- Many pharma transparency policies are vague, ambiguous, and contradictory (statnews.com)
After several years in which drug makers have been pressured to release clinical trial data, a new analysis finds many companies are still doing an incomplete or inconsistent job of being transparent...95 percent of the 42 companies reviewed – including the 25 largest drug makers, based on sales – had a publicly accessible policy...the specifics often varied wildly in terms of what is disclosed and even how to interpret some of the policies...not every drug maker committed to share results within 12 months of completing a trial...policies were found to be vague, ambiguously worded and internally contradictory...every element of best practice around transparency was committed to by at least one company, [which] strongly suggests that they are all deliverable, and that there are no practical barriers to all companies committing to meet all elements of best practice."...The findings follow years of tussling between drug makers, academics and consumer groups over the issue of transparency. A central concern is the ability for researchers to independently verify study results and, consequently, improve patient treatments that can lead to better health and lower costs...Drug makers, however, are not the only ones to have flaunted transparency...many leading medical research institutions flagrantly violated a federal law requiring public reporting of study results. The violations left gaping holes in a U.S. government database used by millions of patients and medical professionals to compare effectiveness and side effects of treatments.
- A Drug Maker Spends Big in Washington to Make Itself Heard (nytimes.com)Dean Heller-Senate-R-NV : $11,000 (datawrapper.dwcdn.net)
Two federal investigations — were only part of the troubles Mallinckrodt faced as the year began. The stock of the drug maker...was tanking. Wall Street worried that Medicare might reduce the half-billion dollars it was spending yearly on a Mallinckrodt drug with limited evidence of effectiveness...This year, the company left the industry trade group Pharmaceutical Research and Manufacturers of America...after the group threatened to kick out companies that did not spend enough on research...Mallinckrodt, however, has been increasing its spending in another area: It has been writing checks to politicians...After making meager donations in 2015, the company’s political action committee began raising its contributions for congressional campaigns last year. Lawmakers in both the House and Senate collected $44,000 from Mallinckrodt in 2017’s first quarter, nearly nine times what they got from the company in the same period two years ago...Mallinckrodt also spent $610,000 lobbying Congress, triple the amount of 2015’s first quarter...This year, a critical and risky one for drug companies, the industry as a whole is ratcheting up campaign donations and its presence on Capitol Hill...The totals do not include contributions from individual executives and lobbyists, or donations to leadership PACs...Drug companies are hedging their bets, writing checks to individual Democrats and Republicans. With Mr. Trump breaking ranks with Republicans to favor reform, "You can’t tell who’s your friend and who’s not,"…"So you have to go to a ground game — a more one-on-one legislator basis."
- U.S. Proposes Deep Cuts to Some Drug Payments Under Medicare (bloomberg.com)The Booming 340B Contract Pharmacy Profits of Walgreens, CVS, Rite Aid, and Walmart (drugchannels.net)The 340B Program Hits $16.2 Billion in 2016; Now 5% of U.S. Drug Market (drugchannels.net)What I Told HHS Secretary Tom Price About the 340B Drug Pricing Program (drugchannels.net)
The U.S. Medicare program...proposed deep cuts to how much it reimburses hospitals to buy drugs under a policy that gives some health facilities access to cheaper medicines...The proposal...by the Centers for Medicare and Medicaid Services, would change the rate paid to hospitals for drugs under what’s known as the 340B program. The program gives hospitals who serve many poor patients access to deep discounts on some drugs. The new proposal would significantly cut the payments hospitals receive for those medications...Medicare would essentially be clawing back most of the discount from hospitals, (without any impact to payment made to drug manufacturers)…Medicare would pay hospitals for drugs purchased through the 340B discount program at a price more consistent with the actual cost hospitals and other providers pay to acquire those drugs. Seniors would see those savings passed on to them in the form of lower copays...The changes would cut seniors’ drug costs in Medicare by $180 million a year...Currently, hospitals are reimbursed for the drugs at their average sales price, plus 6 percent. The proposed rule would pay average sales price, minus 22.5 percent. The proposal would selectively hurt hospitals serving the poor, said Tom Nickels, executive vice president of the American Hospital Association.
