- Drugmakers open new year with price increases on dozens of medicines (biopharmadive.com)
Several major drugmakers, including Allergan, Bristol-Myers Squibb and Eli Lilly, increased prices on a number of their drugs Jan. 1, holding with typical industry practice despite continuing scrutiny of rising pharmaceutical costs...While raising prices in January has become the norm for pharma, this year has become a test of whether recent criticism by lawmakers and the Trump administration would spur companies to hold off on increases. Data cited by Raymond James suggests this could be playing out somewhat: Drugmakers overall have instituted fewer price hikes from Dec. 1 through Jan. 1 than the same period last year, according to a recent note from the investment bank...
- FDA panel backs prescribing opioid overdose reversal drug along with painkillers (reuters.com)
An advisory panel to the Food and Drug Administration....recommended prescribing the opioid overdose reversal drug, naloxone, along with addictive painkillers...The recommendation of the panel underscores concerns about the growing opioid overdose epidemic...FDA studies found that co-prescribing naloxone to all patients who are prescribed painkillers could increase annual healthcare costs by $63.9 billion to $580.8 billion...I think co-prescribing is an expensive way to saturate the population with naloxone. The at-risk population is not necessarily the ones that are being prescribed new narcotics...
- Four Democrats Introduce Senate Bill to Allow HHS to Block “Excessive” Drug Price Increases (centerforbiosimilars.com)S.3754 - A bill to prohibit price gouging in the sale of drugs. (congress.gov)
...Senators Richard Blumenthal, D-Connecticut, Kamala Harris, D-California, Amy Klobuchar, D-Minnesota, and Jeff Merkley, D-Oregon, introduced a bill on the Senate floor, S. 3754, that would allow HHS to block drug price increases that it deems as “excessive.”...The bill, dubbed “A bill to prohibit price gouging in the sale of drugs,” is just the latest in a series of actions that Congressmen and the government alike have taken to address high drug prices.
- Drug Maker Pays $360 Million to Settle Investigation Into Charity Kickbacks (nytimes.com)
The drug maker Actelion Pharmaceuticals has agreed to a $360 million settlement stemming from an investigation into whether the company illegally funneled kickbacks through a patient-assistance charity...Actelion...makes expensive drugs to treat a rare lung condition, is the latest pharmaceutical company to settle federal inquiries into their ties to patient-assistance groups, including whether companies have used the patient programs to increase the price of their drugs...But federal anti-kickback laws prohibit companies from giving such financial assistance to Medicare and Medicaid beneficiaries because doing so is considered an inducement to buy their drugs. For years, drug makers have skirted those laws by instead donating to nonprofit charities, which then give the money to Medicare patients. Such arrangements are legal as long as there is no direct coordination between the pharmaceutical company and the nonprofit organization...Federal prosecutors said Actelion violated the law by collecting detailed data in 2014 and 2015 about the patients receiving help from a nonprofit, the Caring Voice Coalition, and using the data to budget for future donations. As a result, Actelion ensured that the money it donated would be used only to assist patients who used its drugs, and not competing companies’ treatments for the pulmonary condition.
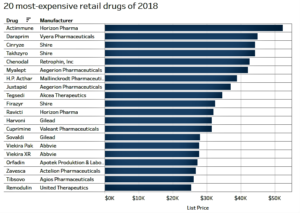
- The 20 most expensive pharmacy drugs in 2018, featuring names big and small (fiercepharma.com)
More and more, specialty drugs carrying eye-popping price tags are winning FDA approvals. While more are certainly on the way—look at Novartis' recent presentation that a gene therapy could be worth millions per patient—analysts at drug pricing website GoodRx have tallied up the most expensive pharmacy-dispensed drugs in the U.S. per month as of November...Some are marketed by small pharma companies such as Vyera Pharmaceuticals, while top biotechs and pharmaceutical companies market others. Some have seen controversy, while others are lesser known. Rare disease meds make up much of the list, while some treatments such as those for hepatitis C treat diseases that affect millions of people...
