- India’s Cipla gets U.S. beachhead with $550M deal for 2 generics operations (fiercepharma.com)
India's Cipla made its name in the west as a leader in challenging Big Pharma patents on expensive drugs in an effort to cut their prices in a country with a large need and little money. Cipla said it will pay $550 million for InvaGen Pharmaceuticals, a Hauppauge, NY-based generics maker with 32 products in the market and 30 in the pipeline, as well as Exelan Pharmaceuticals, a Lawrenceville, GA, operation that markets and sells InvaGen's products into the government and institutional markets.
- Outsourcing Facilities of America Works with Search Engines and Pay-Per-Click Advertising Sources to Eliminate Ads by Non-Compliant Compounding Pharmacies (prweb.com)
Pay-Per-Click advertising sites, primarily search engines, are largely unaware of the new regulations governing 503B Outsourcing Facilities. Pay-per-click advertising has been one of the main ways non-compliant compounding pharmacies have been able to sell their products and services to hospitals, physicians and patients. Outsourcing Facilities of America is working to educate PPC advertising companies on the regulatory requirements… create new policies and procedures to stop non-compliant compounding pharmacies from selling their products in violation of these regulations.
- Biosimilars Council chief answers 6 key questions about biosims in the US (biopharmadive.com)
At the beginning of the year, there was a great deal of anticipation about when the first biosimilar would be approved in the U.S…And then it happened…On March 6, the FDA has approved Novartis/Sandoz's Zarxio (filgrastim-sndz), a biosimilar of Amgen's Neupogen…And as of Thursday, September 3, Zarxio officially hit the U.S. market at a 15% discount to Neupogen… to gain clarification around several key issues concerning biosimilars, we spoke with Dr. Bert Liang, Chair of the Biosimilars Council…
- Last month, you responded to the decision from the Federal Circuit Court of Appeals that recognized the ‘patent dance’ as optional by applauding the decision. Can you explain your reaction?
- When would it be beneficial for a biosimilar company to provide a complete copy of its biosimilar application to the reference manufacturer upon filing?
- You have indicated that the proposed biosimilar naming convention put forth by the FDA in which originator products and biosimilars share the same core name and have different suffixes. Please tell us why you disagree with that.
- What type of approach to naming would work in your opinion?
- How do you see the public hearing hosted by the FDA playing out? Who are your allies in this issue?
- Can the U.S. look to Europe as a model for the evolving role of biosimilars?
- The High Cost of Delays in Biosimilars Hitting the US Market (specialtypharmacytimes.com)
“Biosimilars will do for the biotech market, primarily made up of very expensive injectable drugs, what generic drugs did for traditional oral solid pills a decade ago: lower the cost for safe, effective treatments that improve and save lives,”… “They will revolutionize the category of spending that is among the fastest-growing and most worrisome for payers who want to continue providing sustainable, high-quality benefits for their members.”
- China Resilient: New Pharma Survey Offers Highly Positive Outlook (pharmexec.com)
..survey of the Chinese pharmaceutical market based on an opinion poll conducted with domestic and foreign suppliers attending CPhI’s annual China expo in Shanghai…Despite some structural concerns in the context of slowing macro-economic growth overall, the report offers a very positive outlook…China is the world’s largest producer of active pharmaceutical ingredients. Its massive pharmaceutical market is projected to be worth $158 billion worldwide in 2016, while domestic sales alone will account for $63 billion…China sales will double over the next five years.
- Undeterred by PTO setbacks, Bass slaps new challenges on Acorda patents (fiercepharma.com)
Hedge-fund manager Kyle Bass may have hit some recent speed bumps in his quest to challenge pharma patents through a U.S. Patent and Trademark Office review system. But he's not giving up… not to be deterred, refiled the two Ampyra challenges…with more documentation, and he filed two new challenges against the drug's other IP shields, too… Bass has been using the inter partes review system to wage war on patents from companies such as Celgene, Shire, Biogen and Acorda that he thinks aren't up to snuff,...Bass and his partners want "to line their own pockets at the expense of public pharmaceutical companies and their shareholders,"…
- CVS says its ban on cigarette sales has reduced smoking (reuters.com)
CVS Health Corp said..its decision to stop selling tobacco products last year led to a 1 percent decrease in cigarette sales...The September 2014 decision hurt sales, with general merchandise revenue at CVS pharmacies open at least a year falling 7.8 percent in the second quarter from a year earlier,.., CVS said it benefited in others ways…nicotine patch purchases rose 4 percent…visits to its retail clinics for smoking cessation counseling nearly doubled.
- FDA Grants Malignant Mesothelioma Treatment Orphan Drug Designation (specialtypharmacytimes.com)
Biologic therapy targets genetic defects found in various cancers… FDA granted Orphan Drug Designation to MTG-201 (MTG Biotherapeutics Inc.) for the treatment malignant mesothelioma…drug targets the Dickkopf-3 genetic defect found in various cancers…There is a…need for new treatment options for…mesothelioma…one of the most aggressive and poorly treated cancers. MTG-201 represents a…novel approach to treating this cancer by selectively inducing apoptosis and inducing an immunologic response against the cancer..
- Moody’s upgrades financial outlook for biopharma industry (biopharmadive.com)
Citing the success of product launches, the ratings firm Moody's has raised the outlook for the pharmaceutical sector from "neutral" to "positive."...Projected annual EBIDTA for the global biopharma industry is 4% to 5% over the next 12 to 18 months…One of the industry's biggest areas of growth is oncology, driven largely by the emergence of immuno-oncologic drugs, such as Bristol-Myers Squibb's Opdivo and Merck's Keytruda.
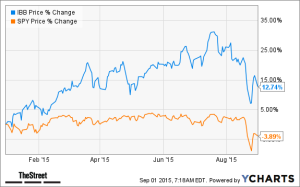
- The Numbers Behind Biotech’s Horrible, No Good, Awful August (thestreet.com)
August was a horrible month for biotech stocks. The breadth of the selling during the month was impressive. Among all U.S.-listed biotech stocks regardless of market cap, only 68 companies managed to end August with higher stock prices, while 267 biotech stocks lost ground,… If you want to be an optimist, note the IBB ETF is still outperforming the broader market for the year.