- Computer-Simulated Tests Eyed at FDA to Cut Drug Approval Costs (bloomberg.com)
Computer simulations may get a role alongside human testing as part of an effort to bring new medications and medical devices to market more quickly and cheaply...The...Food and Drug Administration outlined a proposal...to help integrate computer modeling and virtual testing as part of the regulatory approval process for manufacturers -- a step the agency said could save money while helping find cures for puzzling conditions such as Alzheimer’s disease...The average cost of developing a new medication is about $2.56 billion...and much of that goes to fulfilling the FDA’s rigorous demands for proving safety and effectiveness...The price of new technology affects the ability of people to access these new treatments...We therefore need to be mindful of the costs of our regulatory processes, to the degree that these costs also affect the availability of new innovations, and the way that they are ultimately priced…The idea of computer simulated experiments, sometimes called in silico trials, has been around for years but the FDA hasn’t provided guidance to allow the drug industry to use it in testing...The FDA has begun using computer modeling to build databases to help researchers predict how new treatments...will perform. And the agency is developing a family of "virtual patients" for testing new devices…
- As drug prices drop, generics makers fight back with deals (reuters.com)
Generic drug makers are turning to M&A to shield themselves against a concerted effort by U.S. regulators to crack down on steep drug prices...Impax Laboratories Inc, Perrigo Company Plc and Alvogen Inc have been talking to advisers about strategic options for their generics businesses, ranging from acquisitions to increase scale to an outright sale of the units...Mallinckrodt Plc, one of the largest producers of the generic opioid painkiller oxycodone, has been exploring a sale of its specialty generics unit…To bring down prices, the Food and Drug Administration has committed to eliminating the backlog of drug applications awaiting its approval. This could mean nearly 4,000 new medicines will come onto the market over the next several years, based on FDA estimates of drugs awaiting approval...Even before a potential flood of new products, small and mid-sized drug makers were under pressure as consolidation among generic drug distributors has made it less profitable for them to sell their drugs...A merger or a sale to a rival could alleviate some of the pressure through cost-cutting, reduced competition and new markets and products. It could also help companies negotiate better terms with drug distributors...
- Big Pharma Share Buybacks Not Impacting R&D Budgets (forbes.com)Big Pharma Spends on Share Buybacks, but R&D? Not So Much (nytimes.com)
In her Sunday business column in the New York Times, Gretchen Morgenson went after Big Pharma and the propensity for companies to buy back shares of their own stocks. Entitled "When Big Pharma Spends, Research Isn’t No. 1," Morgenson focuses on an academic study that concludes that "from 2006 through 2015, the 18 drug companies in the Standard & Poor’s 500 index spent a combined $526 billion on buybacks and dividends"--an amount that exceeded by 11% the companies’ R&D spending of $465 billion during these years...Morgenson further quotes the authors: "But there is really very little drug development going on in companies showing the highest profits and capturing much of the gains." The implication is pretty clear: drug companies blame high drug prices on the high costs of R&D, but this is a canard. They spend more money on buying back their stock. Needless to say, industry critics have seized on this as yet another reason why the biopharmaceutical industry can’t be trusted. Unfortunately, the article is misleading.
- Big-Data Analysis Points Toward New Drug Discovery Method (technologynetworks.com)Reversal of cancer gene expression correlates with drug efficacy and reveals therapeutic targets (nature.com)
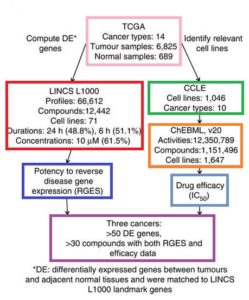
A research team led by scientists at UC San Francisco has developed a computational method to systematically probe massive amounts of open-access data to discover new ways to use drugs, including some that have already been approved for other uses...The method enables scientists to bypass the usual experiments in biological specimens and to instead do computational analyses, using open-access data to match FDA-approved drugs and other existing compounds to the molecular fingerprints of diseases like cancer. The specificity of the links between these drugs and the diseases they are predicted to be able to treat holds the potential to target drugs in ways that minimize side effects, overcome resistance and reveal more clearly how both the drugs and the diseases are working...Our hope is that ultimately our computational approach can be broadly applied, not only to cancer, but also to other diseases where molecular data exist, and that it will speed up drug discovery in diseases with high unmet needs...I’m (Bin Chen, PhD) most excited about the possibilities for applying this approach to individual patients to prescribe the best drug for each...