- Congressional report: Drug companies, DEA, failed to stop flow of millions of opioid pills (washingtonpost.com)Committee Report Details Alleged Opioid-Dumping in West Virginia (energycommerce.house.gov)Red Flags and War ning Signs Ignored: Opioid Distribution and Enforcement Concerns in West Virginia (energycommerce.house.gov)
A report from the majority staff of the House Energy and Commerce Committee found that distributors, which fulfill orders for prescription drugs to pharmacies, failed to conduct proper oversight of their customers by not questioning suspicious activity and not properly monitoring the quantity of painkillers that were being shipped to individual pharmacies...The committee also found that the DEA did not properly use a database that aims to monitor the flow of powerful prescription painkillers from manufacturers to sellers, something that could have allowed federal agents — in real time —...The agency also curtailed enforcement of distributors...and infighting inside the agency affected the way cases were handled...
- Is China the next great hope for the pharma industry? (pharmaceutical-technology.com)
Many top pharma firms have been forced to drop prices to get a slice of the Chinese market, but that hasn’t dampened spirits one bit – on the contrary, CEOs from companies like AstraZeneca and Pfizer have been celebrating dramatic Chinese sales increases in 2018. So how is China’s pharmaceutical market growing and is it really a clean bill of health for any internationals lining up to profit?...In pharmaceuticals specifically, China is comfortably the second-largest national market in the world, with healthcare analytics firm Iqvia putting its value at $122.6bn in 2017 and projecting growth to as much as $175bn by 2022...Multinational pharma firms are starting to make China a major priority for drug sales, as the potential for profit is now widely seen as a decisive counterbalance against the risks of operating under the Chinese regime...So what does China mean to the international pharma industry, and what opportunities and challenges are drug manufacturers encountering as they attempt to crack this lucrative market?
- Demographic changes bring opportunities
- Foreign firms are making big bets on China
- Regulatory reform opens China to foreign innovators
- Chinese competition and policy uncertainty remain a challenge
- Walgreens Boots Alliance’s plans $1 billion in cuts, stock drops (cnbc.com)
Walgreens Boots Alliance announced plans...to its cut annual costs by $1 billion within three years and reported first-quarter results that beat Wall Street’s estimates...The results also showed sales are struggling in Britain, one of its largest markets...Walgreens shares are down more than 2 percent this year bringing its market value to $69.3 billion. The stock has been under pressure as investors worry about the impact Amazon will have as it expands into the pharmacy business. To prepare for increased competition, Walgreens has announced partnerships with Kroger, Alphabet’s Verily and others...
- The Top 5 Biosimilars Articles for the Week of December 10 (centerforbiosimilars.com)
Samantha DiGrande for The Center for Biosimilars®, your resource for clinical, regulatory, business, and policy news in the rapidly changing world of biosimilars.
- Pat-INFORMED: the solution to drug patent procurement issues? (pharmaceutical-technology.com)
Many countries do not have the equivalent to the US’s Orange book, meaning that health officials are wasting time establishing whether patents have been taken out on medicines. Leading biopharmaceutical companies have worked together to develop Pat-INFORMED, a new global platform that could allow government procurement agencies to access information on thousands of individual patents anywhere in the world...Although information on patents is already in the public domain...its technical nature, or the fact that it is presented differently according to national varying requirements, prevents people from understanding what they are about...pharmaceutical companies have been thinking about how to overcome this challenge; many have been disclosing patent information for decades, but there has been no consistent approach for doing so. By way of a solution, the recently established Patent Information Initiative for Medicines (Pat-INFORMED) aims to make it easier for international drug procurement agencies to access a library of clear and understandable patent information...the main purpose of Pat-INFORMED is to help government health agencies and NGOs responsible for procuring medicines to assess the patent status of medicines, as it discloses the patents relating to the active ingredient. The platform includes a channel for procurement agencies to get in touch with the patent holders, so they can speak directly to experts in the relevant company...