- United States Lags Behind Other Nations in Rx-to-OTC Switch Approvals (klinegroup.com)
In contrast to the U.S. FDA, regulatory agencies outside the United States have been much more aggressive in approving switches for conditions outside the typical OTC paradigm…increasing patient choice by making more medicines available without a prescription is a priority for the British government…The United Kingdom leads the world in the range of products available without a prescription…Industry research conducted by Kline for its…suggests that switch receptivity in the United States may be changing. There are a wide variety of categories with switch candidates that may have significant impact on many companies across the OTC and Rx markets.
- Four Fun Facts About the Walgreens-Rite Aid Merger Agreement (drugchannels.net)Sec. and Exch. Com. Form 8-K AGREEMENT AND PLAN OF MERGER Among WALGREENS BOOTS ALLIANCE, INC., RITE AID CORPORATION (files.shareholder.com)
Late last week, Walgreens Boots Alliance filed an 8-K with the full text of its merger agreement with Rite Aid….The document containing the Agreement and Plan of Merger weighs in at a hefty 137 pages. Below, I highlight four fun facts about the deal’s timing, its termination fees, and what the companies will do to achieve antitrust approval.
- The deal must be completed before Halloween 2016.
- If Rite Aid backs out of the deal, then it owes as much as $370 million to Walgreens Boots Alliance.
- WBA could owe Rite Aid a termination fee as large as $650 million.
- To get the deal approved, WBA is willing to dump as many as 1,000 stores.
- Pfizer partners with South Africa to produce pneumococcal vaccine (reuters.com)
Pfizer has partnered with South Africa's Biovac Institute to produce a potentially life-saving pneumonia vaccine for infants…The five-year partnership…will see technology transfer and skills upgraded for the production of 'Prevenar 13' vaccine on a sustainable basis…There is more that we can do to cut the costs of the vaccine, and that is to manufacture the vaccine here in Cape Town…vaccine prices have sky-rocketed over the last decade...
- Eli Lilly to build $70M R&D building in Indianapolis (ibj.com)
Eli Lilly Co…announced plans to add a 130,000-square-foot building to its Indianapolis research-and-development headquarters…The $70 million investment is the latest in a string of moves by the Indianapolis-based drugmaker to bulk up its hometown presence…"We are excited to grow our presence in Indianapolis, which is home to our largest global R&D facility and where our research efforts began,"…Lilly employs 4,400 R&D workers in Indianapolis, the biggest chunk of its 7,000 R&D employees worldwide…Lilly has applied for a real-property tax abatement on the proposed expansion that will save it $6.6 million over a 10-year abatement period…Lilly will still pay about $6.7 million in property taxes during the abatement period on the increased value of the property…This is Lilly's fourth R&D expansion this year. In May…it would build a delivery and device innovation center in Cambridge…In July…announced an expansion of its biotechnology center in San Diego…In October…an expansion of its presence at the Alexandria Center for Life Science in New York City.
- Congress Looks to Halt Rise of Generic Drug Prices with New Rebates (raps.org)
As drug price increases continue to make waves on the national level, Congress is taking the matter into its own hands by requiring generic drugmakers under the Medicaid Drug Rebate Program to pay higher rebates if generic prices rise too quickly…The new penalty for such price increases on generics, which is included in the recently passed budget deal, basically amounts to additional rebates that generic drug manufacturers would have to pay if the price of a generic for a given quarter outpaces the inflation-adjusted baseline Average Manufacturer Price…Under the Bipartisan Budget Act of 2015, the additional rebate...would now be applied to generics, too, beginning in Q1 of 2017...the change could create issues for generic manufacturers around updating their Medicaid price reporting systems, figuring out the potential financial impact of the change, and posing new strategic pricing questions for generic drug launches and market withdrawals.
- Many ‘me-too’ drugs didn’t start off that way: report (pharmalot.com)Nearly All Later Entrants to Drug Classes Were in Clinical Testing or Regulatory Review Before First-in-Class Approval (csdd.tufts.edu)
For many years, drug makers were criticized for racing to market with so-called me-too medicines. These are drugs that arrive in the marketplace after the first in a particular class of medicines is approved for treating a certain malady…Instead of developing something that offers little, if any, improvement over a first-in-class drug…critics have contended that drug makers should use precious R&D resources to focus on developing therapies for ailments that are lacking treatments…Now...a new analysis suggests the me-too phenomenon often occurred because companies were simply engaging in concurrent drug development, rather than opportunistic efforts to ride on the coattails of a successful medicine.
- Prescription drug use has risen in the U.S. (reuters.com)Trends in Prescription Drug Use Among Adults in the United States From 1999-2012 (jama.jamanetwork.com)
Prescription drug users rose from 51 percent of U.S. adults in 1999 to 59 percent of adults in 2011, according to a new study…It’s hard to say why prescription drug use would be on the rise…we know that older adults tend to take more medications than younger adults, and so we’d expect prescription drug use to increase as the U.S. population ages…something beyond the aging…appears to be driving the increase in prescription drug use…New drugs enter the market and old drugs lose patent protection and become less costly...patterns of prescription drug use evolve with scientific advances and with changes in clinical guidelines and policies regarding drug marketing and promotion.
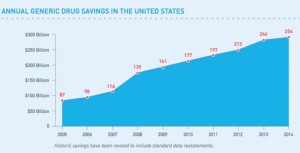
- Record $254 billion in health savings attributed to use of generics in 2014 (drugstorenews.com)Generic Drug Saving in the U.S. Seventh Annual Edition: 2015 (gphaonline.org)
Few factors have played as significant a role in helping curb health care costs over the past decade as generic drugs…since 2005, generics have saved patients $1.68 trillion. In 2014 alone, the report notes, generic drugs trimmed more than $254 billion from health care spending. Slightly more than a third of those savings were from generics taken by people over 65…“This new report reinforces that generic drugs are a critical part of any solution to rising costs for patients, payers and for the entire health care system,”
- Blog: Texas docs plead for relief from ‘meaningless abuse’ (modernhealthcare.com)EHR State of Mind - ZDoggMD (letdoctorsbedoctors.com)
Texas Medical Association wants Congress to intervene and make changes to the federal electronic health-record incentive payment program it's calling "meaningless abuse."…The group says Stage 3 of the program meant to get physicians using EHRs could jeopardize Medicare doc payment rules… wants Congress to lift what it's describing as the $31.6 billion program's “convoluted and tedious” meaningful-use requirements…TMA President Dr. Tom Garcia asked legislators to co-sponsor two bills to alter the meaningful-use landscape…One is the Flex-IT 2 Act...which would delay Stage 3 meaningful-use rules until at least Jan. 1, 2017. The other is the Transparent Ratings on Usability and Security to Transform Information Technology, or TRUST IT Act which is aimed at ensuring health IT systems perform better in the field.
- Valeant sends letter to doctors, seeks to reassure over pharmacy ties (reuters.com)Pharmacist at center of Valeant scandal accuses drugmaker of 'massive fraud' (latimes.com)Charlie Munger Isn't Done Bashing Valeant (bloomberg.com)
Valeant Pharmaceuticals International Inc sought to reassure doctors…that the company's decision to cut ties to a controversial specialty pharmacy would not disrupt doctors' ability to prescribe the company's drugs to patients…In a letter to healthcare professionals…Valeant would pay for the cost of its products through Nov. 8 and make sure patients could fill their prescriptions with no out-of-pocket expenses, wherever possible. Patients on government-run health plans such as Medicare are not eligible…Philidor Rx Services, will file no further insurance claims.