- FDA confronts its limits in push on drug pricing (biopharmadive.com)
Legally, the agency has few levers to pull when it comes to addressing prices. But that hasn't stopped Commissioner Scott Gottlieb from taking a more spirited and vocal approach than past FDA chiefs. The effort comes as eye-popping price tags, some spurred by groundbreaking new treatments but others by manipulation of the drug pricing system, have prompted public and political opposition to rising drug costs...Gottlieb has aggressively used public statements to call out drugmakers for blocking or impeding market competition. And, under his leadership, the FDA has continued to ramp up approval of generic and biosimilar drugs, while pushing the boundaries of regulatory flexibility.
- Indirect effect..."Where the FDA does have authority to get involved is in how much, in what way, and how aggressively it approves drugs, or their generic or biosimilar competitors."...While it's an indirect effect, approving low-cost competitors more quickly can be a powerful force for lowering prices.
- Biosimilars showcase hurdles facing FDA...speeding competitors to market can only go so far if other forces block lower-cost rivals from reaching patients. To date, that's exactly what's played out with biosimilars...Gottlieb has expressed frustration that the FDA's steady increase in approving biosimilars hasn't resulted in greater savings. "Competition is, for the most part, anemic,"
- Naming and shaming...Gottlieb has made full use of his public platform to call out market "shenanigans" by drugmakers...Gottlieb received a lot of media coverage by attacking such practices as "regulatory gaming," and has since kept up pressure on drugmakers through public statements.
- Worth cooperating?...As Health and Human Services Secretary Alex Azar and Gottlieb continue to criticize pricing and anticompetitive practices, drugmakers are faced with a choice: ignore the growing chorus or cooperate.
- U.S. appeals court says GSK cannot be sued over generic drug suicide (reuters.com)
U.S. appeals court...tossed a $3 million verdict against GlaxoSmithKline over the suicide of an attorney who took a generic version of the company’s antidepressant Paxil, finding the company could not be held liable for injuries allegedly caused by a generic copy...The case weighing whether brand-name manufacturers can be sued for injuries blamed on generic drug versions was closely watched within the pharmaceutical and legal industries...Under a 2011 ruling by the U.S. Supreme Court, generic drug companies cannot be sued for failing to provide adequate label warnings about potential side effects because federal law requires them to use the brand-name versions’ labels...decision follows a series of state court rulings in similar cases. The top courts of Massachusetts and California ruled brand-name manufacturers could be sued by generic drug users...West Virginia’s Supreme Court...rejected liability claims against brand-name manufacturers for alleged failures to warn over a generic company’s drug.
- FDA approves first generic of Mylan’s EpiPen (pharmaceutical-technology.com)
The Food and Drug Administration has approved the first generic version of the EpiPen and EpiPen Jr auto-injector for severe allergic attacks, including life-threatening anaphylaxis....The EpiPen was approved in 2007 and is marketed by Mylan. However, since then both the company and its product have been involved in a number of scandals. Notably the price of the EpiPen has increased from $57 in 2007 to approximately $600 in 2016 and in May, the FDA had to place Mylan’s EpiPen on its official shortages list as a result of manufacturing delays...The newly approved generic epinephrine auto-injector will be available in 0.3mg and 0.15mg strengths and is marketed by Teva. This the company’s second attempt to get its generic approved by the FDA; it was rejected in 2016...FDA commissioner Scott Gottlieb said: “Today’s approval of the first generic version of the most-widely prescribed epinephrine auto-injector in the US is part of our longstanding commitment to advance access to lower cost, safe and effective generic alternatives once patents and other exclusivities no longer prevent approval.
- FDA Approves First-of-its-Kind RNA Therapy (biopharminternational.com)
The new drug, Onpattro (patisiran), by Alnylam Pharmaceuticals, is in a new class of drugs called small interfering ribonucleic acid (siRNA) treatment...a first-of-its-kind RNA-based therapy for treating peripheral nerve disease (polyneuropathy) caused by hereditary transthyretin-mediated amyloidosis (hATTR) in adult patients...Polyneuropathy caused by hATTR is a rare, debilitating, and often fatal genetic disease characterized by the buildup of abnormal amyloid protein in peripheral nerves, the heart, and other organs...“This approval is part of a broader wave of advances that allows us to treat disease by actually targeting the root cause, enabling us to arrest or reverse a condition, rather than only being able to slow its progression or treat its symptoms...“New technologies like RNA inhibitors that alter the genetic drivers of a disease, have the potential to transform medicine, so we can better confront and even cure debilitating illnesses.
- Lawmakers push to require price disclosure in DTC drug ads (biopharmadive.com)
The Senate...passed a measure that would provide the Department of Health and Human Services with $1 million to implement proposed rules requiring drugmakers disclose the price of their prescription drugs in direct-to-consumer advertising...Approved as part of a larger spending bill, the bipartisan amendment gives legislative backing for an idea that the Trump administration has pushed as part of its plan to combat rising drug prices...touted the measure as a common-sense step toward greater transparency, it's a proposal likely to generate substantial debate. Drug prices are notoriously opaque, and most patients don't pay the so-called "list price" that such a requirement would likely rely on..."We have FDA and CMS working to examine how to require drug companies to post their list prices in direct-to-consumer advertising. When patients hear about a wonderful new drug, they should know whether it costs $100 or $50,000," said HHS Secretary Alex Azar...The administration's proposal, unsurprisingly, has been met with resistance from drugmakers..."Disclosing list prices in DTC ads would not benefit patients as they are often not the prices insurers pay and are generally not a good indicator of what patients will pay at the pharmacy counter," wrote a spokesperson for PhRMA...
