- Better insurance coverage of non-drug therapies might help ease opioid crisis (reuters.com)Coverage of Nonpharmacologic Treatments for Low Back Pain Among US Public and Private Insurers (jamanetwork.com)
When it comes to non-drug therapies for back pain, U.S. insurance plans vary widely in what they will cover, a new study finds...Private and public insurers are missing important opportunities to promote alternatives to opioids...insurers often provide little or no coverage for evidence-backed interventions for chronic pain such as acupuncture and psychological counseling...“Insurers can be part of the problem or part of the solution,” said study coauthor Dr. Caleb Alexander...at the Center for Drug Safety and Effectiveness...“All too often doctors reach for the quick solution, prescription drugs, especially opioids, to manage pain that would be more effectively and safely treated with non-pharmacological approaches,” Alexander said. “This is a system that is designed with, and fosters, the idea that there is a pill for every ill...
- New Law To Force Payments Disclosure by Drug and Device Manufacturers to Non-Physician Providers (ptcommunity.com)
Sunshine Law Will be Expanded to Include Nurse Practitioners, Physician Assistants, Nurse-Midwives, and Others...Congress is expected to finalize within days a new law to expand transparency surrounding payment to non-physician providers. Beginning in 2020, pharmaceutical and medical device companies must disclose payments made to those providers for speaking or consulting, as they must do now with physicians...In many states, nurse practitioners, physician assistants, and other non-physician health professionals account for a substantial number of prescriptions—often including prescription opioids.” The law also applies to clinical nurse specialists, nurse-anesthetists, and nurse-midwives...Without full transparency, pharmaceutical companies can operate under the cover of darkness, possibly using gifts and payments to influence the prescribing practices of medical professionals like nurse practitioners and physician assistants...
- Brazilian court strips Gilead of its patent for Hepatitis C drug Sovaldi (pharmaceutical-technology.com)
A judge from the 21st Federalist Court of Brasilia has ruled to invalidate US biopharma company Gilead’s exclusivity patent for Sovaldi (sofosbuvir)...Hepatitis C is a major public health issue in Brazil; it has been estimated by Brazilian academics that 1.5 million people in the country were chronically infected with the virus in 2014...A centre-left, environmentalist presidential candidate for the upcoming election, Marina Silva, filed a popular action analysis complaint to the court arguing that Gilead’s patent for Sovaldi should be overturned in order to reduce costs for patients...The patent was granted in 2015 by the National Institute of Industrial Property...Silva and her running mate Eduardo Jorge claim the INPI made the decision without the consent of the National Agency of Sanitary Surveillance, meaning Brazil’s national interest was not adequately recognised in the decision...
- AbbVie’s not the only one facing kickbacks scrutiny. Add Biogen, Sanofi, Gilead and more (fiercepharma.com)
At first glance, it seems like a logical idea: pharma companies teaching doctors' staff how to handle patients using their drugs and helping staffers with reimbursement questions. But a slew of companies are discovering that it's not so simple...In fact, it might just be against the law...Biogen, Sanofi and Gilead Sciences are under investigation by federal and state authorities for offering reimbursement services and clinical education programs, the companies have disclosed in securities filings. Additionally, Bayer, Amgen and Eli Lilly face whistleblower lawsuits over the issue...In Sanofi’s case, the U.S. Attorney’s Office...sent the company a civil investigative deman...“requesting documents and information relating to Sanofi US’s certified diabetes educator program during the period from 2007 to the present.”...Gilead....received a voluntary request from the U.S. Attorney’s Office...seeking information “related to our reimbursement support offerings, clinical education programs and interactions with specialty pharmacies for Sovaldi and Harvoni...
- GSK resumes some doctor payments, backtracking on blanket ban
GlaxoSmithKline, which five years ago stopped paying doctors for promoting its drugs, said...it would allow such payments once again in limited circumstances...The British group’s 2013 no-payment pledge marked a first for an industry battling past scandals over sales practices. But other drugmakers failed to follow suit, leaving it at a competitive disadvantage...Drugmakers have long used so-called key opinion leaders to promote the benefits of their products to other prescribing physicians and the decision to abandon this strategy was questioned by a number of analysts...GSK’s new updated policy...will now permit payments to global experts who speak about the science behind novel new medicines...“These changes are being made for a select number of innovative products in a limited number of countries and apply to restricted time periods in a product’s lifecycle,” the drugmaker said.
