- Drug chains tumble on reports Amazon eyeing their pie (reuters.com)
Shares of drug retailers Walgreens Boots Alliance Inc, CVS Health Corp and Rite Aid Corp tumbled on Friday after reports that Amazon.com Inc was looking to make a move into selling drugs online...Amazon is reported to be in discussions with mid-market pharmacy benefit managers and has been hiring talent to assess the drug retailing market for its entry…We are convinced that AMZN will almost certainly enter the drug distribution value chain within 2 years, evolving into a more disruptive offering over time...Amazon’s entry into pharmaceuticals has been long rumored in the media...Shares of drug retailers Walgreens closed down 5.8 percent, Rite Aid 4.9 percent and CVS Health 4.9 percent.
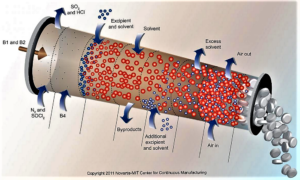
- Continuous Manufacturing: Pfizer, Vertex, AstraZeneca and Others Weigh FDA Plans (raps.org)
The US Food and Drug Administration has been encouraging the adoption of continuous manufacturing techniques...and several companies recently offered the agency some suggestions to refine its work around the developing technology...continuous manufacturing allows companies to move more seamlessly and efficiently...Last week, FDA finalized guidance on how manufacturers can participate in the agency’s program to advance continuous manufacturing...And in a blog post, Michael Kopcha, director of FDA's Office of Pharmaceutical Quality, pointed to Vertex's cystic fibrosis drug Orkambi (lumacaftor/ivacaftor) and Janssen's HIV treatment Prezista (darunavir) as examples of companies successfully using continuous manufacturing after engaging with FDA's emerging technology team...Vertex Pharmaceuticals...noted several contradictions in how batch size is described in an FDA document and sought further clarity and certainty regarding FDA’s understanding and expectations regarding batch size...AstraZeneca...asked if FDA might consider harmonizing the assessment process of the changes relating to continuous manufacturing, including a mechanism by which there could be some mutual recognition across countries participating in the International Council on Harmonization. AstraZeneca also said it "does not see the need" for a full ICH guideline on continuous manufacturing, which the company says FDA has been supporting. But the European drug industry group known as EFPIA is suggesting a Q&A document based on ICH Q8 and AstraZeneca says it "concurs with that approach."
- Medical Technology Is Losing Share Of Venture Investments (forbes.com)
Medical technology continues to lose ground when it comes to all U.S. venture capital investment as value-based care takes hold of the healthcare system and the industry fights to get rid of a device tax...The share of medical technology venture deals dropped to just 4% of total deals last year compared to the industry’s 13% share 25 years ago...There were 420 medical technology venture deals in 2016 out of more than 10,000 total venture deals...medical technology companies had been paying a 2.3% medical device tax on sales under the Affordable Care Act until a two-year moratorium began in January 2016. Before the device tax was put on hiatus, the IRS collected between $1 billion and $2 billion a year in 2013, 2014 and 2015...This move away from fee-for-service medicine to value-based models means insurance companies don't always pay for the medical device a doctor wants to use. Purchasing of devices at large multi-hospital systems has shifted from doctors to “hospital purchasing committees,”.. medtech products...have digital health elements embedded into them...Inherently, digital health solutions are more solution-based...and they have the ability to measure outcomes...Whether medical technology companies can develop products that improve outcomes and measure them will be key...
- Native American tribe moves to dismiss Allergan patent case (reuters.com)
A Native American tribe holding patents for drugmaker Allergan Plc...moved to dismiss a case brought by generic drug company Mylan NV challenging the patents...In a filing to the U.S. Patent Trial and Appeal Board, the Saint Regis Mohawk Tribe asked that Mylan’s case seeking to invalidate Allergan’s patents on dry-eye medicine Restasis be thrown out on the grounds that the board has no jurisdiction over the tribe...The move was expected after Allergan announced on Sept. 8 that it had transferred Restasis patents to the tribe in order to protect them from administrative challenges...the tribe said it is sovereign government that cannot face litigation in an administrative court unless it expressly waives its immunity or the U.S. Congress abrogates immunity...(Allergan) has said the transfer is only intended to shield the patents from review at the patent board, which it has called a flawed forum for patent disputes. Allergan has said it will not invoke tribal immunity in federal courts
- This billion-dollar dry eye drug is behind Allergan’s controversial patent deal (marketwatch.com)With new bill, Sen. McCaskill looks to end 'brazen' tribal licensing strategy (fiercepharma.com)
Last month, drugmaker Allergan announced that it has made a deal with a New York state American Indian tribe to help protect Restasis from competition, using the tribe’s sovereign immunity as a shield against patent challenges. The deal—the first such move by Allergan and, in the pharmaceutical industry, an unusual one at best—has unleashed a firestorm of criticism, including from lawmakers...Originally, patents on Restasis were set to expire in 2014. But Allergan filed more patents, covering the “specific formulation and the method of using” the product, that expire in 2024...Lawmakers and patient advocates, meanwhile, are concerned not only about the implications for Restasis but whether this will set a pharmaceutical industry precedent...The House Oversight committee wrote to Allergan Chief Executive Brent Saunders on Tuesday asking for documents and information regarding the deal...The agreement “may impair competition across the pharmaceutical industry and ultimately dissuade companies from pursuing less-costly generic alternatives to brand drugs,” according to the letter...Allergan said in a statement that it plans to comply with the information requests...patient advocates say they’re worried...Allergan’s sham patent transfer is an effort to circumvent the law to prevent a cheaper generic of Restasis to come to market,” said David Mitchell, the co-founder and president of the non-profit Patients for Affordable Drugs. “Patients will be hurt by this. Patients are being hurt by this. And it’s an outrage.”