- CVS adopts ICER metrics in shift to value-based drug pricing (biopharmadive.com)
CVS Caremark will use the value-based drug pricing system of the Institute for Clinical and Economic Review to exclude some high-cost drugs from plans...The pharmacy benefit manager will incorporate ICER's quality-adjusted life years measure. If a drug is priced greater than $100,000 per QALY at launch, CVS Caremark will allow its clients to exclude the drug from their plan...CVS is hoping the decision will give PBMs more power in pressuring drug prices lower, especially by targeting their launch prices...PhRMA...supports using "rigorous, objective evidence to guide formulary decision-making”...”At the same time, we strongly oppose misuse of subjective, one-size-fits-all cost effectiveness thresholds to deny patient access to life saving treatment options,"..."As many stakeholders have noted, blunt cost-effectiveness thresholds ignore what individual patient and providers value and conflict with the movement toward personalized, 21st century health care."
- Moody’s: CMS proposed changes to Medicare’s outpatient prospective payment system could hurt hospitals (healthcarefinancenews.com)
...the Centers for Medicare and Medicaid Services' proposed changes to outpatient services, including site neutral clinic visits, 340B policy changes and broadening the list of surgeries covered at ambulatory surgical centers, would generally be credit negative and hurt hospital margins.
- Changes include no longer paying more for clinic visits in off-campus hospital or provider-based department clinics compared to a physician's office...
- Proposed changes to the 340B policy could also impact certain hospitals...CMS lowered reimbursement for Part B drugs to the drug's average selling price, minus 22.5 percent from the ASP, plus 6 percent. CMS said this would save Medicare about about $1.6 billion...
- CMS has proposed adding some nonsurgical procedures to those covered at ambulatory surgical centers, which are located off-campus, but are not hospital outpatient surgery centers...
- Exclusive: Pharma sector warns Saudis on German drug curbs (reuters.com)
European and U.S. pharmaceutical associations have waded into a diplomatic row between Germany and Saudi Arabia, warning that ongoing restrictions on German-made drugs could hurt Saudi patients and dampen future investment in the kingdom...In a strongly worded letter to Crown Prince Mohammed bin Salman....he associations highlighted the level of concern in Germany and elsewhere about restrictive procurement measures implemented by Riyadh in response to criticism of its policies...Germany’s foreign minister at the time, Sigmar Gabriel, condemned “adventurism” in the Middle East in comments perceived by some as an attack on increasingly assertive Saudi policies. Riyadh dismissed the comments as “shameful” and withdrew its ambassador to Germany...“For the past six months German healthcare companies have been having trouble doing business in Saudi Arabia,” said Oliver Oehms of the German chamber of commerce and industry in Riyadh. “It is not a general boycott, but the healthcare sector is clearly suffering.”...Saudi Arabia is the largest pharmaceuticals market in the Middle East and Africa, with sales of $7.6 billion last year, according to healthcare information company IQVIA...With a growing burden of chronic diseases tied to a more Western lifestyle, Saudi Arabia’s overall drugs market is growing at 10 percent a year and the tender sector is expanding by about 30 percent.
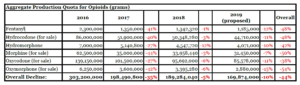
- Trump administration proposes production quota cuts for six opioids (reuters.com)Proposed Aggregate Production Quotas for Schedule I and II Controlled Substances and Assessment of Annual Needs for the List I Chemicals Ephedrine, Pseudoephedrine, and Phenylpropanolamine for 2019 (justice.gov)
The Trump administration on Thursday proposed that U.S. drugmakers cut production quotas of the six most abused opioids by 10 percent next year to fight a nationwide addiction crisis....the U.S. Justice Department and Drug Enforcement Administration said the proposed cut would be in keeping with President Donald Trump’s effort to cut opioid prescription fills by one-third within three years...
- Icahn raises white flag in Cigna-Express Scripts tiff (biopharmadive.com)Express Scripts Reminds Stockholders to Vote "FOR" the Merger with Cigna (bizjournals.com)
Activist investor Carl Icahn has given up his fight to block Cigna's proposed $67 billion acquisition of Express Scripts. Icahn just last week warned shareholders that the deal was among the "worst acquisitions in corporate history" in an open letter...Cigna responded to Icahn's criticism with a lengthy defense. In its response, Cigna called Icahn's opposition "misguided and short-sighted" and said it "demonstrates a clear lack of understanding of the dynamics of the healthcare industry."...Icahn had raised three primary arguments against the acquisition in his open letter to Cigna shareholders: The insurer is paying too much, it would be diving directly into a risky sector embroiled in a political debate over drug pricing, and would soon have to face off with Amazon, which is gearing up to take the industry by storm...Instead, the activist investor proposed an alternative to the acquisition: That Cigna engage in a multi-year partnership with Express Scripts until those regulatory and competitive concerns are ironed out.