- AmerisourceBergen to pay $625 million in U.S. civil fraud settlement (reuters.com)
AmerisourceBergen Corp...will pay $625 million to resolve civil fraud charges over the sale of syringes containing drugs for cancer patients, double billing, and providing kickbacks to doctors...The settlement...boosts AmerisourceBergen’s total payout to $885 million over its repackaging and distribution of pre-filled syringes that were not approved by the Food and Drug Administration...AmerisourceBergen admitted that from January 2001 to January 2014, its Medical Initiatives Inc pharmacy unit in Alabama shipped millions of syringes for patients undergoing chemotherapy that contained drugs prepared in an unsterile environment...Authorities said AmerisourceBergen would harvest “overfill” from the original vials of such drugs as Aloxi, Anzemet, Kytril and generic Kytril, Neupogen and Procrit...That enabled the company to create more doses than it bought, and generate at least $99.6 million of extra profit...
- Scientists behind game-changing cancer immunotherapies win Nobel medicine prize (reuters.com)2018 Nobel Prize in Medicine Awarded to 2 Cancer Immunotherapy Researchers (nytimes.com)
American James Allison and Japanese Tasuku Honjo won the 2018 Nobel Prize for Physiology or Medicine on Monday for game-changing discoveries about how to harness and manipulate the immune system to fight cancer...The seminal discoveries by the two Laureates constitute a landmark in our fight against cancer...Allison...worked on a protein known as CTLA-4 and realized that if this could be blocked, a brake would be released, unleashing immune cells to attack tumors...Honjo...separately discovered a second protein called PD-1 and found that it too acted as an immune system brake, but with a different mechanism...The discoveries led to the creation of a multibillion-dollar market for new cancer medicines. In particular, drugs targeting PD-1 blockade have proved a big commercial hit, offering new options for patients with melanoma, lung and bladder cancers...
- Banner Health, Ascension executives join hundreds of hospital CEOs defending 340B program (fiercehealthcare.com)
CEOs from more than 700 hospitals sent a letter to congressional leaders...calling on them to protect the 340B drug discount program, among them Banner Desert Medical Center...The 340B program requires drugmakers to provide discounted drugs to hospitals, health centers, and other healthcare providers that serve disproportionate numbers of low-income and rural patients...But the program has been under increased scrutiny after enjoying years of bipartisan popularity, including three expansions in Congress. Critics argue that hospitals are taking advantage of the discounts to line their wallets and are snapping up oncology providers to boost the number of pricey specialty drugs they can prescribe under the program...the letter said the group is concerned cuts to the 340B program would increase the prices of need and existing drugs...In our larger disproportionate share hospitals, the program enables us to provide a wider range of comprehensive services to people who are uninsured and traditionally underserved—services that would need to be scaled back along with any 340B reductions...For our more rural critical access hospitals, cuts to 340B would threaten the only places for people in the communities to get their care in the first place...
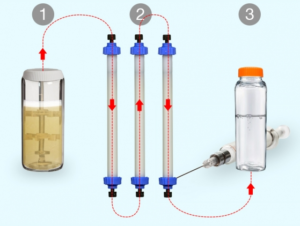
- A new way to manufacture small batches of biopharmaceuticals on demand (news.mit.edu)
Biopharmaceuticals, a class of drugs comprising proteins such as antibodies and hormones, represent a fast-growing sector of the pharmaceutical industry. They’re increasingly important for “precision medicine” — drugs tailored toward the genetic or molecular profiles of particular groups of patients...Such drugs are normally manufactured at large facilities dedicated to a single product, using processes that are difficult to reconfigure. This rigidity means that manufacturers tend to focus on drugs needed by many patients, while drugs that could help smaller populations of patients may not be made...To help make more of these drugs available, MIT researchers have developed a new way to rapidly manufacture biopharmaceuticals on demand. Their system can be easily reconfigured to produce different drugs, enabling flexible switching between products as they are needed...Traditional biomanufacturing relies on unique processes for each new molecule that is produced...We’ve demonstrated a single hardware configuration that can produce different recombinant proteins in a fully automated, hands-free manner...
- Novartis, axing 19% of its workforce, faces big challenges with thinned ranks (fiercepharma.com)
....CEO Vas Narasimhan’s plan to reduce Novartis’ overall workforce by at least 19%, from 124,000 today to under 100,000 by 2022, he told reporters...Most of those cuts will come from manufacturing and the company’s 2019 spinoff of its Alcon eye unit. But a restructuring of business services will result in an additional 700 jobs lost, the company said...Novartis plans to move some management operations from its Basel headquarters to the five service centers in India, Malaysia, Mexico, the Czech Republic and Ireland...The primary drivers for this intention are the benefits that standardization, simplification and a sound global services location strategy bring over time...Novartis stands out for the sheer size and scope of its downsizing plan—and for the challenges the company will still have to address with a rapidly shrinking workforce. Novartis is grappling with marketing and manufacturing issues surrounding Kymriah, for example, and it called off planned price hikes in the face of increasing scorn from Washington on Big Pharma pricing practices. Novartis is also facing pricing pressure on key products, including psoriasis drug Cosentyx and its stable of generic drugs produced by its Sandoz division, which is facing a sales decline in the U.S. even as its volume continues to grow.