- Pfizer joins DOJ probe into claims pharma bribes funded Iraqi terrorists (fiercepharma.com)Veterans' lawsuit claims Big Pharma bribes in Iraq helped finance terrorism (fiercepharma.com)Roche, Johnson & Johnson pulled into Justice Department probe of alleged terrorist bribes (fiercepharma.com)
Pfizer has joined three of its Big Pharma peers in a Department of Justice probe examining allegations that the companies paid bribes to a terrorist-run health ministry in Iraq...The Justice Department's inquiries stem from a lawsuit, filed last fall, in which veterans and their families accused Pfizer, AstraZeneca, Roche and Johnson & Johnson of paying bribes to win business from the Iraqi ministry of health at a time when the ministry was controlled by terrorists...The suit alleges the companies paid bribes to terrorists that "openly controlled the Iraqi ministry in charge of importing medical goods." The plaintiffs contend the drug companies "obtained lucrative contracts from that ministry by making corrupt payments to the terrorists who ran it."
- Washoe County School District prepares to renew health insurance contract with Renown; Saint Mary’s contests the process (thenevadaindependent.com)Washoe County school board delays renewing Renown contract after legal concerns raised about pursuing alternatives (thenevadaindependent.com)
The Washoe County School District is poised to renew a contract...with Renown Hospital and its insurance arm to provide health care to teachers and other district employees, but the hospital’s biggest competitor, Saint Mary’s Regional Medical Center, is crying foul over the process, saying it can provide care more cost-effectively and wasn’t given a fair shot to prove it can do so...Saint Mary’s Health Network, which includes the hospital, other health-care facilities and networked providers, submitted a proposal to the school district earlier this summer for a comprehensive health plan that it says could have saved the district anywhere between $5.4 million and $15.9 million in 2019. But the school district rejected the proposal as “non-responsive” because the district had only requested information on hospital services, not a full health plan, adding in a letter that it only sent out the bid in the first place in response to “constant badgering and lobbying” by Saint Mary’s.
- FDA OKs first generic under new approval pathway (drugstorenews.com)
This new approval pathway was created to expedite the development and review of a generic drug for products that lack competition...The FDA gave the nod for Apotex’s potassium chloride oral solution...“Today’s approval marks the successful implementation of a new program designed to encourage generic drug development for products with inadequate generic competition,” said FDA commissioner Scott Gottlieb, in a press statement...“The quick implementation of this new pathway is part of our broader effort to foster generic competition and help address the high cost of drugs. So are our efforts to narrow the time it takes for generic drugs to reach the market by reducing the number of review cycles that generic applications typically undergo. This new generic drug application was also approved in its first cycle of review. This approval demonstrates that the competitive generic therapy pathway is efficient and open for business. This pathway is a key step in making safe and effective generic drugs available to patients quickly and ensuring there’s adequate competition so patients have affordable access to the treatments they need,” said Gottlieb.
- Brexit Guidance: MHRA Outlines What to Expect (raps.org)
The UK’s Medicines & Healthcare products Regulatory Agency...explained what pharmaceutical and medical device companies can expect during the period that the Brexit agreement is implemented...During the implementation period, which will end in December 2020, pharmaceutical firms will be able to continue UK batch release testing and Qualified Person certification in the UK, which will be recognized by the EU and vice versa, MHRA said...But one of the setbacks is that for medicines, the UK will not have voting rights in EMA and EU committees during the implementation period, MHRA said...“Marketing authorisation holders and qualified persons for pharmacovigilance will continue to be able to be based in the UK and access EU markets. There will be continued mutual recognition of manufacturing and distribution licences, as well as associated inspections such as good manufacturing practice (GMP),” the regulator added.
- Nevada Medicaid approves policy requiring prior approval after 5 therapy sessions (thenevadaindependent.com)
Nevada Medicaid approved a requirement that therapists receive prior approval before providing more than five therapy sessions to a patient...The new policy, which takes effect on Oct. 1, will require psychologists, therapists and other mental health providers to submit written documentation to the state’s third-party vendor demonstrating the medical necessity of treatment and receive prior approval to continue providing both talk therapy and neurotherapy services after five sessions with a patient. The final policy is a scaled back version of earlier proposals from Medicaid to require prior authorization before the first session or after three sessions, both which received significant pushback from the mental health community over the last few weeks...The policy will only apply to patients enrolled in Medicaid’s fee-for-service program, in which Nevada Medicaid reimburses individual providers for services rendered, and not those who are covered under Medicaid managed care, where the state pays an insurance company a flat fee to provide health services to a patient. About one in four of the 650,000 Nevadans on Medicaid are enrolled in the fee-for-service-program.