- Trump’s FDA Commissioner on Drug Prices, Regulations, Science (bloomberg.com)
Trump vs. Big Pharma: Can He Bring Drug Prices Down?...U.S. Food and Drug Administration Commissioner Scott Gottlieb spoke with Bloomberg News about drug pricing, new medicine and regulations. This transcript of the interview has been edited for clarity and length.
- What’s the FDA’s role to play in drug pricing and what can the agency do, given that it hasn’t traditionally had a mandate to address the issue?
- Is it just small, opportunistic drug companies that are "gaming" the system, or is this something bigger companies do as well?
- What about the rest of the administration? Trump talked a lot about drug pricing on the campaign trail and after, yet we haven’t seen much action other than yours.
- What about an executive order on drug pricing -- there was talk that Trump was going to come out with something. Is that still being worked on?
- What about drugs like EpiPen, would you come out with new rules there to create more competition? [EpiPen, made by Mylan NV, is what’s known as a drug-device combination, where both the medicine and the device that administers it can have patent protections.
- But is it reasonable to assume you’re looking at doing something like this, on these types of devices?
- One of the things we’ve seen from the administration is, get rid of regulation, get rid of regulation, get rid of regulation. How does your philosophy as a regulator –- one of the biggest regulators in the U.S. government –- how does that line up with what the Trump administration has called for?
- Some of the things you’re doing to create more competition among drugs, people could interpret as a loosening of standards. I wonder if you think that’s the case?
- Through the years we’ve seen the agency go through cycles, of pushing drugs out into the market faster, versus being much more conservative about safety. Is the balance at the FDA changing?
- How broadly can you apply new standards or guidelines for drug approval? There’s been talk about it in cancer, but what about other diseases?
- EU watchdog concerned drug agency EMA may be too close to companies (reuters.com)
The European Medicines Agency, eager to accelerate access to promising new drugs, may be getting too cosy with the pharmaceutical companies it regulates...That is the concern of the watchdog charged with overseeing administrative irregularities in the European Union, which is launching a strategic inquiry to see if early-stage interactions with firms could influence agency approval decisions...The probe by the European Ombudsman will focus on meetings and discussions between drugmakers and agency officials before a new medicine is formally submitted for regulatory approval...Such "pre-submission activities" have grown in importance in recent years as the EMA has sought to fast-track important new treatments through special arrangements designed to streamline the approval process...Some in Europe, however, fear that moves to accelerate marketing authorizations, including by promoting "adaptive pathways" for clinical trials and designating some products as priority medicines, will increase risks. Critics include Germany's IQWiG, which plays a key role in determining the cost-effectiveness of drugs.
- Updating drug labels would greatly help patients — but few companies do it (statnews.com)
Unfortunately, information contained in the label often lags far behind the evolution of a drug’s use. Drug makers can update product labels to include additional uses when new data supports such modifications. In many cases, though, they don’t do this because there’s little incentive to do so, especially when a drug is no longer being actively marketed or when its patent has expired and it faces generic competition...The problem is widespread...Friends of Cancer Research, found that leading oncologists recommended additional uses beyond those listed on product labels for 79 percent of cancer drugs approved between 1999 and 2011. This means that a large number of well-accepted uses of cancer drugs that emerge over time are completely absent from drug labels...Take the drug methotrexate. Despite being the standard of care for a large number of rheumatologic conditions, the drug label lists only three. When labels are missing critical information like this, particularly regarding diseases other than cancer, insurers often refuse coverage, putting added strain on patients...In the long run, letting labels fall out of date will do a lot of damage by undermining reliance on the drug approval process, which was put in place to protect patients and safeguard public health…