- Lawyers Want the Testimony From OxyContin Company’s Ex-Chief (usnews.com)
A lawyer for one of the country's largest manufacturers of prescription painkillers says the public has "no right of access" to the testimony of its former president about how the company marketed the dangerously addictive OxyContin...The Kentucky attorney general's office sued Purdue Pharma in 2007, accusing it of misleading the public about just how addictive the opioid-based painkiller really was. They said that helped fuel a wave of addiction in Appalachia and beyond that cost the state millions of dollars in health care costs...Former Democratic Attorney General Jack Conway settled the case in 2015 for $24 million with an agreement to keep some court documents hidden from public view. They included a deposition from Richard Sackler, a former Purdue Pharma president and member of the family that still controls the company. His testimony could offer important insights into how much the company knew about the drug's addictive qualities.
- The Benefits of Pharmaceutical Representatives in Health Care (pharmacytimes.com)
I know I am probably going to get some disagreement here and probably some emails (please feel free to!), but I feel that drug representatives have their place in the health care system. It is obvious that their role is to sell the drug they are tasked with selling and as pharmacists we must keep that in mind as we work with them. It is our job to protect and take care of our patients, which is definitely a different goal; however, given the right scenario, it does not have to be a mutually exclusive goal. With that in mind, here are my top reasons I work with and keep in touch with drug representatives:
- If they are selling an innovator or particularly beneficial drug (or vaccine), then both your goal and theirs is to increase the use of that product.
- They can help you with financial assistance programs for their products.
- They can help you when you are in a crunch.
- They can connect you with physician offices.
- They can help market your programs and pharmacy.
- Working Smarter: Establishing an Effective Serialization Architecture (pharmtech.com)Serialization and the Drug Quality & Security Act (pharmamanufacturing.com)
The upcoming serialization requirements in the European Union and the United States have presented pharmaceutical companies with the challenge of balancing data integrity with performance when designing the appropriate information architecture...The pharma sector is currently undergoing the process of introducing new systems and processes for serialization...Companies offering networks for the storing and sharing of vast amounts of serialization data are challenged with creating a shareable world that is also scalable. For pharmaceutical companies, serialization will require a paradigm shift in IT architecture to deal with the combination of the vast amount of data stored within network databases and the operational processes associated with the upcoming requirements...In an industry as highly regulated and safety critical as the pharmaceutical sector, the integrity of the real world must meet the volume and performance of the virtual world. Cloud networks for serialization must offer security, together with speed and scalability especially with the introduction of new drug traceability requirements across the globe...
- EU court: Vaccines can be blamed for illnesses without proof (ktvn.com)
The highest court of the European Union ruled…that courts can consider whether a vaccination led to someone developing an illness even when there is no scientific proof…The decision was issued…in relation to the case of a Frenchman known as Mr. J.W., who was immunized against hepatitis B in late 1998-99. About a year later, Mr. J.W. was diagnosed with multiple sclerosis. In 2006, he and his family sued vaccine-maker Sanofi Pasteur in an attempt to be compensated for the damage they claim he suffered due to the vaccine. Mr. J.W. died in 2011… the EU's top court said that despite the lack of scientific consensus on the issue, a vaccine could be considered defective if there was "specific and consistent evidence," including the time between a vaccine's administration and the onset of a disease, an individual's previous state of health, the lack of any family history of the disease and a significant number of reported cases of the disease occurring following vaccination.
- NCPA, APA File Brief in Federal Appeals Court in PBMs’ Case Against Arkansas Drug Pricing Transparency Law (ncpanet.org)
The National Community Pharmacists Association and the Arkansas Pharmacists Association have filed an amici curiae brief in the United States Court of Appeals for the Eighth Circuit in support of the state of Arkansas and against a challenge by the Pharmaceutical Care Management Association to Act 900, passed by the Arkansas General Assembly in 2015. The ongoing legal battle has prevented Arkansas from implementing a more transparent system under which pharmacy benefit managers determine generic prescription drug reimbursements to pharmacies. In response, NCPA...issued the following statement:..."The Arkansas legislature approved Act 900 because it recognized that PBMs' secretive approach to determining generic prescription drug reimbursement was unfair to pharmacies and patients. Pharmacies deserve to know what their reimbursement will be for a medication, and that they can at least break-even on what they dispense. That's the kind of essential transparency Act 900 enacted. PCMA seems less interested in fairness and more interested in filling PBM corporations' pockets. They continue to spout specious arguments that would undermine legislators' intent...
