking System.
- Gilead accused of manipulating HIV patents (statnews.com)
In the latest legal challenge to Gilead Sciences and its register-ringing strategy, the AIDS Healthcare Foundation has filed a lawsuit accusing the drug maker of manipulating the patent system in order to thwart competition to its HIV medicines. And the nonprofit alleged that countless HIV patients have been prevented from accessing treatment — and may have been harmed — as a result...At issue is tenofovir, or TDF, which is a cornerstone of the combination HIV treatments that Gilead sells. The patent on the TDF compound expires in December 2017 and Gilead hopes to replace it with a modified version known as TAF (Genvoya)....TAF is more potent and causes fewer side effects, notably bone damage and kidney toxicity. The foundation argued that Gilead knew of these differences thanks to animal testing dating back to 2001, but that the company delayed testing TAF in humans until 2011. In doing so, Gilead extended patent protection by a few years, the foundation charged in the lawsuit...the foundation argued that TAF is an obvious modification of TDF and, therefore, does not deserve patent protection... The nonprofit....seeks to have the TAF patents invalidated...Gilead’s refusal to make TAF available as a stand-alone drug appears to be a calculated, anticompetitive maneuver aimed at keeping competing TAF drugs off the market for years despite the weakness of Gilead’s patents covering TAF, the lawsuit claimed. The failure to make TAF available as a stand-alone drug highlights Gilead’s motive of avoiding competition at all costs...Gilead "reevaluated TAF as part of its ongoing R&D review process" and "decided to prioritize" TAF development...the company believes its patents are valid.