- Evidence to support ‘breakthrough’ drugs often very limited: study (reuters.com)
The 46 medicines given approval through 2017 as part of the Food and Drug Administration’s Breakthrough Therapy program have often been sent to patients without a large double-blind study, direct measurement of benefit, or comparison with a placebo or existing treatment, according to a new analysis...“FDA approval of these breakthrough therapies is generally based on shorter and smaller clinical trials than those that support FDA approval of non-breakthrough therapy drugs,” coauthor Dr. Joseph Ross....expediting drug approvals raises concerns that important safety or effectiveness information will be missed, potentially heightening risk of patient harm...The study, published in the Journal of the American Medical Association, did not examine whether the drugs were actually as effective and safe as pre-approval testing suggested. In some cases the FDA doesn’t require such post-marketing studies for “breakthrough” drugs...people, when they hear ‘breakthrough designation,’ assume the ‘breakthrough’ is based on great clinical trial evidence but it’s actually more of an expectation that this is promising...My expectation is that this is what the public and clinicians (and Congress!) wants - more novel therapies coming to market as quickly as is reasonably possible, while still assuring drug safety and efficacy...questions persist on whether a larger trial will produce a different result, whether the benefit will fade as treatment continues, whether a drug will prove to be dangerous over the long term and whether a drug will really make a difference in long-term quality of life or survival...
- US prison ‘fraudulently’ obtained sedative for lethal injection, claims Alvogen (in-pharmatechnologist.com)
Alvogen has won a court order preventing the Nevada Department of Corrections from using its sedative drug product, midazolam, in an execution...A temporary restraining order has blocked the Nevada Department of Corrections from using Alvogen’s midazolam in an execution due to take place yesterday (7/11). The drug was intended to be used as a sedative, prior to delivery of the lethal injections...Alvogen had claimed the Department fraudulently obtained the drug. “Alvogen does not accept direct orders from prison systems or departments of correction,”...“Alvogen also works with its distributors and wholesalers to restrict any sale, either directly or indirectly, of our midazolam product to any prison system or department of corrections,”...Alvogen “does not condone the use of any of its drug products, including midazolam, for use in state sponsored executions,”...
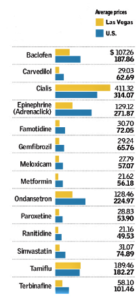
- Las Vegas fares well in prescription drug price comparison (reviewjournal.com)Here are the Most, and Least, Expensive Cities for Prescription Medications (goodrx.com)
Las Vegas ranks near the bottom in many health care categories, but this time it’s a good thing...The prescription drug-tracking website GoodRx rated Las Vegas one of the least-expensive metropolitan areas in the country for out-of-pocket prescription drugs in a new report...The reason for the city’s No. 9 ranking is unclear...Las Vegas’ prices were 9.4 percent below the national average. Columbus, Ohio, had the lowest prices, with drugs costing 21.7 percent below the national figure on average...In some cities, higher cost of living corresponded to higher prices, but not so in Las Vegas, where the Council for Community and Economic Research reports the cost of living to be 2.5 percent above the national average...Leiana Oswald, associate professor of pharmacy practice at Roseman University of Health Sciences in Henderson, said tools like GoodRx can be useful for consumers and pharmacists. She said customers should also get in the habit of asking for coupons at the pharmacy counter, checking for discounts through the drug manufacturer and comparing a drug’s cash price to the cost after insurance...“I think that the best thing that patients can do is to be that squeaky wheel, to ask those questions,” she said.
- EMA Recommends First CART-T Cell Therapies (biopharminternational.com)
...the European Medicines Agency recommended Novartis’ Kymriah (tisagenlecleucel) and Kite Pharma’s Yescarta (axicabtagene ciloleucel), chimeric antigen receptor (CAR) T-cells therapies for blood cancer, for approval in the European Union...Kymriah and Yescarta are the first CAR T-cell treatments to be recommended by the agency. In August 2017, Kymriah became the first CAR-T therapy approved by FDA in the United States, with Yescarta becoming the second in October 2017...Both drugs are also the first treatments supported through EMA’s Priority Medicines scheme to receive positive opinions from the Committee for Medicinal Products for Human Use...
- EMA Report: Clinical Data Published on 50 Medicines in One Year (raps.org)European Medicines Agency Clinical Data: Online Access to Clinical Data for Medicinal Products for Human Use (clinicaldata.ema.europa.eu)Clinical data publication ( Policy 0070) report Oct 2016 - Oct 2017 (ema.europa.eu)
The European Medicines Agency...published its first report on implementing its policy on the publication of clinical data whereby researchers, academics and others can access data from clinical reports submitted by pharmaceutical companies to EMA for new medicines as of 1 January 2015...The 27-page report covers one year from the launch of EMA’s clinical data website on 20 October 2016, and lists the 50 medicines for which clinical data were published...EMA is the only regulatory authority to provide open access to clinical data submitted by companies in support of their marketing authorisation applications...The report unveils the documents published, the amount of commercially confidential information redacted and the anonymization techniques used...EMA accepted about one third of CCI redactions proposed by pharmaceutical companies, though only 0.01% of 1.3 million pages published contained CCI redactions...anonymization techniques to protect personal data, the report suggests conducting a “proper assessment” of the impact of the anonymization technique on data utility and improving the quality of the anonymization reports...