- Mylan board survives ouster attempt, but shareholders reject rich pay packages (statnews.com)
Mylan stockholders voted to reelect its board at the annual meeting...rebuffing an unusual and highly public campaign from some top shareholders determined to oust many of the directors, including the CEO...But the company also suffered a rebuke as its shareholders voted against a measure to approve executive compensation. That vote doesn’t force Mylan to reduce its pay packages, but most companies do try to be responsive to such votes. Mylan issued a statement saying it would take the shareholder views into account in designing future compensation packages...The fact that the vote over executive compensation failed is unusual for a company of this size...Usually companies respond by making reforms and we certainly hope Mylan will listen to shareholders and discontinue excessive pay, but they seem to shrug off even the most well-founded criticism…
- Drugmaker Mylan gets boost from unlikely source: coal (reuters.com)
Mylan N.V. is best known for producing EpiPen emergency allergy treatments and generic drugs...But a non-pharmaceutical offering – refined coal – has quietly generated hundreds of millions of dollars of tax credits for the company over the last six years that have boosted its bottom line...Since 2011, Mylan has bought 99 percent stakes in five companies across the U.S. that own plants which process coal to reduce smog-causing emissions. It then sells the coal at a loss to power plants to generate the real benefit for the drug company: credits that allow Mylan to lower its own tax bill...Mylan is one of only a few public companies, and the only publicly-traded pharmaceutical maker, that uses these tax credits...
- ‘Pharma bro’ Martin Shkreli heads into fraud trial (reuters.com)‘Pharma Bro’ defies advice to keep quiet before fraud trial (cnbc.com)
Martin Shkreli, the pharmaceutical entrepreneur vilified as the "pharma bro" for raising the price of a life-saving drug by 5,000 percent, will go on trial...for what U.S. prosecutors called a Ponzi-like scheme at his former hedge fund and a drug company he once ran…The charges that led to his arrest in December 2015...focus on Shkreli's management at Retrophin and the hedge fund MSMB Capital Management between 2009 and 2012...Prosecutors said Shkreli lied about MSMB's finances to lure investors and concealed devastating trading losses from them. They said he paid the investors back with money stolen from Retrophin…
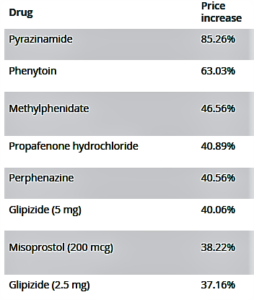
- Why did these generic drugs’ prices jump as much as 85%? (marketwatch.com)
On June 13, as members of a Senate health panel gathered to discuss the rising cost of prescription drugs, the prices of 14 common medications were increased by some 20% to 85%...The blame for rising drug prices once fell squarely on drug makers. But it has shifted at least in part to industry middlemen, with critics saying those companies benefit from high prices and help drive them up. In turn, the middlemen point back to drugmakers, charging that they set the prices in the first place...The corporate blame game can make it hard to determine who is actually benefiting from higher drug prices. June 13’s price increases are a case in point as to why this has become a harder question to answer than it might at first seem...When it comes to drug pricing, AmerisourceBergen is best known as a bellwether for industry trends...competitors including McKesson and Cardinal Health make money based on drug sales and can thus predict how much prices will rise or fall in a given year. And, if they negotiate prices with a pharmacy, they can also play a role in setting prices...when questioned about these increases, executives from drug manufacturers...have pointed instead to pharmaceutical middlemen, which benefit from higher prices, too...But the role of these industry players, including pharmacy-benefit managers...may be shifting out of the regulatory limelight.
- FDA moves to prevent Pharma from ‘gaming’ generic drug system (reuters.com)
The...Food and Drug Administration moved...to prevent pharmaceutical companies from "gaming" the system to block or delay entry of generic rivals…FDA Commissioner Scott Gottlieb said...that the agency plans to hold a public meeting...to identify ways pharmaceutical companies are using FDA rules to place obstacles in the way of generic competition..."We know that sometimes our regulatory rules might be 'gamed' in ways that may delay generic drug approvals beyond the time frame the law intended, in order to reduce competition,"..."We are actively looking at ways our rules are being used and, in some cases, misused."...These obstacles can include limiting the availability of branded products for testing by generic companies, or prolonging negotiations with generic companies over the implementation of shared risk-management programs...