- Big pharma, big data: why drugmakers want your health records (reuters.com)
Drugmakers are racing to scoop up patient health records and strike deals with technology companies as big data analytics start to unlock a trove of information about how medicines perform in the real world...Studying such real-world evidence offers manufacturers a powerful tool to prove the value of their drugs...Real-world evidence involves collecting data outside traditional randomized clinical trials, the current gold standard for judging medicines, and interest in the field is ballooning...The ability to capture the experience of real-world patients, who represent a wider sample of society than the relatively narrow selection enrolled into traditional trials, is increasingly useful as medicine becomes more personalized...It’s getting more expensive to do traditional clinical trial research, so industry is looking at ways it can achieve similar goals using routinely collected data...The thing that has made all this possible is the increasing digitization of health records...Food and Drug Administration...believes more widespread use of real-world evidence (RWE) could cut drug development costs and help doctors make better medical choices...Under the 21st Century Cures Act, the FDA has been directed to evaluate the expanded use of RWE...As the breadth and reliability of RWE increases, so do opportunities for FDA to also make use of this information...
- FDA to Launch New Pilot Program for Orphan Designation Requests (raps.org)
With more than 700 orphan designation requests last year, the Food and Drug Administration...announced a new pilot program to make the request process more efficient...The pilot will include a new form intended to make the submission process easier for sponsors to complete orphan designation requests, and to make the process more efficient for FDA...FDA also released an on-line tutorial to guide sponsors through the submission process and Gottlieb noted there is a new inter-center consult process to streamline and standardize communications...In addition to the pilot, FDA vowed to eliminate the orphan drug designation backlog, and is planning a public meeting to discuss the scientific and regulatory issues related to cancer treatments that target a tumor’s genetic features rather than its location in the body...“We’ll also consider the appropriate application of orphan incentives to this new paradigm of drug development, and how we apply designations to these indications,”...
- Middlemen Play Gatekeeper in Battle to Curb Soaring Drug Costs (bloomberg.com)
Jeff Bezos is teaming up with two fellow billionaires in what could be a prelude to bigger plans to undercut the dominance of intermediaries in the country’s complex health-care system. Pharma companies are trying to shift the blame onto them for soaring drug prices...In response, the middlemen known as pharmacy-benefit managers, or PBMs, are getting more aggressive with the tactics they use to control costs. Express Scripts Holding Co., CVS Health Corp. and UnitedHealth Group Inc.’s OptumRx are increasingly denying coverage of some drugs, negotiating discounts on others or requiring high co-pays for expensive treatments...“It’s becoming more common that we have to play the gatekeeper role,”...The strategy has had some success. Express Scripts said clients’ drug costs rose 1.5 percent last year, the slowest in more than two decades. For 2018, Express Scripts and CVS each refused to cover about 150 drugs -- typically in cases where they negotiated a better deal for alternatives...The tension between PBMs and drugmakers is likely to intensify for two sets of promising drugs coming soon: treatments that help prevent migraines, and drugs for a little-known but widespread liver disease called NASH...
- FDA’s Scott Gottlieb wants to use funding boost to create a Center of Excellence on Digital Health (fiercehealthcare.com)
The Food and Drug Administration plans to use a proposed $400 million boost in federal funding to focus on a range of innovative approaches to speed the approval of new medical devices and create a new center that would support digital health oversight and address cybersecurity concerns...FDA Commissioner Scott Gottlieb, announced...That would include specific carve-outs planned for a new Center of Excellence on Digital Health and furthering the agency’s ability to use EHR data to evaluate medical devices...The Center for Excellence on Digital Health would oversee a revamped regulatory paradigm created through the FDA’s new software precertification program launched with nine companies in September. But the center would also create a cybersecurity unit to “enhance its ability to coordinate device-specific responses to cybersecurity vulnerabilities and incidents.” Over the past several years, medical device cybersecurity has emerged as particular concern for industry and regulators...Gottlieb also highlighted an expanded effort to integrate real world data into pre-market and post-market reviews of drugs and medical devices. The additional funding would allow FDA to develop analytic tools and pull real-time data out of EHRs associated with at least 10 million individuals across a range of healthcare settings...“Toward these ends, an expanded use of natural language processing for the assessment of information submitted to the agency would be developed in an effort to markedly speed recognition and remediation of emerging safety concerns,” he said. “The effort would cover a broad range of medical products, including drugs, biologics and medical devices.”
- Why Your Pharmacist Can’t Tell You That $20 Prescription Could Cost Only $8 (nytimes.com)
...states across the country are moving to block “gag clauses” that prohibit pharmacists from telling customers that they could save money by paying cash for prescription drugs rather than using their health insurance...Many pharmacists have expressed frustration about such provisions in their contracts with the powerful companies that manage drug benefits for insurers and employers. The clauses force the pharmacists to remain silent...“the pharmacist cannot volunteer the fact that a medicine is less expensive if you pay the cash price and we don’t run it through your health plan.”...In North Dakota, a new law explicitly bans gag orders. It says that a pharmacy or pharmacist may provide information that “may include the cost and clinical efficacy of a more affordable alternative drug if one is available.”...also says that a pharmacy benefit manager or insurer may not charge a co-payment that exceeds the actual cost of a medication...the Pharmaceutical Care Management Association, has filed suit in federal court to block the North Dakota law, saying it imposes “onerous new restrictions on pharmacy benefit managers.”