- FDA to Consumers: Stay Away from Maximum Powerful (ptcommunity.com)
The Food and Drug Administration is advising consumers not to purchase or use Maximum Powerful, a product promoted for sexual enhancement that contains sildenafil, the active ingredient in Viagra. This product was identified during an examination of international mail shipments...The announcement is part of the agency’s effort to inform the public of a growing trend of dietary supplements or conventional foods with hidden drugs and chemicals. The culprit products are typically promoted for sexual enhancement, weight loss, and bodybuilding and are often represented as being “all natural.”...the FDA noted that it is unable to test and identify all products marketed as dietary supplements that have potentially harmful hidden ingredients. As a result, the agency issued a blanket warning that consumers should exercise caution before purchasing any product touted as improving sexual enhancement, helping with weight loss, or building up muscle...
- What Does a Drug Cost? It Depends on Where You Live. (nytimes.com)Here are the Most, and Least, Expensive Cities for Prescription Medications (goodrx.com)
A new analysis finds the price of certain drugs varies widely depending on what city they are sold in...Thomas Goetz, the chief of research at GoodRx, said many factors are likely playing a role, like the prevalence in some areas of big-box stores like WalMart and Costco, which sell generic drugs at cheap prices...But that can’t entirely explain what’s going on. Much can still be chalked up to the “drug prices make no sense” theory, he said. Generic drug manufacturers often charge different prices for versions of the same drug, and pharmacies can then mark up the drug in a variety of ways...The study, which looked at 500 commonly used drugs in 30 American cities...differ significantly from coast to coast...
...average cash prices are at pharmacies in five major cities - Paroxetine - Generic for Paxil, used to treat depression
- Birmingham — $50.53
- Boston — $47.34
- Columbus — $20.87
- New York — $73.55
- San Francisco — $53.78
- FDA looks at incentives, manufacturing ‘interventions’ to solve drug shortages (fiercepharma.com)
The FDA will consider creating a list of “essential drugs” and financial incentives to drugmakers that manufacture them as it continues to grapple with the drug shortages that plague U.S. hospitals and caregivers...FDA Commissioner Scott Gottlieb, M.D...announced the formation of a new Drug Shortages Task Force and named Keagan Lenihan, the FDA’s associate commissioner for strategic initiatives to lead it...“I’m charging the shortages task force to delve more deeply into the reasons why some shortages remain a persistent challenge,” Gottlieb said. “The charge to this new task force is to look for holistic solutions to addressing the underlying causes for these shortages.”...The FDA is already taking steps to support new technologies that can improve manufacturing and help reduce the chance that supply disruptions will occur...the FDA may want to consider “more significant interventions” than it currently employs. “We want to make sure we aren’t discouraging investment for manufacturing drugs that are more likely to go into shortage, and thus working against our own goals." One suggestion is to grant the FDA authority to require applicants of certain drugs to conduct a “risk assessment to identify the vulnerabilities in their drug supply that could cause a shortage," and to establish risk mitigation plans in advance to address those weaknesses...
- Pfizer Caving to Trump Has Wall Street Asking Who’s Next (bloomberg.com)Sen. Wyden probes 'secret, sweetheart' deal between Pfizer, Trump (fiercepharma.com)
Pfizer’s decision to delay planned drug price hikes after President Trump attacked the company on Twitter may cause another shakeout throughout the health-care industry. Analysts at Goldman Sachs, Morgan Stanley and SunTrust expect other drugmakers will have to show more restraint on price increases, and say drug distributors may also find themselves under pressure. Height Capital Markets earlier cautioned about the risk to pharmacy-benefit managers, saying that Pfizer’s move ups the ante for the White House to propose policy changes on the use of rebates...“Drug stocks will not react favorably to this news, given the chilling effect this will likely have on others looking to take price increases. On the one hand, the price increases taken in July are only a small number of increases taken over the past year or several years, so the impact of the rollback to the healthcare system is insignificant in the big picture. However, the impact on the broader [dialogue] is much larger.”
- Drugmakers try evasion, tougher negotiations to fight new U.S. insurer tactic (reuters.com)
In the escalating battle over U.S. prescription drug prices, major pharmaceutical companies are scrambling to limit the economic damage from a new U.S. insurer tactic that coaxes patients away from expensive drugs...The latest move by insurers - which effectively forces drug companies to pay more to assist patients with their copays - is causing a decline in real U.S. drug prices this year, and is expected to become more widely adopted...Drugmakers are working on ways to counter copay accumulator programs, fearing that more employer health plans will adopt them...They include new payment options to evade detection by the pharmacy benefits managers so that a patient still benefits from the financial aid...Larger drugmakers may have the financial flexibility to monitor how these accumulator programs affect revenue over time, while those reliant on a small number of drugs may not be able to wait it out...Drugmakers are also taking a tougher stance when negotiating prices or new discounts for payers, according to insurance industry executives and pharmaceutical consultants...