- Grocery chain Albertsons to acquire Rite Aid (cnbc.com)
Grocery chain Albertsons announced plans...to acquire Rite Aid, as the traditional grocery industry continues to look for growth by broadening offerings, not just store base...A combined Albertsons and Rite Aid would have a value of roughly $24 billion, including debt. When the deal closes, Albertsons shares will trade on the New York Stock Exchange...Rite Aid will own a 28 percent to 29.6 percent stake in the combined company and current Albertsons shareholders will own a 70.4 percent to 72.0 percent, on a fully diluted basis...The two will have about 4,900 locations, 4,350 pharmacy counters and 320 clinics...Most Albertsons' pharmacies will be rebranded as Rite Aid, and the company will continue to operate Rite Aid's stand-alone stores...The deal with Albertsons underlines the change in course that retailers are taking, no longer looking to expand only by real estate footprint, but also by capability. Increasingly, retailers are looking to pharmacies for this expansion, which can take advantage of the frequency with which people buy prescription drugs. There is also the opportunity to use store footprints as a base for drug delivery and pick up...
- Report: Opioid Manufacturers Gave Millions to Advocacy Groups (ptcommunity.com)
Senator’s investigation found a “lack of transparency” surrounding the donations...A new report from Senator Claire McCaskill found that five opioid manufacturers paid nearly $9 million to 14 outside groups between 2012 and 2017, alleging that the advocacy groups often “amplified messages favorable to increased opioid use.”...The groups—many of which work on chronic pain and other opioid-related issues—lobbied to defeat prescriber limits on opioids...and many criticized facets of 2016 guidelines from the Centers for Disease Control and Prevention that limited the prescribing of painkillers...“The financial relationships between these groups and opioid manufacturers should be clear to the general public,” McCaskill said. “We passed a law ensuring the public had information on payments to doctors by pharmaceutical companies, and I can’t imagine why the same shouldn’t be done in this space.”...
- Gilead’s freshly approved Biktarvy faces patent hurdle (drugstorenews.com)
Gilead Sciences’ new HIV treatment Biktarvy has been approved by the Food and Drug Administration — but its novel component is also the subject of patent infringement litigation from GSK...Biktarvy is a triple-therapy HIV treatment that brings together bictegravir’s unboosted integrase strand transfer inhibitor with the dual nucleoside transcriptase inhibitor backbone of emtricitabine, tenofovir alafenamide — which are the two components of Gilead’s Descovy...However, now bictegravir is at the center of GSK’s lawsuit, which was filed in the U.S. District Court for the District of Delaware, as well as in Canadian Federal Court in Toronto. GSK’s majority-owned Viiv Healthcare said it is looking to prove that Biktarvy’s use of bictegravir infringes on a patent covering its dolutegravir and other compounds that use dolutegravir’s chemical scaffold. The company said it would seek financial redress...
- Rite Aid Says It’s Halfway Home On Store Transfers To Walgreens (forbes.com)
Rite Aid...has now transferred 1,114 stores to Walgreens Boots Alliance as part of a larger deal with the nation’s largest pharmacy chain...In all, Walgreens will over the next few weeks buy 1,932 stores and three distribution centers from Rite Aid for nearly $4.4 billion in cash. Rite Aid...has received “cash proceeds of $2.424 billion, which the company continues to use to reduce debt.”...Before agreeing in September to buy 1,932 Rite Aids, Walgreens had been trying to buy all of Rite Aid before antitrust scrutiny from the Federal Trade Commission that ultimately led to the deal’s undoing...As a smaller pharmacy chain, Rite Aid is focusing on eight states largely on the East and West Coats with plans to invest heavily in its pharmacy benefit manager EnvisionRx, which works with employers and government health programs like Medicare to better control drug costs. PBMs are the middlemen between drug makers and patients when it comes to buying prescription drugs and getting discounts for their customers.
- Pot Pharm: Booming Canada weed sector plots next-wave medicines (reuters.com)
Canopy Growth Corp, one of the world’s biggest medical marijuana producers, now wants to take on the world’s pharmaceutical giants...The Canadian firm launched Canopy Health Innovation in late 2016 to build a portfolio of patented and federally approved cannabinoid-based medicines...aiming to compete with established drugs treating diseases ranging from anxiety and chronic pain to multiple sclerosis and childhood epilepsy...They’re developing research-backed formulations to be sold as pills, inhalers, solutions and creams, with the goal of convincing doctors and insurers to embrace marijuana as a mainstream medicine...Canopy Health and other firms now aim to craft new formulations with varying cannabinoid levels; to find the best dosage delivery systems, such as rapid-release or long-acting tablets or metered-dose inhalers; or to combine cannabinoids with other drugs or supplements to improve effectiveness...Medical marijuana firms have an unusual advantage in that they can continue to sell a legal but largely unregulated medicine as they raise money for research into fully regulated products that might have “enormous potential” in the pharmaceutical market...“We’re allowed to sell a product that many people view as being a possible replacement for many pharmaceutical products,” he said. “But we’re not obligated to take it through the traditional Health Canada approval process.”