- Three chambers of commerce join together to offer new association health plan (thenevadaindependent.com)Clark County chambers offer ‘very decent’ health insurance plans (reviewjournal.com)
Three chambers of commerce in Southern Nevada are banding together to take advantage of a new Trump administration rule that makes it easier for small businesses to collectively purchase health insurance...The Henderson Chamber of Commerce, Latin Chamber of Commerce and Boulder City Chamber of Commerce have partnered under the banner of the Clark County Health Plan Association to offer so-called association health plans to their members...The Department of Labor released a rule in June that loosened the rules on association health plans, exempting them from providing the essential health benefits required under the Affordable Care Act, eliminating restrictions based on geography and allowing companies in different industries in the same region to provide coverage together.
- Albertsons, Rite Aid terminate merger (drugstorenews.com)Rite Aid, Albertsons Call Off Merger Amid Investor Opposition (wsj.com)
Albertsons and Rite Aid on Wednesday evening announced that they had mutually decided to terminate the proposed merger...The termination came a day before Rite Aid was set to hold a special shareholders meeting at which the company has been urging its shareholders to vote in favor of the merger...“Albertsons believes that the strategic rationale of the Rite Aid combination was compelling, including the $375 million of cost synergies and $3.6 billion of identified revenue opportunities,” Albertsons said. “We disagree with the conclusion of certain Rite Aid stockholders and third-party advisory firms that although they acknowledged the strategic logic of the combination, did not believe that Albertsons was offering sufficient merger consideration to Rite Aid stockholders.”...Rite Aid chairman and CEO John Standley said that despite believing in the merits of the merger, it had listened to its stockholders and has committed to moving forward as a standalone company...
- Tuesday’s execution in Nebraska the 1st in US to use fentanyl (reviewjournal.com)
Nebraska is preparing to carry out its first execution since 1997 on Tuesday in a bewildering about-face driven largely by the state’s Republican governor...Carey Moore, is scheduled to be executed at the Nebraska State Penitentiary...with a never-before-tried combination of drugs. Moore was condemned to die for the 1979 shooting deaths of...Maynard Helgeland and Reuel Van Ness Jr., and is one of the nation’s longest-serving death row inmates...The combination of drugs for Tuesday’s execution has never been used to put a person to death, according to the Lincoln Journal Star. Three of the drugs — diazepam, fentanyl and cisatracurium — have never been used as part of an execution protocol. The fourth drug, potassium chloride, has been challenged as having the potential to cause serious pain for the inmate.
- Citing appeals court ‘anarchy,’ Amgen asks SCOTUS to weigh its PCSK9 patent spat with Sanofi (fiercepharma.com)
Amgen made an aggressive legal bid to push Sanofi and Regeneron's rival PCSK9 drug off the market when the two were first duking it out in 2015. It didn't work—but Amgen hasn't given up. It's aiming to take the battle to the U.S. Supreme Court instead...After falling short with its arguments in federal appeals court, Amgen is asking the Supreme Court to strike up a review of the case. Amgen claimed Sanofi and Regeneron stepped on its PCSK9 patents and initially won an injunction against their drug Praluent...Amgen now argues that the U.S. Court of Appeals for the Federal Circuit incorrectly interpreted and ruled on patent law. The company says the court has created its own standard that has resulted in “jurisprudential anarchy.” Amgen markets the PSCK9 cholesterol drug Repatha in a head-to-head battle with Praluent; both products have fallen short of expectations, mostly because payers limited access to the high-priced drugs.
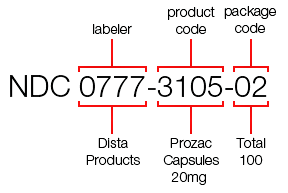
- FDA Anticipates Need for Expanding National Drug Code, Calls for Industry Input (raps.org)
The Food and Drug Administration announced...it plans to hold a public discussion on expanding the National Drug Code format and the impact on industry...FDA currently assigns 10-digit NDCs for the purposes of identifying drugs in the country and the format is comprised of three codes regarding the labeler, the product and the package...“FDA anticipates that it will run out of 5-digit labeler codes in approximately 15 years,” the agency wrote in the notice on the 5 November public hearing. “FDA will begin assigning 6-digit labeler codes at some point in the future due to exhaustion of 5-digit labeler codes.”...Prior to implementing the new formats for longer configurations, FDA officials will seek feedback from meeting participants on the potential impact on companies’ bottom line and drug safety, as well as “issues associated with the current lack of NDC uniformity in the marketplace.”