- Nevada explores options as Children’s Health Insurance Program expires (reviewjournal.com)
Nevada officials are exploring alternative avenues for insuring more than 26,300 children in the state covered under the Children’s Health Insurance Program if Congress doesn’t reauthorize the program, which is set to expire Saturday...Congress is expected to reauthorize the CHIP program through a bipartisan bill, though that could be weeks away. And with the turmoil in Congress lately, state officials are taking no chances...The Department of Health Care Financing and Policy is working with state partners to identify alternative sources of funding for the program if it is not reauthorized, deputy administrator Cody Phinney said ...“We would have to find other funding sources and we’d have to look at our options for limiting the services that are available, but our first role is to maintain those services,” Phinney said...the federal government pays nearly the entire annual $43 million cost of the program in Nevada...The state has ‘reserve funding’ to operate the health-care program for the next few months. However if Congress does not quickly reauthorize CHIP, states like Nevada will need to either send notices of termination to program beneficiaries or develop alternative funding...
- Moody’s Sees Drug Price Increases Slowing (healthleadersmedia.com)
Despite the slowing rate, rising drug costs and potential changes to Medicare 340B payments for outpatient drugs would further reduce hospitals' margins...Inpatient drug costs will continue to rise for not-for-profit hospitals, but the pace will slow under growing scrutiny of drug makers' pricing practices, Moody's Investors Service said...Drug costs have outpaced hospital revenue growth in recent years...the median growth rate for supply costs, which include drugs, slowed between 2015 and 2016. But the gap between how fast supply costs grew versus (revenue growth) revenues grew widened...Price increases in recent years were extraordinarily high for certain branded hospital inpatient drugs, but drug manufacturers are pulling back on these increases...On the generic drug side, we expect that some of the pressure will ease as the U.S. Food and Drug Administration approves more generic drugs for the first time...The proposed reduction of Medicare Part B outpatient drug reimbursement to 340B hospitals by roughly 30% would represent another headwind for hospitals already facing pressure...
- New Jersey sues Insys as opioid maker settles with Massachusetts (reuters.com)
New Jersey...accused Insys Therapeutics Inc of engaging in a fraudulent scheme to boost sales of a fentanyl-based cancer pain drug, as Massachusetts announced a $500,000 settlement with the drugmaker to resolve similar allegations...The lawsuit by New Jersey Attorney General Christopher Porrino accused Insys of illegally directing its sales force to push prescriptions of Subsys for a broader range of patients than the opioid drug was approved for, and at higher doses...The lawsuit...alleged Insys also paid kickbacks, including sham speaker fees to medical practitioners to prescribe Subsys and defrauded insurers into paying for it...The lawsuit said Insys’ greed put hundreds of lives in jeopardy and led to the 2016 overdose death of a New Jersey woman who was prescribed Subsys to treat fibromyalgia.
- NCPA-commissioned study: DIR fees could cost CMS $3.4B in next 10 years (drugstorenews.com)
Retroactive pharmacy payment reductions, a portion of direct and indirect remuneration fees, could cost the federal government $3.4 billion between 2018 and 2027. That’s according to a new study from Wakely Consulting...This...study is vitally important in showing that DIR legislation will actually save taxpayers $3.4 billion over 10 years without subtracting any benefits seniors currently receive...For pharmacies, banning these after-the-fact fees is the fair way to achieve predictability in the reimbursement for the medications they buy and dispense...The report evaluates the impact of the Improving Improving Transparency and Accuracy in Medicare Part D Drug Spending Act, which has been introduced in both the Senate and House of Representatives and prohibits retroactive pharmacy payment reductions as claims without any defect, impropriety or fraud in Medicare Part D...The legislation is part of NCPA’s efforts to end DIR Fees, which make up a small amount of overall DIR within Medicare Part D, the majority of which is made up of manufacturer rebates
- FDA warns ‘critical’ drug shortages possible after Hurricane Maria battered Puerto Rico (usatoday.com)
Patients could experience "critical shortages" of key pharmaceuticals, the U.S. Food and Drug Administration is warning after Hurricane Maria brought Puerto Rico's drug manufacturing industry to a standstill...The FDA said...it is taking active measures to help redirect production and preserve existing treatments to avoid a ballooning health crisis from Maria's destruction...The agency did not identify any specific medications that could be at risk of a shortfall, and a spokesperson was not immediately available to provide details...But there are "several" cases where "we may soon face critical shortages if we don’t find a path for removal or ways to get production back up and running," FDA Commissioner Scott Gottlieb said in a statement...Some companies are beginning to move product off of the island, and they’ve been communicating with the FDA about that and what potential challenges and limiting factors they see ahead...Drugs made on the island include AstraZeneca's cholesterol treatment Crestor, Abbvie arthritis drug Humira and Johnson & Johnson-owned HIV drug Prezista. Those three companies have said supplies of their drugs are in good shape...the catastrophic storm wiped out electricity for the entire island, devastated telecommunications and made travel nearly impossible for many employees of the island's nearly 50 pharmaceutical factories...