- Nordic project takes ‘manufacturing-on-demand’ approach to future drugs (in-pharmatechnologist.com)
Nordic Universities will investigate 3D printing, electrospraying, and microfluidics in an industry supported collaboration aimed at revolutionising production in an age of personalised medicine.
The collaboration, known as Nordic POP (Patient-Oriented Products), will use 35m DKK ($6m) of funding from NordForsk...to create flexible and translational approaches to personalised medicine manufacturing…Jukka Rantanen...University of Copenhagen...said that new patient-oriented and personalised drug products require a “totally new mindset” in the drug development process...“The project team is aiming to create new innovative drug products, where the dose, release mechanism, and size/shape of the product could be easily personalised based on the patient needs...“Instead of one-size-fits-all medication, the potential of new patient oriented products considering gender, age, lifestyle, genetic profile, metabolic capacity, and microbiota will be explored,”... - US OKs medical isotope system that isn’t based on bomb-grade uranium (cnbc.com)
The federal government...approved a device made by a private company...that will allow the first domestic production of a medical imaging isotope...a move the government said would enhance national security by reducing the need to transport weapons-grade uranium...The Food and Drug Administration granted the approval to NorthStar Medical Radioisotopes, which said it would begin delivering systems to make technetium-99...the most common isotope in medicine and is used in 40,000 procedures a day in the United States...consumers have long had to depend on a complicated and risky supply chain for the materials...The current process involves shipping weapons-grade, or highly enriched, uranium from the United States to research reactors in Australia, South Africa and Europe where it is irradiated to make molybdenum-99, which decays into technetium-99..."This is a win for our national security," said Peter Hanlon, an official with the National Nuclear Security Administration office of material management and minimization...
- What Gilead taught pharma about pricing a cure (biopharmadive.com)
Debate over about 'how to price a cure' entered the pharma industry lexicon when Gilead Sciences Inc. priced its highly effective hepatitis C drug Sovaldi (sofosbuvir) at $84,000 per treatment course...The uproar has died down a bit and there's now much less debate about whether Sovaldi was actually cost effective. It has since been shown the drug could save the health system millions by helping to prevent hepatitis C patients from progressing to liver failure...“Gilead bought a launch-ready drug ... and so they launched very quietly,"..."But any analysis that you run shows it's very cost-effective. And I don't think they had to do any discount scheme until after other competitors launched. So one could say that they had a good product and a good story, but they did not tell it."...Gilead...has tried to squeeze every dollar out of the franchise before falling new patient starts and competition erodes away its market. All told, the four antiretrovirals Gilead developed on the sofosbuvir backbone have earned the company roughly $55 billion since Sovaldi's approval in 2013...
- Purdue Pharma to stop promoting oxycodone to U.S. doctors (pharmacist.com)
Purdue Pharma says it will stop promoting oxycodone (OxyContin) and other opioids to U.S. doctors. The company will continue selling the products, but Purdue's sales force "will no longer be visiting offices to engage in discussions about opioid products," the company says. Doctors and other prescribers who have questions about the drugs will have to contact Purdue's medical affairs department, the company says. Purdue is also cutting its U.S. sales force by more than 50%, to about 200 people. The remaining sales representatives will market non-opioid products. Purdue's halting of its opioid marketing comes as the company faces growing legal scrutiny. More than a dozen states and about 400 cities and counties in the U.S. have sued Purdue or other opioid analgesic makers, accusing them of fueling addiction by misrepresenting the risks of their drugs. In response to the suits, Purdue has said it is "dedicated to being part of the solution" to the opioid crisis.
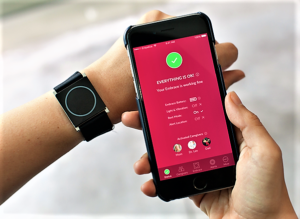
- FDA Clears the First Smart Watch for Use in Neurology (ptcommunity.com)
Wearable device identifies convulsive epileptic seizures and sends alerts to caregivers...The FDA has cleared the Embrace smart watch (Empatica, Inc.) for use by patients with epilepsy. Embrace uses advanced machine learning to monitor for the most dangerous kinds of seizures, known as “grand mal” or “generalized tonic-clonic” seizures, and sends an alert to summon caregivers’ help...The smart watch stands apart from any seizure detection system in that it measures multiple indicators of a seizure. Its unique property is its use of electrodermal activity, a signal used by stress researchers to quantify physiological changes related to sympathetic nervous system activity, also known as the "fight-or-flight" response. Embrace has been approved in Europe as a medical device for seizure monitoring and alert since April 2017.