- A South Texas county drags PBMs into nationwide lawsuit over opioids (statnews.com)
A massive lawsuit over the nation’s opioid crisis has largely ignored an influential group of companies in the prescription drug business...pharmacy benefit managers, these companies secretly make the rules that determine the availability of drugs, and how much patients must pay out of pocket to get them...a community in South Texas is calling PBMs into the fight, filing what its lawyers believe is the first case in the country to name these companies as defendants in a municipal opioid lawsuit...The complaint...was filed in January. And earlier this month it was absorbed into a larger lawsuit in Ohio that aggregates claims from cities and states across the country. That, in turn, could put these companies on the hook nationwide for settlement dollars and court orders meant to change the way the drug industry operates...The county’s suit includes claims against...Express Scripts, CVS Health, and OptumRx — along with two smaller companies that also operate in South Texas, Prime Therapeutics and Navitus Health Solutions...It also includes fraud, conspiracy, and racketeering charges against major drug manufacturers and wholesale distributors, as do many other complaints filed across the country. But Webb County’s case is unusual because of the legal claims and language directed at PBMs, entities the suit calls “the gatekeepers to the vast majority of opioid prescriptions filled in the United States.”
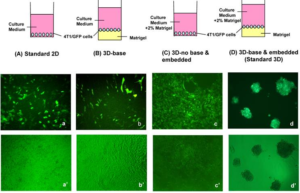
- Cancer research breakthrough shows a better way to predict drugs that will work (cnbc.com)Patient-derived organoids model treatment response of metastatic gastrointestinal cancers (science.sciencemag.org)
Cancer patients need time and drugs that, over time, are effective. They might have more of both according to the results of a new study...The study...showed that the creation of what the researchers are calling microtumors can help predict drug effectiveness in cancer patients better than the current standard method of testing the drugs on rodents. Researchers took biopsies from 71 colorectal cancer patients and made "cancer organoids," or cell culture models of cancerous organs. Researchers then treated these microtumors with drugs and observed the effectiveness in the laboratory...The microtumors grow in a 3-D matrix called a matrigel, and the whole process only takes six to eight weeks...If doctors can more accurately predict what drugs will treat someone's cancer, they can select the right drugs for the right patients. If they know for sure what drugs won't work, they can spare patients the painful side effects..."You can avoid unnecessary toxicity to patients who won't benefit,"..."You can switch to another treatment”...
- EU-US Mutual Recognition Agreement on Inspections: Four Member States Added (raps.org)
The European Medicines Agency...announced that the Food and Drug Administration has confirmed the capabilities of regulators in the Czech Republic, Greece, Hungary and Romania to participate in a mutual recognition agreement focused on drug manufacturing inspections...Under the agreement, FDA can now rely on inspections conducted in 12 member states (the first eight were added in October 2017 – Austria, Croatia, France, Italy, Malta, Spain, Sweden and UK) to replace its own inspections. EMA said that plans are still on track for the mutual recognition agreement to be operational in all member states by 15 July 2019...Mutual benefits under the agreement for both EU and FDA authorities, which has been in place almost exactly a year, include: Avoiding duplicative inspections and reducing the administrative burden and costs, improving the use of inspection resources to focus on manufacturing sites of higher risk, and improving the ability to identify and address potential problems at manufacturing sites before they become a public health risk.
- DOJ to support lawsuits against companies selling opioids (ktvn.com)
The Justice Department said...it will support local officials in hundreds of lawsuits against manufacturers and distributors of powerful opioid painkillers that are fueling the nation's drug abuse crisis...The move is part of a broader effort to more aggressively target prescription drugmakers for their role in the epidemic, Attorney General Jeff Sessions said. The Justice Department will file a statement of interest in the multidistrict lawsuit, arguing the federal government has borne substantial costs as a result of the crisis that claimed more than 64,000 lives in 2016...the Justice Department...which has also sought to crack down on black market drug peddlers and doctors who negligently prescribe...Targets of the lawsuits include drugmakers such as Allergan, Johnson & Johnson, and Purdue Pharma, and the three large drug distribution companies, Amerisource Bergen, Cardinal Health and McKesson. Drug distributors and manufacturers named in these and other lawsuits have said they don't believe litigation is the answer but have pledged to help solve the crisis.
- Amazon breaks into drug sales with Perrigo’s store-brand OTC medications (fiercepharma.com)
While industry watchers were eagerly anticipating Amazon’s move into prescription drugs, the e-commerce behemoth snuck into over-the-counter sales with help from store brand expert Perrigo...Amazon rolled out a line of consumer health products, called Basic Care...The portfolio—consisting of 60 Perrigo-made treatments—sets Amazon up to put the squeeze on retailers of OTC therapies...Amazon already sells branded OTC products...including those from Perrigo’s GoodSense brand...But with Basic Care, Amazon can undercut its competition to reap just a hair in the profit margin column...just because Amazon’s got the lowest prices doesn’t mean consumers will consistently order the Perrigo brands online instead of running out to a Walgreens or CVS when they need something.